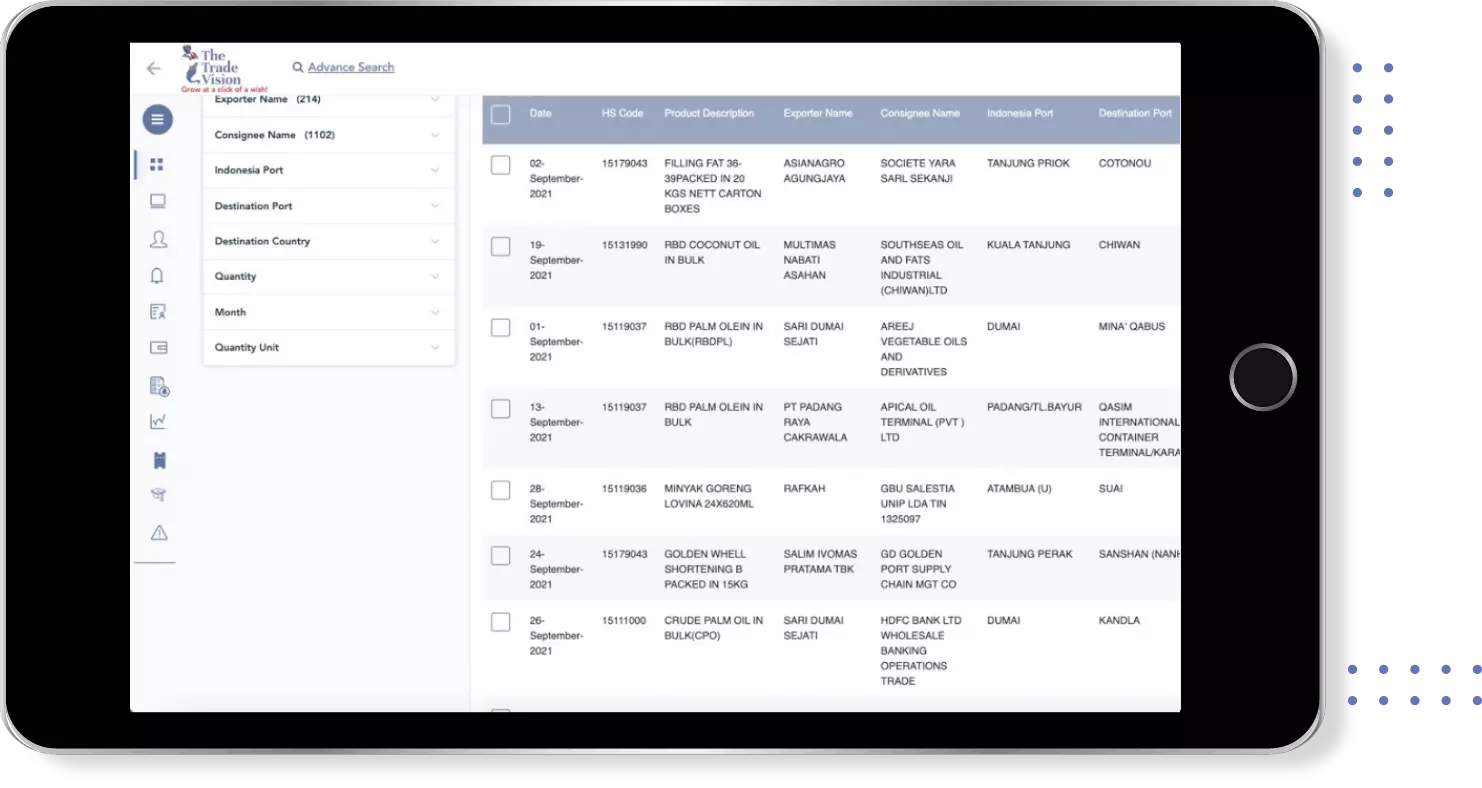
Find verified buyers and sellers of zentel in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- zentel import export data
Overview
- The top 3 supplier countries for zentel are “PAKISTAN” , “UKRAINE” , “ECUADOR” ,
- The top 3 Buyer countries for zentel are “VIETNAM” , “UKRAINE” , “PERU” , .
- Top 3 Product Categories for zentel are 300490, 300420, 481910,
The above summary is based on TTV’s Global Export Import data of zentel, compiled from 180+ countries export import shipments updated till .
Global Buyers of Zentel
TOO GSK KAZAKHSTAN

KAZAKHSTAN
HS-Code
Products
3004900002
1 . ZENTEL TABLET 400MG 1 X1 ON 1 TABLET IN THE BELL PACK. 1 BELL PACK IN A CARDBOARD PACK. S/Y 30. 04. 2027
3004900002
1 . ZENTEL TABLET 400MG 1 X1 ON 1 TABLET IN A BELL PACK. ON 1 BELL PACK IN A PACK FROM CARDBOARD. S/G 31 . 07.2026
3004900002
1 . ZENTEL TABLET 400MG 1 X1 ON 1 TABLET IN THE BELL PACK. ON 1 BELL PACK IN A PACK FROM CARDBOARD. S/G 31 . 1 2. 2026
GLAXOSMITHKLINE PHARMACEUTICAL KENYA LIM

KENYA
HS-Code
Products
3004900000
ZENTEL SUSPENSION 2 PERCENT 1X20ML
3004900000
1X40HR CONTR STC: 28 PALLETS OF 273 120.0EA ZENTEL TABLET 400MG 1X1 - BATCH NOS: CB7K / D48E - MANUF.DATE: 12-JAN-2021 / EXP.DATE: 31-DEC-2025. PO.4502340320.
3004900000
MEDICAMENTS (ZENTEL SUSPENSION 2PERCENT 1X20ML) BATCH NO. X024/5 MFD. 19/20.OCT.2020 EXP. 19/20.SEP. 2023
LLC CURATIO PHARM

UZBEKISTAN
HS-Code
Products
3004900002
1 . ZENTEL TABLETS 400 MG NO 1 (BLISTERS). IN QUANTITY OF 2961 0 PACKS IN 1 24 CARTON BOXES. SERIES: 9F6L TO 01 . 04. 2027. INN: ALBENDAZOLE. MANUFACTURER: GLAXOSMITHKLINE EXPORT LIMITED UK FOR GLAXOSMITHKLINE CONSUMER HEALTHCARE SOUTH AFR
3004900002
1 . ZENTEL TABLETS 400 MG NO 1 (BLISTERS). IN THE QUANTITY OF 6240 PACKS IN 26 CARTON BOXES. SERIES: 3K5L TO 01 . 1 2. 2026. INN: ALBENDAZOLE. MANUFACTURER: GLAXOSMITHKLINE EXPORT LIMITED UK FOR GLAXOSMITHKLINE CONSUMER HEALTHCARE SOUTH AFRIC
3004900002
1 . ZENTEL TABLETS 400 MG NO 1 (BLISTERS). IN THE QUANTITY OF 1 3440 PACKS. SERIES: 6X9C TO 01 . 07. 2026. INN: ALBENDAZOLE. MANUFACTURER: GLAXOSMITHKLINE EXPORT LIMITED UK FOR GLAXOSMITHKLINE CONSUMER HEALTHCARE SOUTH AFRICA (PTY) LIMITED SOUTH AFRICA.
OOO NOVOTEK

UZBEKISTAN
HS-Code
Products
3004900002
MEDICINES FOR HUMANS: 1) ZENTEL TABLETS 400 MG N1 (1 X1) (BLISTERS) INN: (ALBENDAZOLE QTY: 1 4400 SERIES: 3K5L EXPIRY DATE: 01.12.2026 PRODUCTION: GLAXOSMITHKLINE EXPORT LIMITED UK MADE: GLAXOSMITHKLINE CONSUMER HEALT
3004900002
MEDICINES FOR HUMANS1) ZENTEL TABLETS 400 MG N1 (1 X1) (BLISTERS) INN: (ALBENDAZOLE QTY: 8640 SERIES: 3K5L EXPIRY DATE: 01.11.2031 PRODUCTION: GLAXOSMITHKLINE EXPORT LIMITED MANUFACTURED: GLAXOSMITHKLINE CONSUMER HEALTHC
3004900002
MEDICINES FOR HUMANS 1 ) ZENTEL TABLETS 400 MG N1 (1 X1 ) (BLISTERS) INN: (ALBENDAZOLE QTY: 1 2960 SERIES: KL4H EXPIRY DATE: 01.1.2.2026 PRODUCTION: GLAXOSMITHKLINE EXPORT LIMITED UK MADE: GLAXOSMITHKLINE CONSUMER HEALTH
Global Suppliers of Zentel
GLAXOSMITHKLINE EXPORT PANAMA SA

GERMANY
HS-Code
Products
300490910000
MEDICINES FOR HUMAN USE (ZENTEL 4%)
300490910000
MEDICINES FOR HUMAN USE (ZENTEL 4%)
300490990099
MEDICINES FOR HUMAN USE (ZENTEL 4% RAL SUSPENSION BOX X 1 BOTTLE X 1 0ML)
GLAXOSMITHKLINE TRADING SERVICES LTD

GERMANY
HS-Code
Products
300490290000
ZENTEL SUSPENSION 4% 1X10ML|| GSK PHARMACEUTICALS SA
300490290000
ZENTEL SUSPENSION 4% 1X10ML|| GSK PHARMACEUTICALS SA
300490290000
ZENTEL SUSPENSION 4% 1X10ML|| GSK PHARMACEUTICALS SA
GSK TRADING SERVICES LTD

SOUTH AFRICA
HS-Code
Products
300490000000
1X40HR REEFER CONTR STC: 21 PALLETS OF 203 850 EA ZENTEL TABLET 400MG 1X1 - BATCH NO. B88K; MANUF DATE: 12-JAN-2021 - EXP DATE: 31-DEC-2025.
300490000000
ZENTEL TABLET 400MG 1X1 (QTY; 288 240); ZENTEL; PK; 30; 400MG; THERAPEUTIC; 31.
300490000000
1X40HR CONTR STC: 28 PALLETS OF 273 120.0EA ZENTEL TABLET 400MG 1X1 - BATCH NOS: CB7K / D48E - MANUF.DATE: 12-JAN-2021 / EXP.DATE: 31-DEC-2025. PO.4502340320.
GLAXOSMITHKLINE EXPORT PANAMA S A

GERMANY
HS-Code
Products
300490990099
MEDICINES FOR HUMAN USE (ZENTEL 4%)
300490910000
MEDICINES FOR HUMAN USE (ZENTEL 4%)
300490910000
MEDICINES FOR HUMAN USE (ZENTEL 4%)
Global Export Import Trade Data for zentel
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 PAKISTAN PAKISTAN | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SINGAPORE SINGAPORE | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SWITZERLAND SWITZERLAND | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 MALDIVES MALDIVES | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
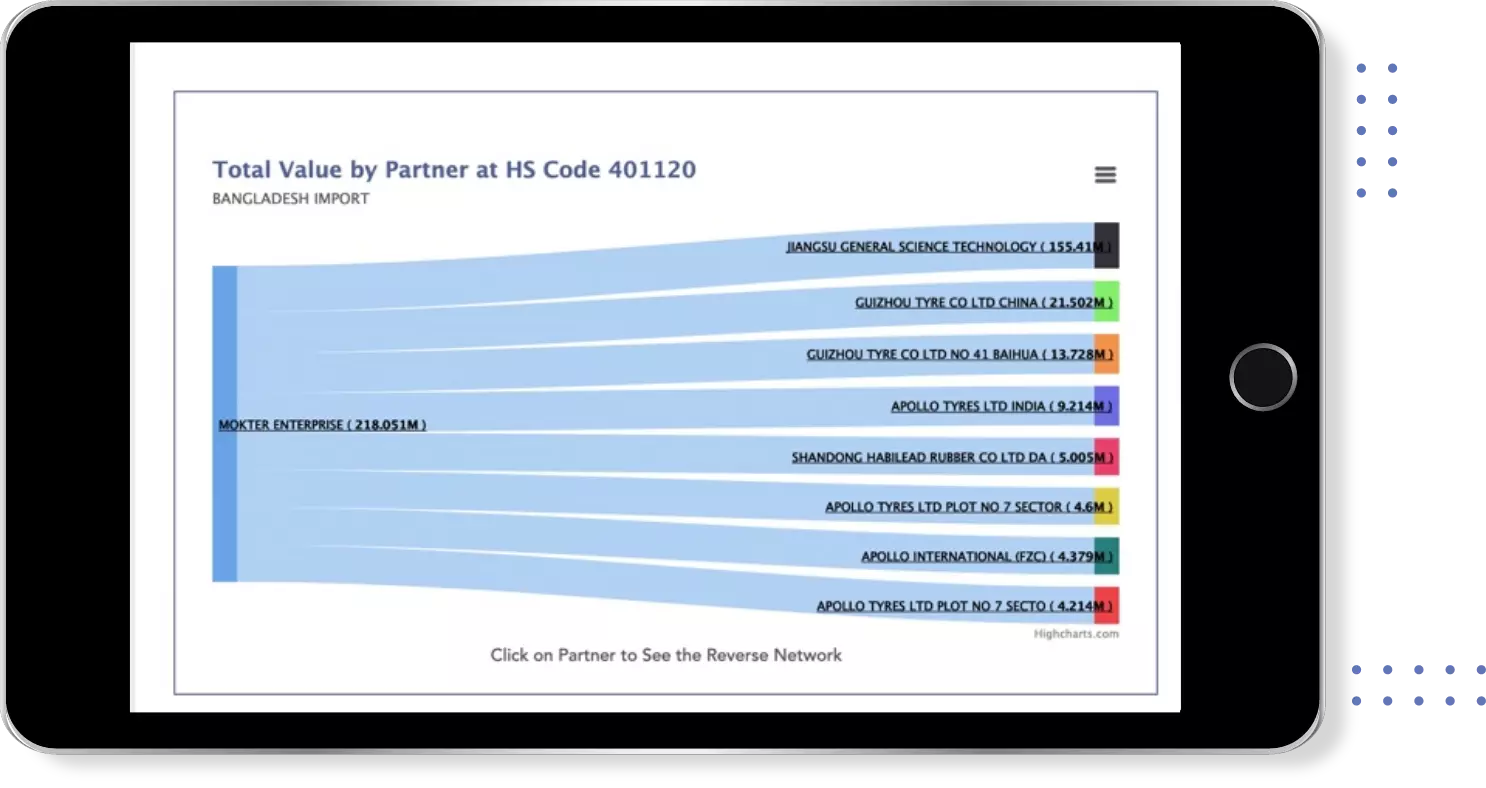
Get most profitable Buyers & Suppliers
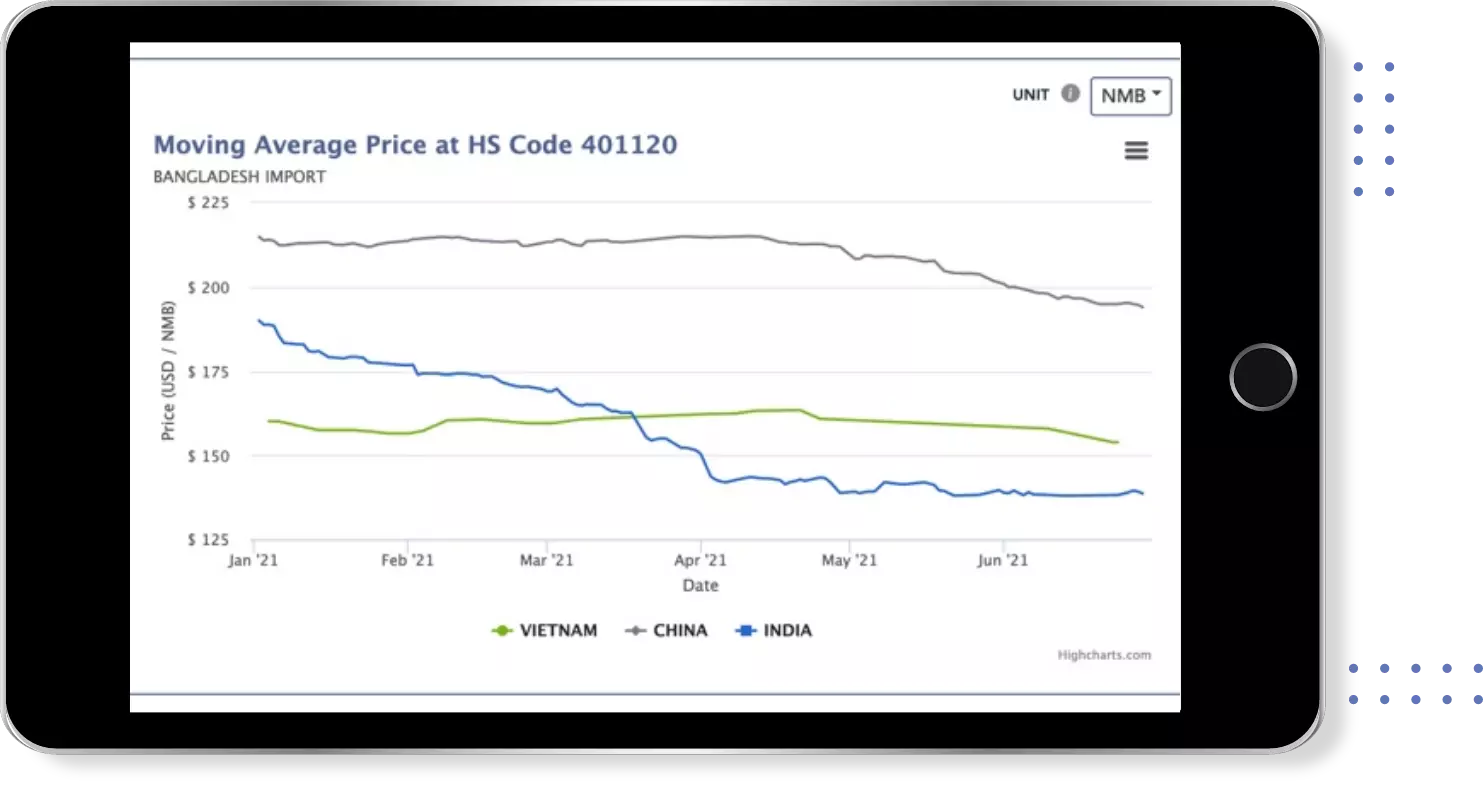
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries