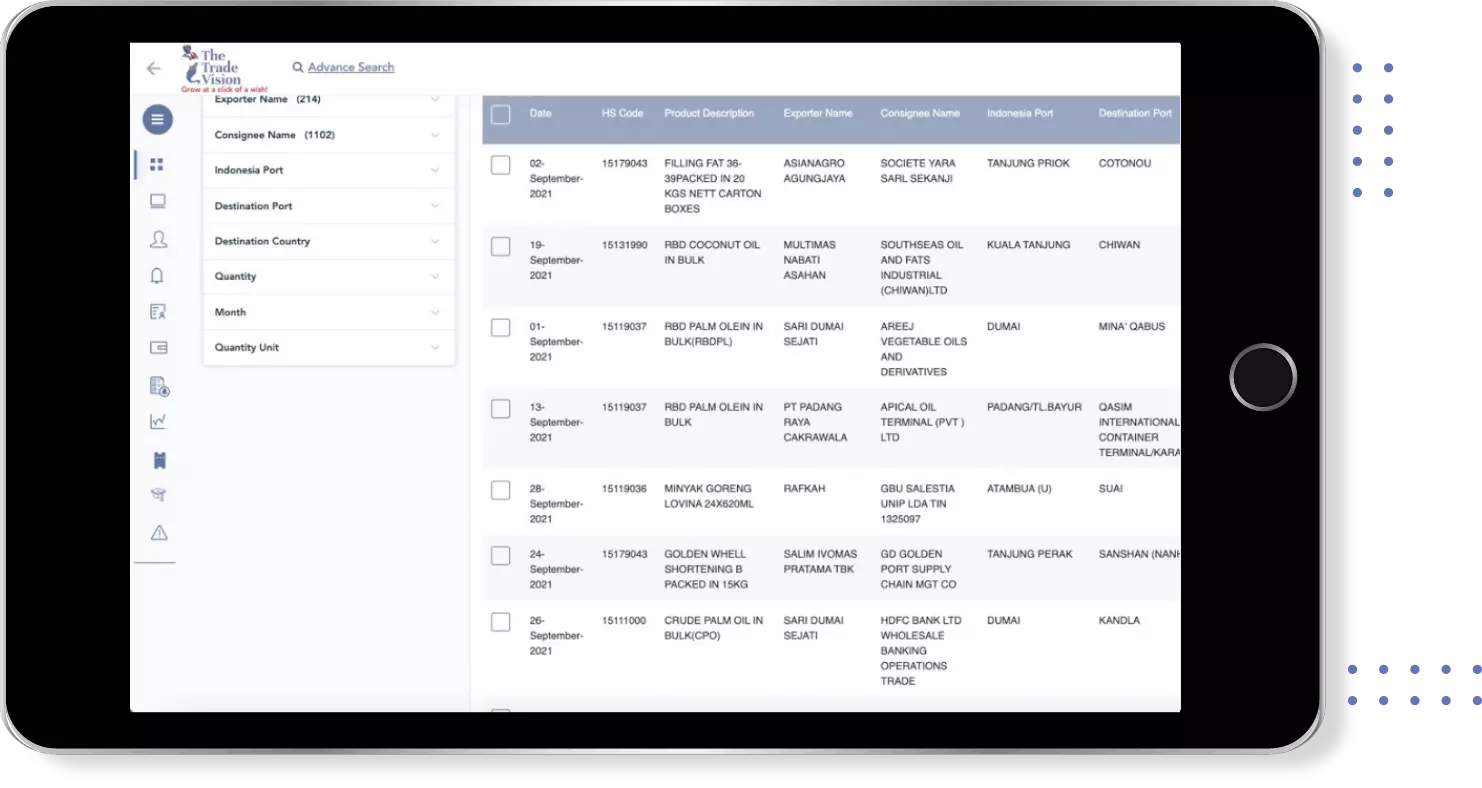
Find verified buyers and sellers of vaccine anti rabies in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- vaccine anti rabies hs code 300241 import export data
Overview
- The top 2 supplier countries for HS Code 300241 are “CHINA” , “FRANCE” ,
- The top 3 Buyer countries for HS Code 300241 are “UZBEKISTAN” , “KAZAKHSTAN” , “VIETNAM” , .
- Top 1 Product Categories for HS Code 300241 are 300241,
The above summary is based on TTV’s Global Export Import data of HS Code 300241, compiled from 180+ countries export import shipments updated till .
- HS Code: 300241
Global Buyers of vaccine anti rabies under HS-Code 300241
LTD PARTNERSHIP KNYAZ PHARMA LTD

KAZAKHSTAN
HS-Code
Products
3002410000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE (CONCENTRATION) 2.5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF THE ACTIVE SUBSTANCES RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVATED WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 03072AY (1 1 880DOZ/PACK. ). DATE PR. - MAR. 01 2021 . EXPIRY DATE DO - FEB. 29 2024 SERIES - 2021 06221 AY(91 055DOZ/PK) SERIES - 2021 06224BX(1 5505DOZ/PK) DATES PR. - JUN. 1 8 2021 . DATES BEFORE - JUN. 1 7 2024. NOT FOR VETERINARY SCIENCE. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
3002410000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE (END) 2.5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF ACTIVE SUBSTANCES RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVE WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 03072AY (79570DOZ/PACK. ). DATE OF PRODUCTION - MAR. 01 2021 . EXPIRY DATE UP TO - FEB. 29 2024. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
3002410000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE 2. 5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF THE ACTIVE SUBSTANCES: RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVE WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 1 0345BY (20725 DOS). PRODUCTION DATE - SEPTEMBER 24 2021 EXPIRY DATE - SEPTEMBER 23 2024 FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
GRAND CROSS MEDICAL LLC

UZBEKISTAN
HS-Code
Products
3002410000
1) RABIES VACCINE FREEZE-DRIED (VERO CELL) FOR HUMAN USE 2.5IU DOSE VIALS NO. 5 0.5ML DILUENT AMPOULES NO. 5 (INN: ANTI-RABIES VACCINE (VERO CELLS)) LYOPHILIZED POWDER FOR INJECTION 2.5 IU NO. 5 (FLASK) SERIES: 202208112AX 0.5 ML SOLUTION INCLUDED. WATER FOR
3002410000
1) RABIES VACCINE FREEZE-DRIED (VERO CELL) FOR HUMAN USE 2.5IU DOSE VIALS NO. 5 0.5ML DILUENT AMPOULES NO. 5 (INN: ANTI-RABIES VACCINE (VERO CELLS)) LYOPHILIZED POWDER FOR INJECTION 2.5 IU NO. 5 (FLASK) SERIES: 202202026AY 0.5 ML SOLUTION INCLUDED. WATER D
OOO MEDBIOLINE

UZBEKISTAN
HS-Code
Products
3002410000
1. RABIES VACCINE (VERO CELL) FOR HUMAN USE FREEZE-DRIED / ANTI-RABIES VACCINE LYOPHILIZED (VERO CELL) BIOSHOOT. DOSAGE FORM: LYOPHILIZED POWDER FOR THE PREPARATION OF INJECTION SOLUTION NO. 5 (BOTTLES) COMPLETE WITH SOLVENT - IN
3002410000
1. RABIES VACCINE (VERO CELL) FOR HUMAN USE FREEZE-DRIED / ANTI-RABIES VACCINE LYOPHILIZED (VERO CELL) BIOSHOOT. DOSAGE FORM: LYOPHILIZED POWDER FOR THE PREPARATION OF INJECTION SOLUTION NO. 5 (BOTTLES) COMPLETE WITH SOLVENT - IN
OOO GRAND CROSS MEDICAL

UZBEKISTAN
HS-Code
Products
3002410000
1) RABIES VACCINE FREEZE-DRIED (VERO CELL) FOR HUMAN USE 2.5IU DOSE VIALS NO. 5 0.5ML DILUENT AMPOULES NO. 5 (INN: ANTI-RABIES VACCINE (VERO CELLS)) LYOPHILIZED POWDER FOR INJECTION 2.5 IU NO. 5 (FLASK) SERIES:: 202210166?? 0.5 ML SOLUTION INCLUDED. WATER DL
Global Suppliers of vaccine anti rabies under HS-Code 300241
LIAONING CHENG DA BIOTECHNOLOGY CO LTD

CHINA
HS-Code
Products
300241000000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE (END) 2.5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF ACTIVE SUBSTANCES RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVE WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 03072AY (79570DOZ/PACK. ). DATE OF PRODUCTION - MAR. 01 2021 . EXPIRY DATE UP TO - FEB. 29 2024. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
300241900000
NEW DRUG (VACCINE): SPEEDA (PURIFIED RABIES VIRUS ANTIGEN L-PASTEUR PV-2061 STRAIN >= 2.5IU). BOX OF 5 VIALS OF LYOPHILIZED POWDER 5 TUBES OF DISTILLED WATER SOLVENT FOR INJECTION X0.5ML. LOT: 202304051AY.SX:03/23.HD:03/26. QLVX-1041-17.
300241900000
RABIO ANTI RABIES VACCINE RABIES ANTIGEN2.5IU WHO SPECIFICATIONS 0.5 ML PER VIAL AFTER RECONSTITUTION WITH DILUENTS 0.5 ML FOC REGISTRATION NO. 59094 5 DOSE PACK PART N
SHENZHEN MELLOW HOPE PHARM INDUSTRIAL CO LTD

CHINA
HS-Code
Products
300241000000
1. RABIES VACCINE (VERO CELL) FOR HUMAN USE FREEZE-DRIED / ANTI-RABIES VACCINE LYOPHILIZED (VERO CELL) BIOSHOOT. DOSAGE FORM: LYOPHILIZED POWDER FOR THE PREPARATION OF INJECTION SOLUTION NO. 5 (BOTTLES) COMPLETE WITH SOLVENT - IN
Global Export Import Trade Data for vaccine anti rabies under HS-Code 300241
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UZBEKISTAN UZBEKISTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 KAZAKHSTAN KAZAKHSTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 CAMBODIA CAMBODIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SINGAPORE SINGAPORE | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 CHINA CHINA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
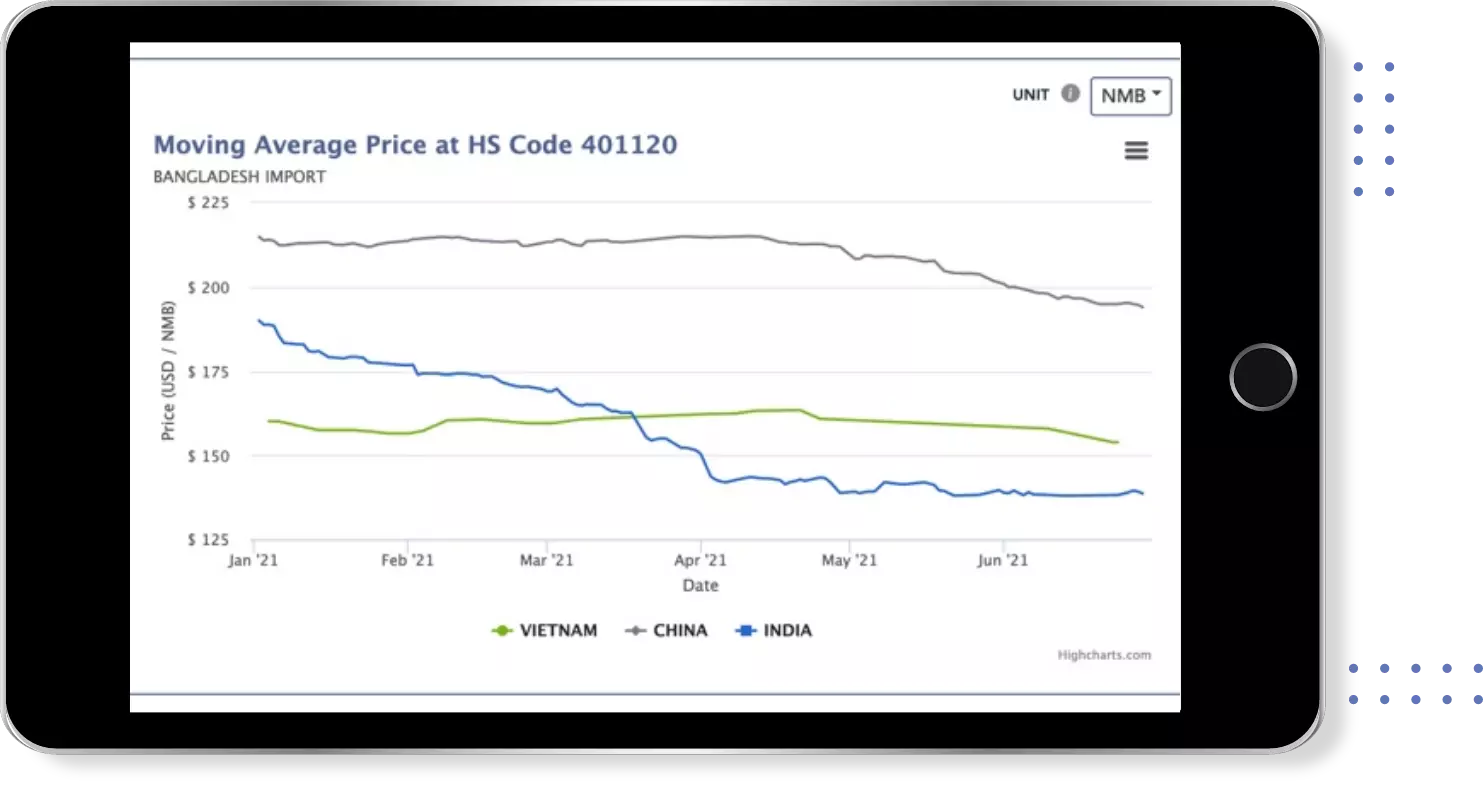
Instant insights on Price , Quantity & Value trends
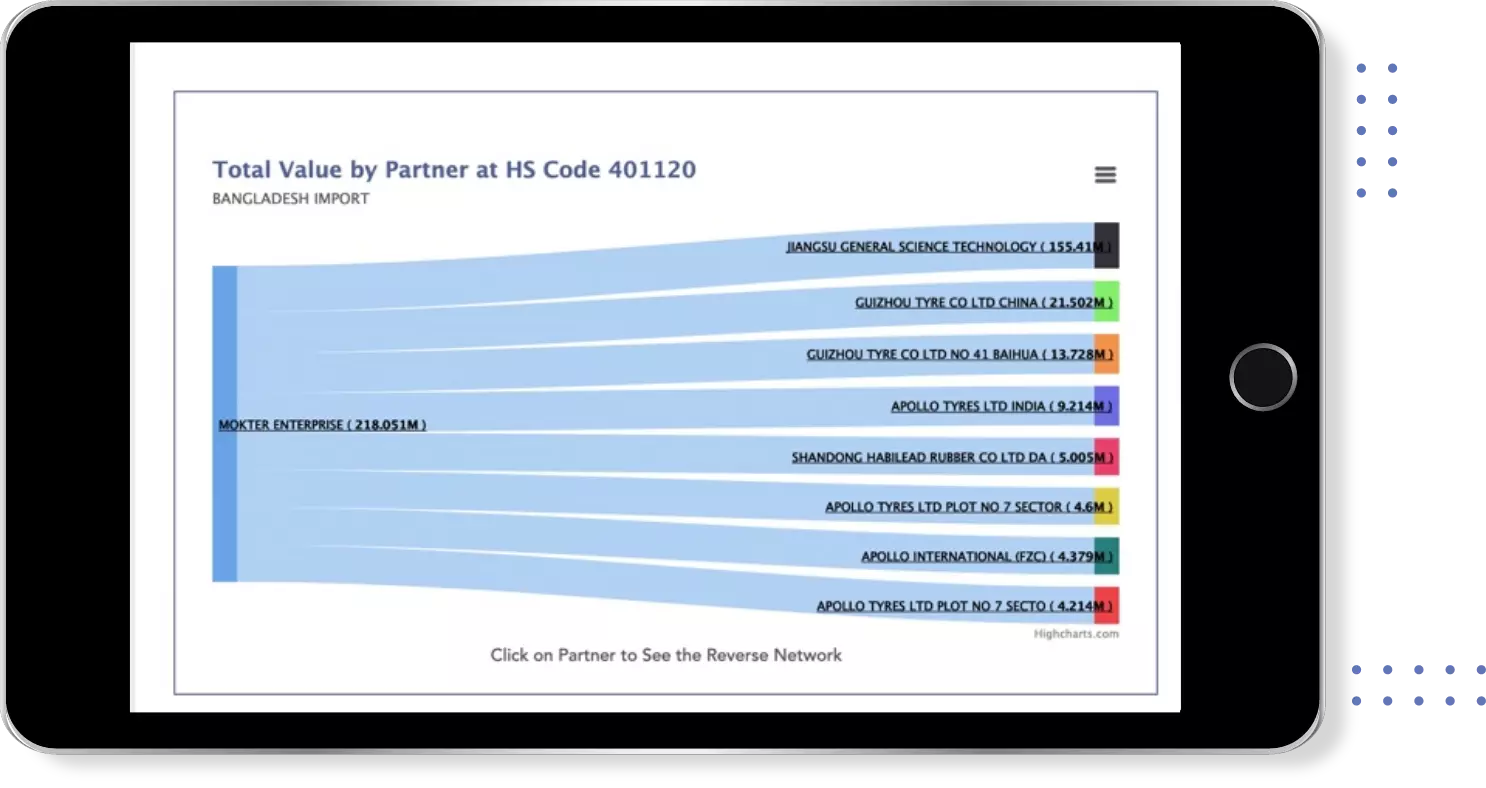
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries