Find verified buyers and sellers of s pantoprazole vial in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- s pantoprazole vial buyers
Global Buyers of s pantoprazole vial
TOO RAUZA ADE

KAZAKHSTAN
HS-Code
Products
3004900002
1 . PROTECTOPASE (PANTOPRAZOLE) 40 MG LYOPHILIZED POWDER FOR PREPARATION OF SOLUTION FOR INJECTION. POWDER IN A VIAL. ON 1 5 10 OR 20 VIALS IN A CARDBOARD PACK. S/Y 1 1 /2023
3004900002
1 . PARASTAMIK (PANTOPRAZOLE) 40 MG LYOPHILIZED POWDER FOR PREPARATION OF SOLUTION FOR INTRAVENOUS ADMINISTRATION. DRUG IN A VIAL. ON 1 VIALS IN A CARDBOARD BOX. S/Y 1 2/2023
3004900002
1 . PARASTAMIK (PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG LYOPHILIZED POWDER FOR PREPARATION OF SOLUTION FOR INTRAVENOUS ADMINISTRATION DRUG IN A VIAL. ON 1 BOTTLE IN A CARDBOARD BOX. S/Y 1 0/2024
IP OOO PHARMACORE LIFE SCIENCES

UZBEKISTAN
HS-Code
Products
3004900002
1. MEDICINES: PANZOVI-I (PANTOPRAZOLE FOR INJECTION 40MG)/PANZOVI-I LYOPHILISATE (POWDER) FOR THE PREPARATION OF SOLUTION FOR INJECTION 40 MG VIALS. INN: PANTOPRAZOLE; SERIES: SD22H008E; PERIOD ONE YEAR UNTIL: 04.2024 - 10200 PACK. MANUFACTURER: LLP SANJARPHARM
3004900002
1 . MEDICINES: PANZOVI-I (PANTOPRAZOLE FOR INJECTION 40MG) / PANZOVI-I LYOPHILISATE (POWDER) FOR PREPARING SOLUTION FOR INJECTION 40 MG VIALS. INN: PANTOPRAZOLE; SERIES: SD22E002E; DEADLINE YEAR UNTIL: 04. 2024 -25500UP. MANUFACTURER: LLP SANJARPHAR
3004900002
1. MEDICINES: PANZOVI-I (PANTOPRAZOLE FOR INJECTION 40MG)/PANZOVI-I LYOPHILISATE (POWDER) FOR THE PREPARATION OF SOLUTION FOR INJECTION 40 MG VIALS. INN: PANTOPRAZOLE; SERIES: SD22H008E; PERIOD ONE YEAR UNTIL: 07.2024 - 20100 PACK. MANUFACTURER: LLP SANJARPHARM
THE PHARMA NETWORK LLP (ZE PHARMA NETWORK)

KAZAKHSTAN
HS-Code
Products
3004900002
1 . PAN IV 40 MG INJECTION (KAZAKHSTAN) EACH VIAL CONTAINS PANTOPRAZOLE SODIUM EQUIVALENT TO PANTOPRAZOLE 40 MG
3004900002
1 . PAN IV 40 MG INJECTION (KAZAKHSTAN) EACH VIAL CONTAINS PANTOPRAZOLE SODIUM EQUIVALENT TO PANTOPRAZOLE 40 MG BATCH NO.
3004900002
1 . PAN IV 40 MG INJECTION (KAZAKHSTAN) EACH VIAL CONTAINS PANTOPRAZOLE SODIUM EQUIVALENT TO PANTOPRAZOLE 40 MG BATCH NO.
LLC OLMOS DUR FARM

UZBEKISTAN
HS-Code
Products
3004900009
1 . MEDICINES IN RETAIL FORMS NOT FOR VETERINARY SECTION: PANTOGAINE FORM: POWDER FOR SOLUTION FOR INJECTION EACH POWDER VIAL CONTAINS: ACTIVE SUBSTANCE: PANTOPRAZOLE SODIUM STERILE MIXTURE PANTOPR EQUIVALENT
3004900009
1 . MEDICINES IN RETAIL FORMS NOT FOR VETERINARY SECTION: PANTOGAINE FORM: POWDER FOR SOLUTION FOR INJECTION EACH POWDER VIAL CONTAINS: ACTIVE SUBSTANCE: PANTOPRAZOLE SODIUM STERILE MIXTURE PANTOPR EQUIVALENT
3004900009
1 . MEDICINES IN RETAIL FORMS NOT FOR VETERINARY SECTION: PANTOGAINE FORM: POWDER FOR SOLUTION FOR INJECTION EACH POWDER VIAL CONTAINS: ACTIVE SUBSTANCE: STERILE PANTOPRAZOLE SODIUM MIXTURE PANTOPR EQUIVALENT
OOO MEROS PHARM

UZBEKISTAN
HS-Code
Products
3004900002
1) NOLPAZA (INN: PANTOPRAZOLE) POWDER FOR SOLUTION FOR INJECTION 40 MG N1 (VIALS) -1 201 5UP. SER. C81 001 C81 000. MANUFACTURER KRKA DD SLOVENIA. : WEIGHT: 304.58KG; 2) TYPE AND QUANTITY OF PACKAGING: 2 PALLETS;
3004900002
1) NOLPAZA (INN: PANTOPRAZOLE) POWDER FOR SOLUTION FOR INJECTION 40 MG N1 (VIALS) -24000UP. 1 34 CARDS COR SER. C80873. PR-L KRKA D. D. NOVO MESTO. SLOVENIA. : WEIGHT: 608.4 KG; 2) TYPE AND QUANTITY OF PACKAGING: 4 PALLETS;
3004900002
1) NOLPAZA (INN: PANTOPRAZOLE) POWDER FOR SOLUTION FOR INJECTION 40 MG N1 (VIALS)-1 2000 UP. 67CARD. KOR. SER. C81001. PR-L. KRKA DD SLOVENIA. : WEIGHT: 304.2 KG; 2) TYPE AND QUANTITY OF PACKAGING: 2 PALLETS;
RIN PHARM LLP (RIN PHARM)

KAZAKHSTAN
HS-Code
Products
3822190009
1 . STANDARD SAMPLE: ULSEPAN TABS 40 MG N7 (WM) - PANTOPRAZOLE SODIUM SESQUIHYDRATE (COMPANY STANDARD) - 200 MG (X 1 VIAL EVEREST) ??WMWS220345
3822190009
1 . REF: ULSEPAN LYOF POWD (MEFAR) - PANTOPRAZOLE SODIUM SESQUIHYDRATE (USP) - 1 80. 44 MG (1 VIAL EVEREST) ??- WMWS220363
3004900002
1 . MEDICINE: ULSEPAN LYOPHILIZED POWDER FOR PREPARING SOLUTION FOR INJECTION 40MG NO. 1 PACKAGING: POWDER IN A VIAL. ON 1 BOTTLE IN BELL PACK. ON 1 BELL PACK IN A CARDBOARD BOX. COMPOSITION OF THE ACTIVE SUBSTANCES: PANTOPRAZOLE SODIUM SESQUIHYDRATE
TOO WELFAR KAZAKHSTAN (WELFAR KAZAKHSTAN)

KAZAKHSTAN
HS-Code
Products
2942000000
1 . REFERENCE MATERIALS OF PHARMACEUTICAL SUBSTANCES FOR LABORATORY STUDIES: SODIUM PANTOPRAZOLE SESQUIHYDRATE BATCH: BPZLE/2009026 EXPIRY DATE: 07.2024 230 MG IN A VIAL
2942000000
1 . REFERENCE MATERIALS OF PHARMACEUTICAL SUBSTANCES FOR LABORATORY STUDIES: SODIUM PANTOPRAZOLE SESQUIHYDRATE FOR CRS SUITABILITY SYSTEM BATCH: 8 5 MG PER VIAL
2934998000
1 . REFERENCE MATERIAL FOR PHARMACEUTICAL SUBSTANCES: PANTOPRAZOLE NARIA SESQUIHYDRATE (SERIES: 1 . 2) IN A VIAL FOR LABORATORY TESTING OF PARASTAMIK
AUROLOGISTICS LLC AUROMEDICS PHARM

UNITED STATES OF AMERICA
HS-Code
Products
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40 MG(PANTOPRAZOLE SODIUM FOR INJECTION 40MG/VIAL -10ML VIAL -)(13950X10'S)
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40 MG(PANTOPRAZOLE SODIUM FOR INJECTION 40MG/VIAL -10ML VIAL )(6756X10'S)
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40 MG/VIAL-10ML VIAL (14200X10'S)
EUGIA US LLC

UNITED STATES OF AMERICA
HS-Code
Products
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40 MG/VIAL (13416X10 S)
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40 MG/VIAL (14088X10 S)
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40 MG/VIAL (13944X10 S)
SUN PHARMACEUTICAL INDUSTRIES INC

UNITED STATES OF AMERICA
HS-Code
Products
300490390000
PLACEBO OF PANTOPRAZOLE SODIUM FORINJECTION 40 MG/VIAL ( BATCH NO: HACT204A (MFG DT.: 10/2021 & EXP DT.:03/2024
300490390000
PLACEBO OF PANTOPRAZOLE SODIUM FORINJECTION 40 MG/VIAL ( 1 BOX CONTAIN 50VIALS=50 NOS)
300490390000
PANTOPRAZOLE SODIUM FOR INJECTION 40MG/VISL PACK BOX10 VIAL BATCH HAD2506AMFD 19.07.2022 EXP 31.12.2023 BATCH
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
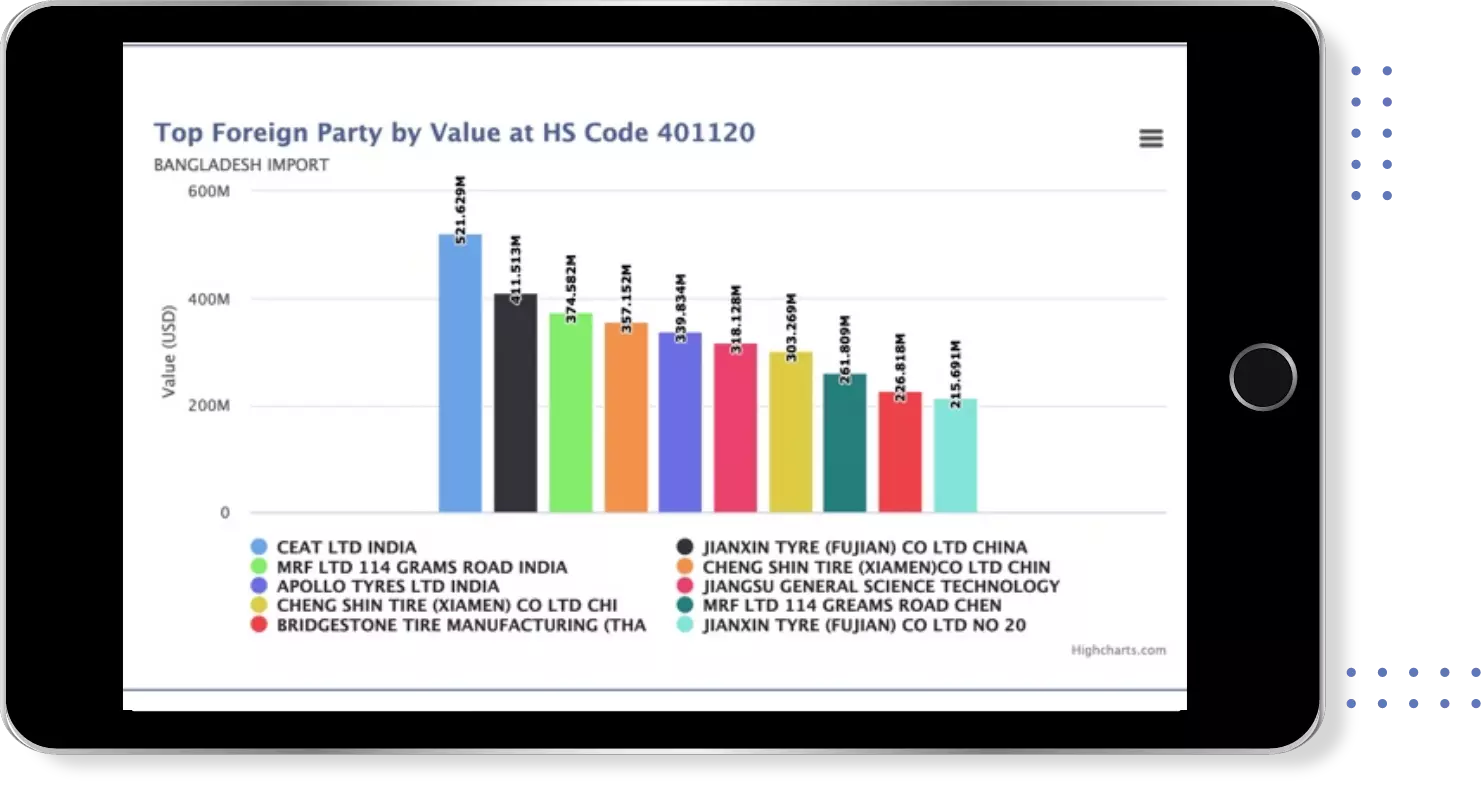
Get most profitable Buyers & Suppliers
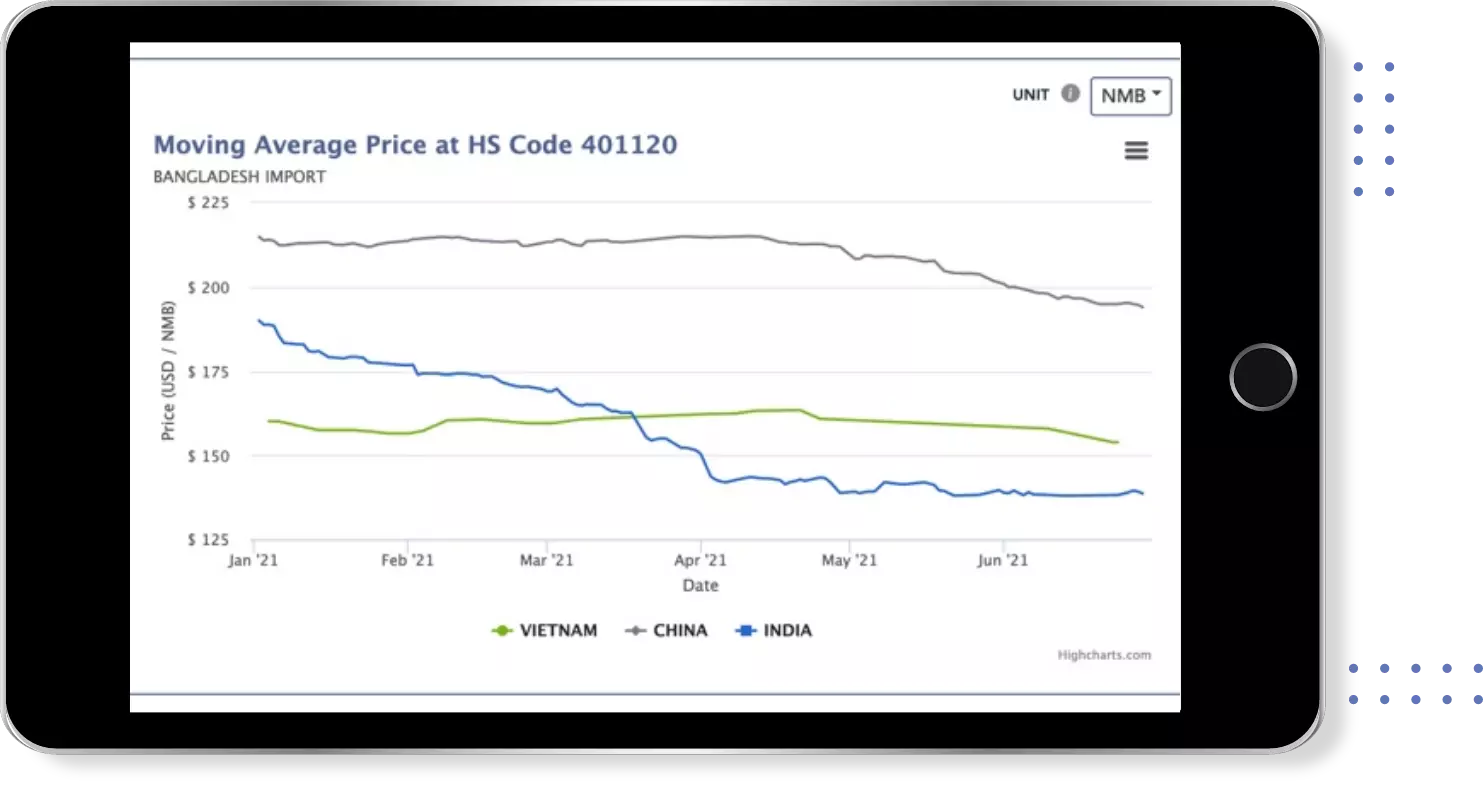
Instant insights on Price , Quantity & Value trends
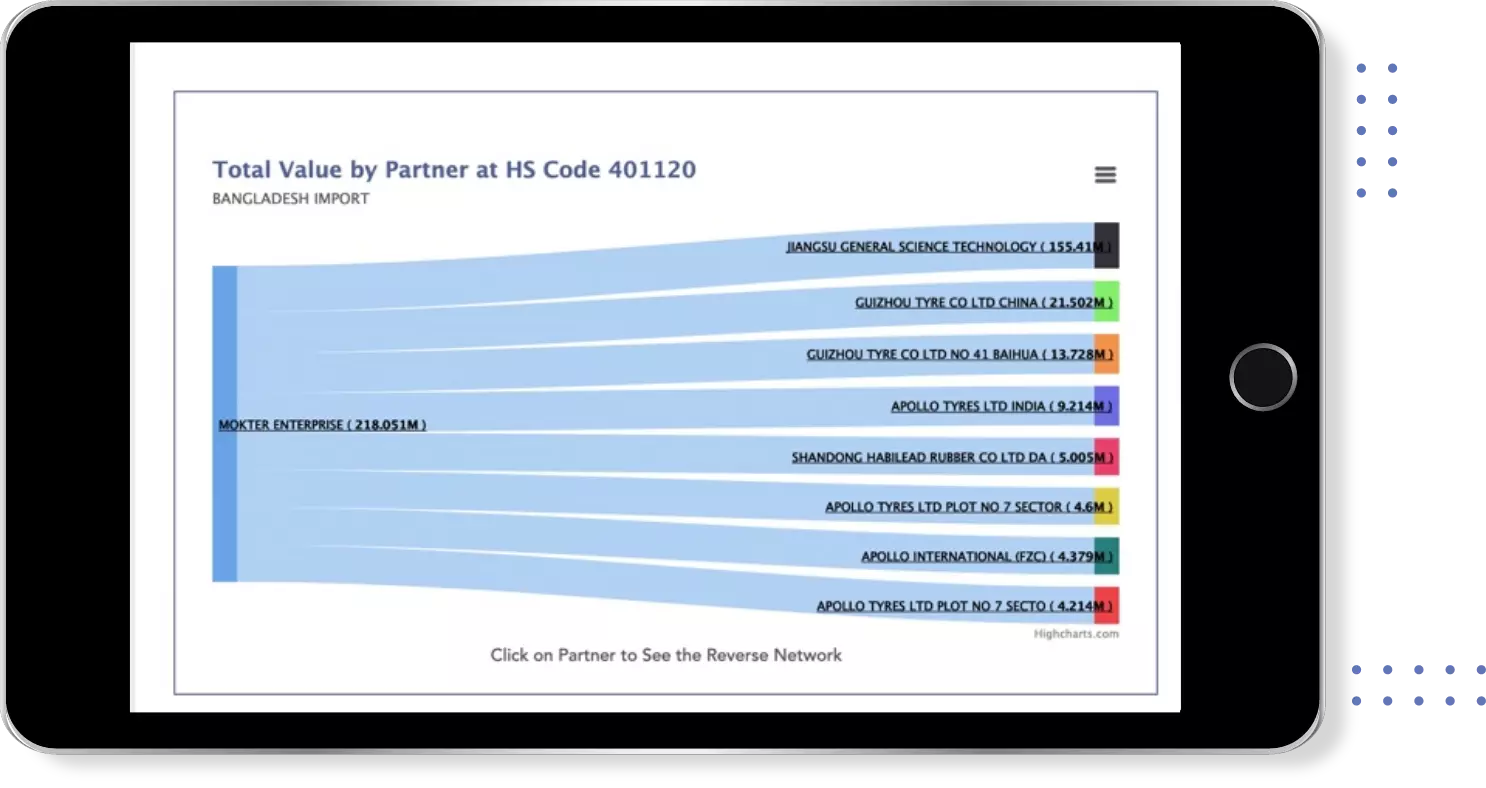
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
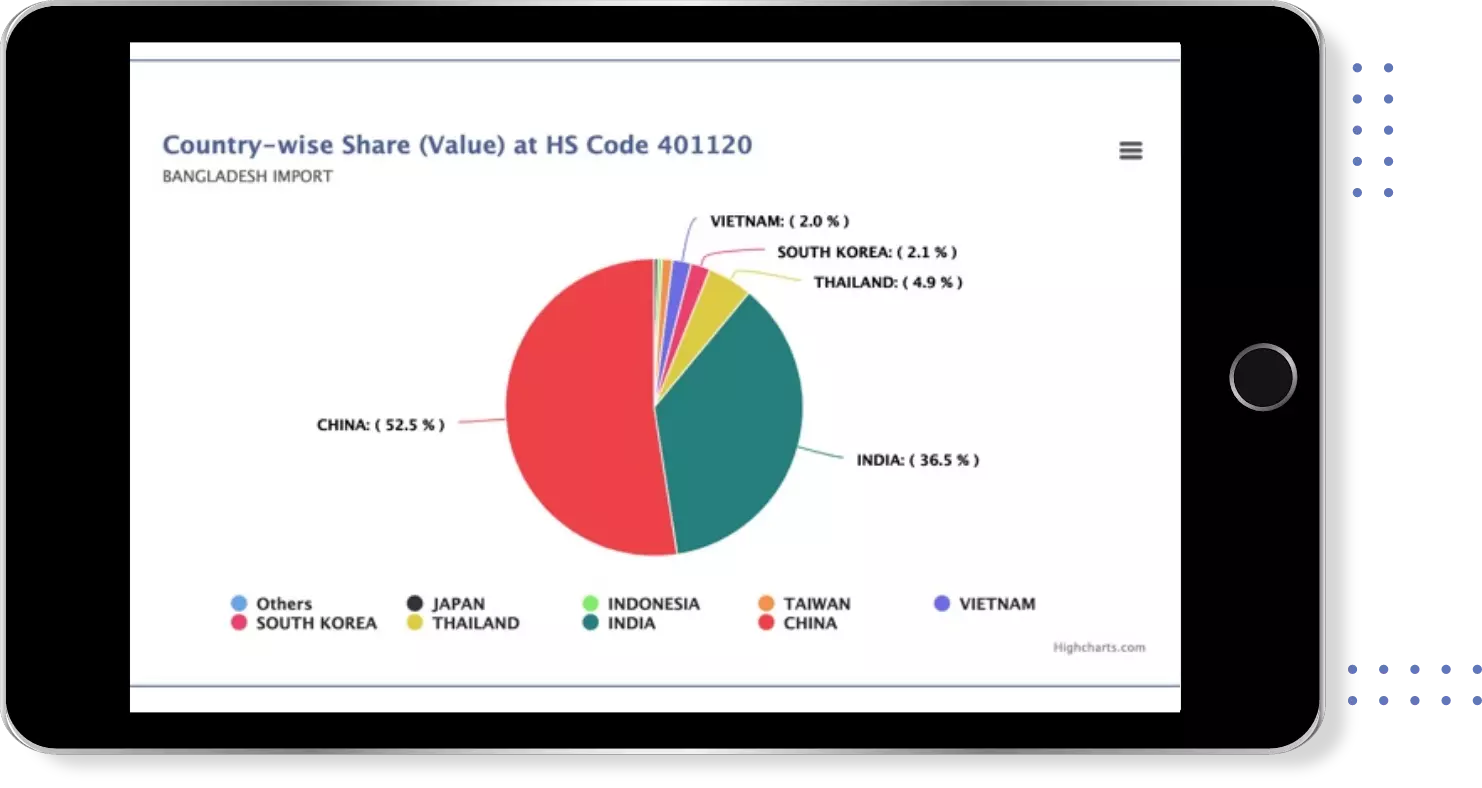
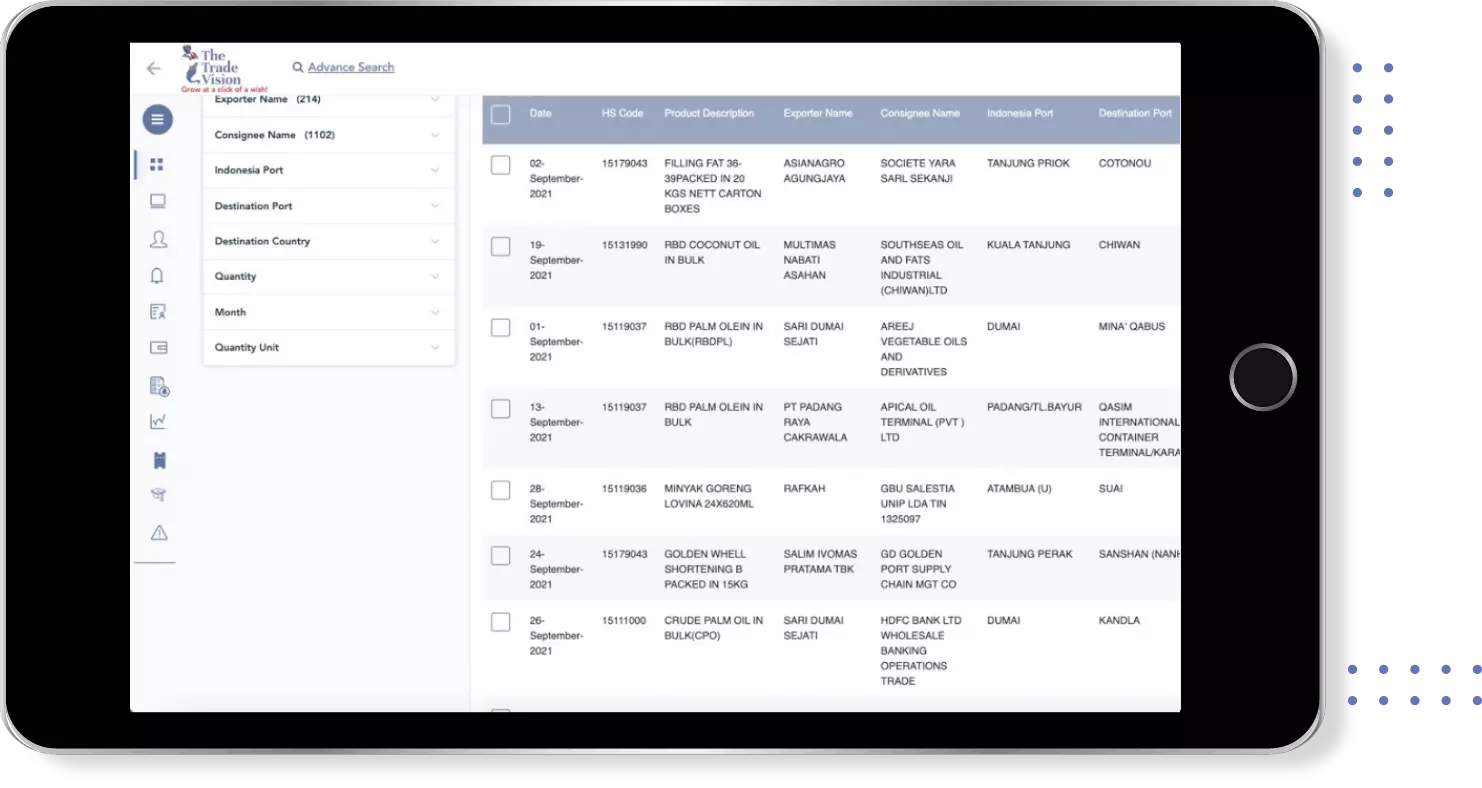
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.