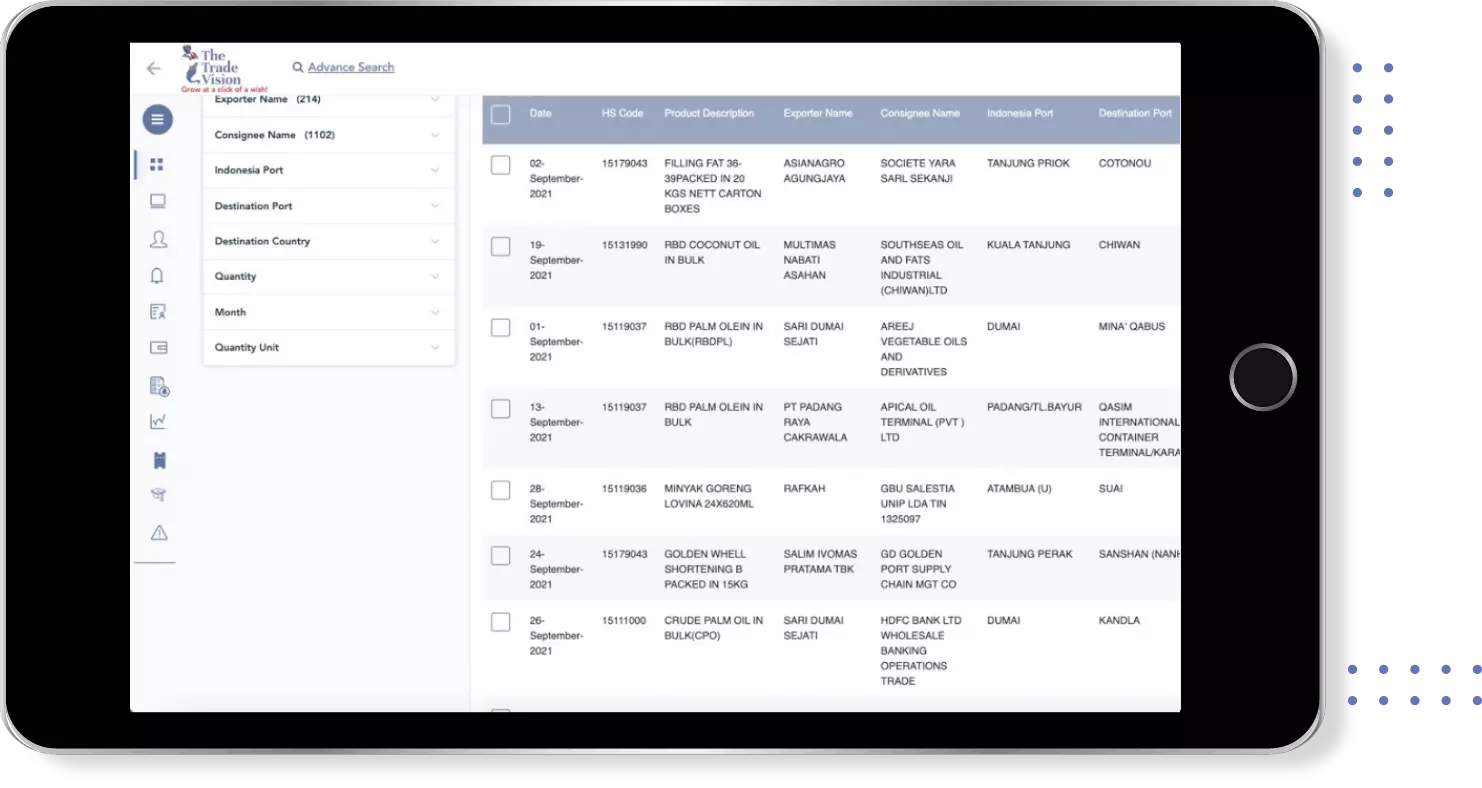
Find verified buyers and sellers of rabies vaccine human use in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- rabies vaccine human use import export data
Overview
- The top 3 supplier countries for rabies vaccine human use are “UKRAINE” , “CHINA” , “FRANCE” ,
- The top 3 Buyer countries for rabies vaccine human use are “PAKISTAN” , “PHILIPPINES” , “UZBEKISTAN” , .
- Top 3 Product Categories for rabies vaccine human use are 300220, 300241, 300212,
The above summary is based on TTV’s Global Export Import data of rabies vaccine human use, compiled from 180+ countries export import shipments updated till .
Global Buyers of Rabies vaccine human use
GRAND CROSS MEDICAL LLC

UZBEKISTAN
HS-Code
Products
3002410000
1) RABIES VACCINE FREEZE-DRIED (VERO CELL) FOR HUMAN USE 2. 5IU DOSE VIALS NO5 0. 5ML DILUENT AMPOULES NO5 (FLAC) SERIES:2021 1 1 380AX INCLUDED SOLUTION 0.5ML. WATER
3002410000
1)RABIES VACCINE FREEZE-DRIED (VERO CELL) FOR HUMAN USE 2.5IU DOSE VIALS NO5 0.5ML DILUENT AMPOULES NO5 (FLAC) INCLUDED SOLUTION 0.5ML. WATER FOR INJECTION NO. 5 (AM
3002200009
1)RABIES VACCINE FREEZE-DRIED (VERO CELL) FOR HUMAN USE 2.5IU DOSE VIALS NO5 0.5ML DILUENT AMPOULES NO5 (FLAC) SERIES: 2021 09342AX INCLUDED SOLUTION 0.5ML. WATER
LTD PARTNERSHIP KNYAZ PHARMA LTD

KAZAKHSTAN
HS-Code
Products
3002410000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE (CONCENTRATION) 2.5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF THE ACTIVE SUBSTANCES RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVATED WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 03072AY (1 1 880DOZ/PACK. ). DATE PR. - MAR. 01 2021 . EXPIRY DATE DO - FEB. 29 2024 SERIES - 2021 06221 AY(91 055DOZ/PK) SERIES - 2021 06224BX(1 5505DOZ/PK) DATES PR. - JUN. 1 8 2021 . DATES BEFORE - JUN. 1 7 2024. NOT FOR VETERINARY SCIENCE. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
3002410000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE (END) 2.5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF ACTIVE SUBSTANCES RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVE WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 03072AY (79570DOZ/PACK. ). DATE OF PRODUCTION - MAR. 01 2021 . EXPIRY DATE UP TO - FEB. 29 2024. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
3002410000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE 2. 5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF THE ACTIVE SUBSTANCES: RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVE WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 1 0345BY (20725 DOS). PRODUCTION DATE - SEPTEMBER 24 2021 EXPIRY DATE - SEPTEMBER 23 2024 FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
OOO MEDBIOLINE

UZBEKISTAN
HS-Code
Products
3002200009
1 . RABIES VACCINE(VERO CELL) FOR HUMAN USE FREEZE-DRIED PHARMACEUTICAL FORM: LYOPHILIZED POWDER FOR PREPARING SOLUTION FOR INJECTION NO. 5 (VIALS) SERIES: 202202006/DRUG TERM
3002410000
1. RABIES VACCINE (VERO CELL) FOR HUMAN USE FREEZE-DRIED / ANTI-RABIES VACCINE LYOPHILIZED (VERO CELL) BIOSHOOT. DOSAGE FORM: LYOPHILIZED POWDER FOR THE PREPARATION OF INJECTION SOLUTION NO. 5 (BOTTLES) COMPLETE WITH SOLVENT - IN
3002410000
1. RABIES VACCINE (VERO CELL) FOR HUMAN USE FREEZE-DRIED / ANTI-RABIES VACCINE LYOPHILIZED (VERO CELL) BIOSHOOT. DOSAGE FORM: LYOPHILIZED POWDER FOR THE PREPARATION OF INJECTION SOLUTION NO. 5 (BOTTLES) COMPLETE WITH SOLVENT - IN
LTD LIABILITY PARTNERSHIP KNYAZ PHARMA LTD

KAZAKHSTAN
HS-Code
Products
3002410000
1. SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED. LYOPHILIZED POWDER FOR PREPARATION OF SOLUTION FOR INTRAMUSCULAR ADMINISTRATION COMPLETE WITH SOLVENT. DOSAGE (CON
3002410000
1. SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED. LYOPHILIZED POWDER FOR PREPARATION OF SOLUTION FOR INTRAMUSCULAR ADMINISTRATION COMPLETE WITH SOLVENT. DOSAGE (CON
Global Suppliers of Rabies vaccine human use
LIAONING CHENG DA BIOTECHNOLOGY CO LTD

CHINA
HS-Code
Products
300241000000
1 . SPEEDA - RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED DOSAGE (END) 2.5 IU. PACKAGING: ONE DOSE IN A GLASS VIAL. 0.5 ML SOLVENT IN GLASS AMPOULE. 5 VIALS WITH PREPARATION AND 5 AMPOULS WITH SOLVENT IN A CARDBOARD BOX. ATC CODE: J07BG01 RABIES VIRUS - INACTIVE WHOLE. COMPOSITION OF ACTIVE SUBSTANCES RABIES VIRUS ANTIGEN (RABIES VIRUS L. PASTEUR PV-2061 GROWN ON VERO CELLS AND INACTIVE WITH BETA-PROPIOLACTONE) 2.5 IU. SERIES - 2021 03072AY (79570DOZ/PACK. ). DATE OF PRODUCTION - MAR. 01 2021 . EXPIRY DATE UP TO - FEB. 29 2024. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
300220900000
RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE DRIED 90945 DOSES
300241900000
RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE DRIED 88530 DOSE
LIAONING CHENG DA BIOTECHNOLOGY

CHINA
HS-Code
Products
300220160000
FOR HUMAN USE RABIES VACCINE FREEZE-DRIED (VERO CELL) I.P.(INACTIVATED) 43900 DOSE/INACTIVATED RABIES VACCINE VERO CELL
300241160000
FOR HUMAN USE RABIES VACCINE FREEZE-DRIED (VERO CELL) I.P.70730 DOSE
300241160000
FOR HUMAN USE RABIES VACCINE FREEZE-DRIED (VERO CELL) I.P. 88655 DOSE
LIAONING CHENG DA BIOTECH

CHINA
HS-Code
Products
300220900000
90725 DOSE RABIES VACCINE FOR HUMAN USE (VERO CELL) FREEZE-DRIED
300220900000
102 CARTONS (91005 DOSES) RABIES VACCINE FOR HUMAN USE(VERIO CELL) FREEZE-DRIED
300220900000
91 015 DOSE RABIES VACCINE FOR HUMAN USE (SPEEDA VERO CELL 2.5 I.U 0.5ML) FREEZE-DRIED POWDER FOR INJECTION
LIAONING CHENG DA BIOTECHNOLOG

CHINA
HS-Code
Products
300220900000
RABIES VACCINE VERO CELL HUMAN USE QTYNW 12 LTR BATCH NO E202012013. MEG.DEC 04.2020.MEF EXP MAR 03.2021 REG NO .091880 ADC NO.05521 DT.14.01.2021
300220900000
RABIES VACCINE (VERO CELL) HUMAN USE (QTYN/W 54 LTR ) BATCH NO E22A003. MEG.20 04.2022.EXP 19.07.2022 REG NO .091880 ADC NO.F-1355762223264 DT.28.05.2022
300241900000
RABIES VACCINE (VERO CELL) HUMAN USE (QTY 54) BATCH NO E22A006 MEG.15.072022 EXP 14.10.2022 REG NO .091880 ADC F-2412962224827 DT 24.08.2022(HIGH COURT PETITION NO 5233 DT 08.09.2022)
Global Export Import Trade Data for rabies vaccine human use
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 PHILIPPINES PHILIPPINES | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 CHINA CHINA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 CHINA CHINA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 JAPAN JAPAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
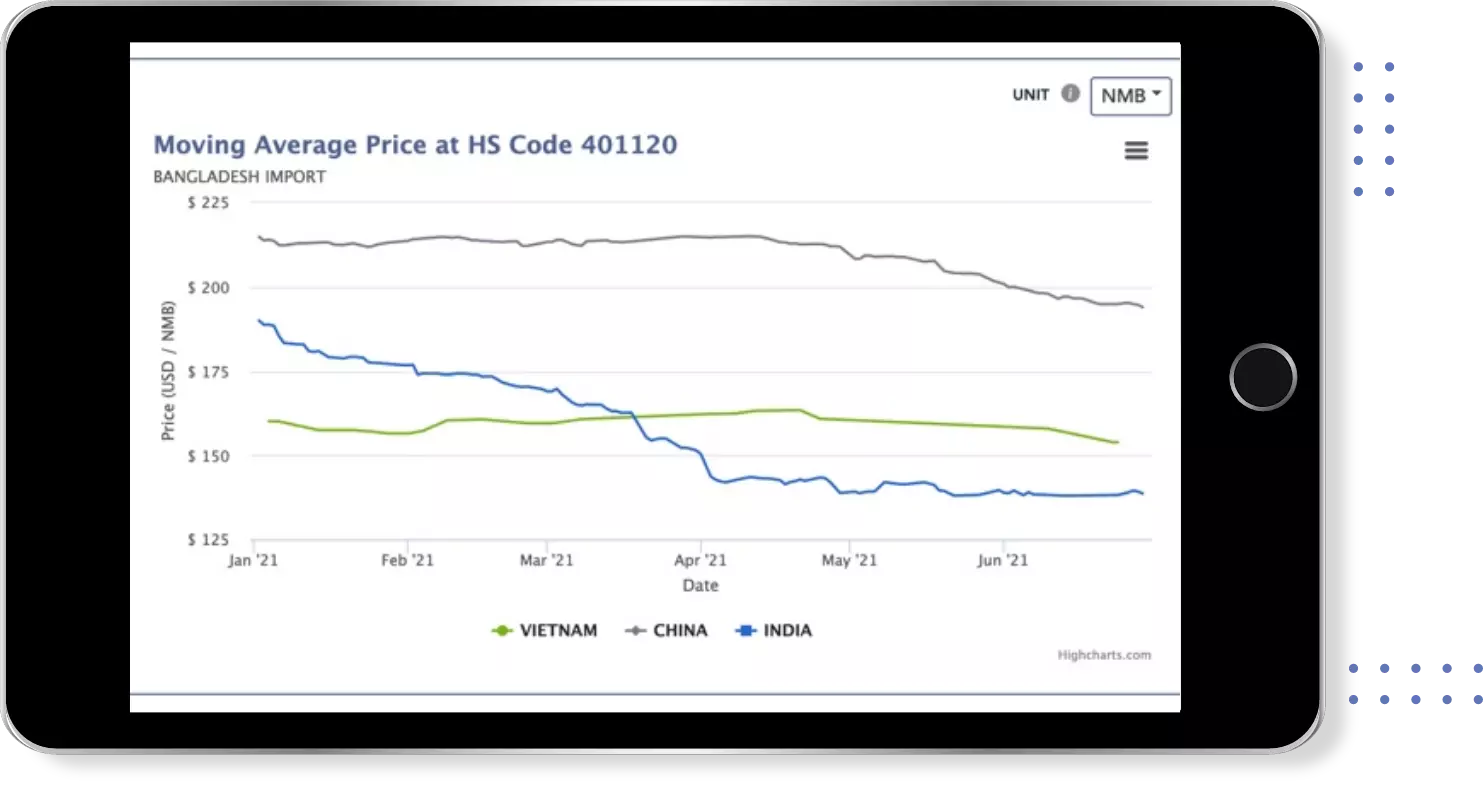
Instant insights on Price , Quantity & Value trends
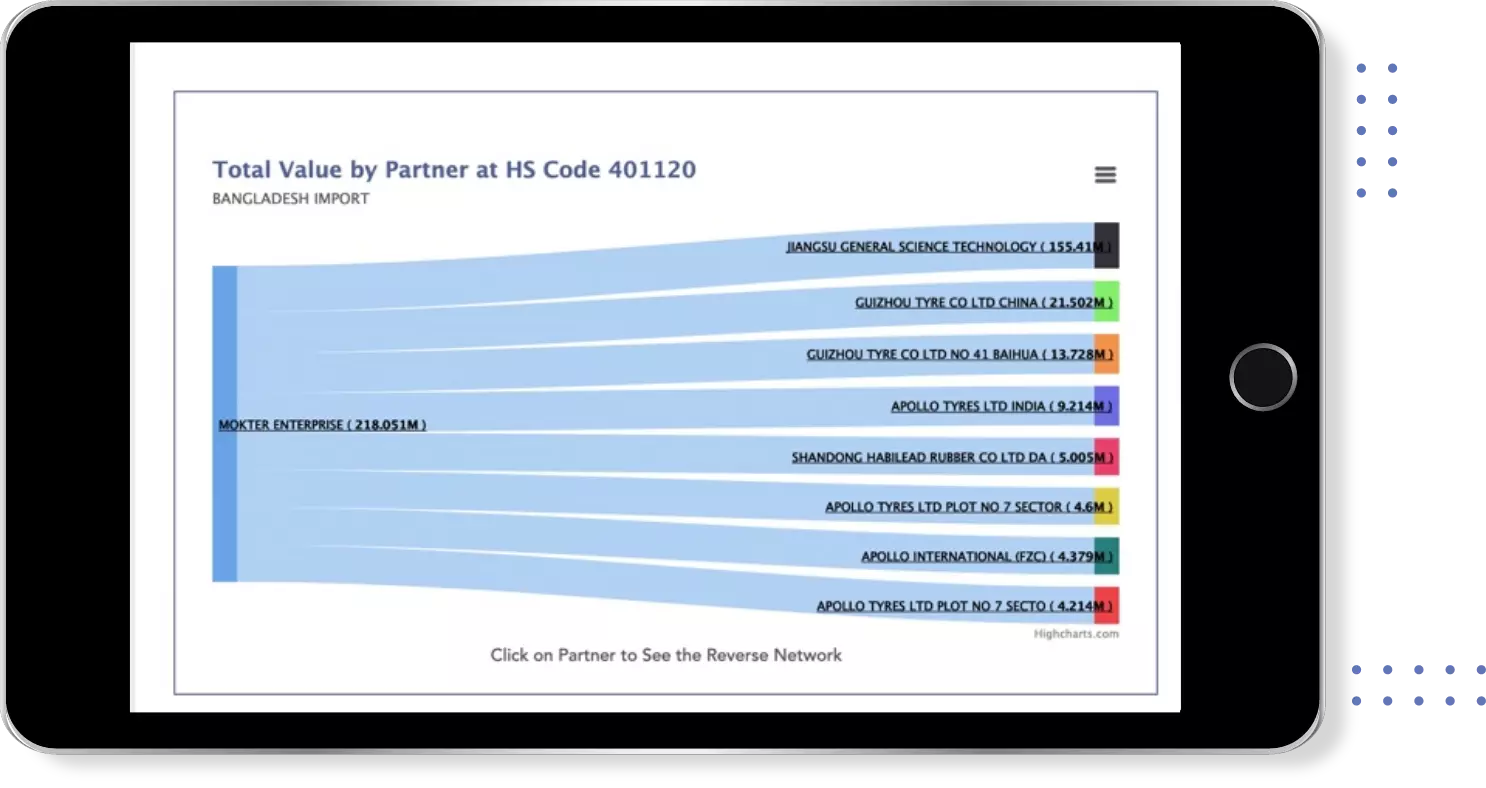
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries