Find verified buyers and sellers of k gold in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- k gold hs code 300490 import export data
Overview
- The top 3 supplier countries for HS Code 300490 are “VIETNAM” , “UNITED STATES OF AMERICA” , “UZBEKISTAN” ,
- The top 3 Buyer countries for HS Code 300490 are “UZBEKISTAN” , “PAKISTAN” , “UKRAINE” , .
- Top 1 Product Categories for HS Code 300490 are 300490,
The above summary is based on TTV’s Global Export Import data of HS Code 300490, compiled from 180+ countries export import shipments updated till .
- HS Code: 300490
Global Buyers of k gold under HS-Code 300490
OOO FARM LYUKS INVEST

UZBEKISTAN
HS-Code
Products
3004900002
1 ) GOLD STAR POWDER (ORANGE) 1 5 G NO1 0 INN: COMB. DRUG (PARACETAMOL PHENYLEPHRINE PHENIRAMINE ASCORBIC ACID) SERIES 0 80921 0 90921 - 3600 PACK. BEFORE: MANUFACTURING: DANAPHA PHARMACEUTICAL JSC (VIETNAM) : WEIGHT: 660 KG; 2) TYPE AND NUMBER OF PACKS
3004900002
1 ) GOLD STAR POWDER (RASPBERRY) 1 5 G NO1 0 INN: COMB. DRUG (PARACETAMOL PHENYLEPHRINE PHENIRAMINE ASCORBIC ACID) SERIES 1 1 0921 1 20921 - 3622 PACK. EXPIRY BEFORE: MANUFACTURING: DANAPHA PHARMACEUTICAL JSC (VIETNAM) : WEIGHT: 664.03 KG; 2) TYPE AND QUANTITY OF PACKAGING
3004900002
1 ) GOLD STAR POWDER (ORANGE) 1 5G. NO1 0 (BAGS) INN: COMB. DRUG (PARACETAMOL PHENYLEPHRINE PHENIRAMINE ASCORBIC ACID) SERIES 0 40321 - 3685 PACK. EXPIRY UNTIL: 03/24. MANUFACTURED: DANAPHA PHARMACEUTICAL JOINT STOCK COMPANY VIETNAM : WEIGHT: 675.58 KG; 2)
LLC PHARMA CHOICE

UZBEKISTAN
HS-Code
Products
3004900002
MEDICINES 1) GOLD STAR BALM PENCIL FOR INHALATION 1.3G (TUBE). SERIES: 0301 21 UZ TO 1 0. 01 . 26G. =1 0080UP. (INN:COMB. DRUG (MENTHOL CAMPHOR CAJEPUT OIL OCIMUM OIL CINNAMON OIL). MANUFACTURER DANAPHA PHARMACEUTICAL JSC VIETNAM - WEIGHT: 1 20 KG ;
3004900002
MEDICINES 1) GOLDEN STAR POWDER FOR PREPARATIONS. SOLUTION WITH RASPBERRY FLAVOR 15G. NO. 10 (SACHET). SERIES: 020223 TO 02/01/26 = 540 PACK. INN: COMB.DRUG (PARACETAMOL PHENYLEPHRINE PHENIRAMINE ASCORBIC ACID) DANAPHA PHARMACEUTICAL JSC VIETNAM - WEIGHT : 99 KG
3004900002
MEDICINES 1) GOLDEN STAR POWDER FOR PREPARATIONS. SOLUTION WITH RASPBERRY FLAVOR 15G. NO. 10 (SACHET). SERIES: 020223=540 PACK. - WEIGHT: 99 KG ; 2)0 PALLETS;
OOO MEROS PHARM

UZBEKISTAN
HS-Code
Products
3004900002
1 ) ROMPHARM GOLD TAPE (INN: IBANDRONIC ACID) LYOPHILISATE FOR PREPARING SOLUTION FOR INFUSION 4 MG N1 (BOTTLES) -600UP. SER. 2203272 221 4971 . PR-L. SC ROMPHARM COMPANY SRL RUMINIA. : WEIGHT: 18.329 KG; 2) TYPE AND QUANTITY OF PACKAGING: PART OF PALLETS;
3004900002
1) GOLD STAR BALM (INN: MENTHOL CAMPHOR CAJEPUT OIL OCIME OIL CINNAMON OIL) OINTMENT 4 G - 33480UP 47CART. COR SER. 1 80621 USD. MFR: DANAPHA PHARMACEUTICAL JOINT STOCK COMPANY VIETNAM: WEIGHT: 372 KG; 2) TYPE AND QUANTITY OF PACKAGING: 2 PALLETS
3004900002
1 ) GOLD STAR BALM (INN: MENTHOL CAMPHOR CAJEPUT OIL OCIME OIL CINNAMON OIL) LIQUID BALM 5 ML - 6240UP 1 3CART. COR SER. 0201 21 USD. MFR: DANAPHA PHARMACEUTICAL JOINT STOCK COMPANY VIETNAM: WEIGHT: 182 KG; 2) TYPE AND NUMBER OF PACKS
TOO DANSON KZ

KAZAKHSTAN
HS-Code
Products
3004900002
1 . BALM GOLD STAR OINTMENT FOR EXTERNAL NOTE 4 Y EXPIRED UNTIL 07. 1 1 . 2026 SERIES 251 1 21 KZ
3004900002
1 . BALM GOLD STAR LIQUID 5 ML IN A BOTTLE EXPIRED UNTIL 10.08.2026 SERIES 050821 KZ
3004900002
1 . BALM GOLD STAR LIQUID 5 ML IN A BOTTLE IS GOOD UP TO 1 1 . 08. 2026 SERIES 060821 KZ
Global Suppliers of k gold under HS-Code 300490
NATUR PRODUKT ZDROVIT SP Z O O

ITALY
HS-Code
Products
300490990000
DIABETES TREATMENT: GOLDDICRON (GLICLAZIDE 30MG) .S?K: VN-18660-15. LOT: 202970 202971 202908 202965. NSX: 01/2022 01/2022 12/2021 12/2021. HD: 01/2025 01/2025 12/2024 12/2024.WITH 100%.FOC.
300490990000
NEW MEDICINE GOLDDICRON (GLICLAZIDE 30MG) BOX OF 5 BLISTERS X 20 CONTROLLED RELEASE TABLETS.S?K: VN-18660-15 LOT: 193018 193028.NSX: 2/ 2021.HD: 1/2024. ROAD. 100% NEW .FOC.
300490990000
GOLDDICRON DIABETES TREATMENT (GLICLAZIDE 30MG). BOX OF 5 BLISTERS X 20 CONTROLLED RELEASE TABLETS. S?K: VN-18660-15. LOT: 203525. NSX: JANUARY 24 2022. HD: JANUARY 23 2025. 100%NEW. FOC.
SHENZHEN TRIMANSON EXPRESS LTD

CHINA
HS-Code
Products
300490500000
PK120000257827 ?BUY 1 GET 1 FREE?GOLDEN VISION EYE DROPS 0.16 KG TOTAL HOUSE 160 CONTAINING 27 KG DE-MINIMIS 157 CONTAINING 26 KG RELEASED 3 CONTAINING 1 KG(SPEEDAF)
300490500000
PK120000470259 ?BUY 1 GET 1 FREE?GOLDEN VISION EYE DROPS 0.109 KG
300490500000
PK120000368755 ?BUY 1 GET 1 FREE?GOLDEN VISION EYE DROPS 0.135 KG
BOEHRINGER INGELHEIM ANIMAL HEALTH DO BRASIL LTDA

BRAZIL
HS-Code
Products
300490
10 PACKAGES WITH PHARMACEUTICAL PRODUCTS NW 210.06 KG HS-CODE 300490<BR/>26 PACKAGES WITH PHARMACEUTICAL PRODUCTS NW 1065.30 KG 02 40RF CONTAINERS 1.574 BOXES ON 36 PALLETS CONTAINING ZIMECTERIN GOLD 1 SYRINGE X40 US BATCH AD052 20 EXPIRE DATE NOV 23 PO N
300490
2025 BOXES WITH PHARMACEUTICAL PRODUCTS NW 3098.35 KG 02 40RF CONTAINERS SHIPPERS LOAD AND COUNT 3.386 BOXES ON 71 PALLETS CONTAINING ZIMECTERIN GOLD 1 SYRINGE X40 US BATCH DD052 20 EXPIRE DATE NOV 23 PO NUMBER 0300608380 ZIMECTERIN GOLD 1 SYRINGE X40 US
300490
2025 BOXES WITH PHARMACEUTICAL PRODUCTS NW 3098.35 KG 02 40RF CONTAINERS SHIPPERS LOAD AND COUNT 3.386 BOXES ON 71 PALLETS CONTAINING ZIMECTERIN GOLD 1 SYRINGE X40 US BATCH DD052 20 EXPIRE DATE NOV 23 PO NUMBER 0300608380 ZIMECTERIN GOLD 1 SYRINGE X40 US
AO PHARMASYNTEZ

RUSSIA
HS-Code
Products
300490000200
1. OTHER MEDICINES: GOLDA MV (GOLDA MV) TABLETS WITH MODIFIED. RELEASE 60 MG (3X10) CONTOUR CELL PACKAGING NO. 30 INN: GLICLAZIDE SERIES 1500822 - 1500 PCS/PACK (9 CARD BOXES) NOT FOR VETERINARY MEDICINE PRODUCED BY: PHARMASINTEZ-TYUMEN LLC RUSSIA - WEIGHT : 30 KG 2. ME
300490000200
1. OTHER MEDICINES: GOLDA MV (GOLDA MV) TABLETS WITH MODIFIED. RELEASE. 30 MG (6X10) CONTOUR CELL PACKAGING NO. 60 INN: GLICLAZIDE SERIES 1470822 - 1500 PCS/PACK (9 CARD BOXES) NOT FOR VETERINARY MEDICINE MANUFACTURED: PHARMASINTEZ-TYUMEN LLC RUSSIA - WEIGHT : 45 KG 2. ME
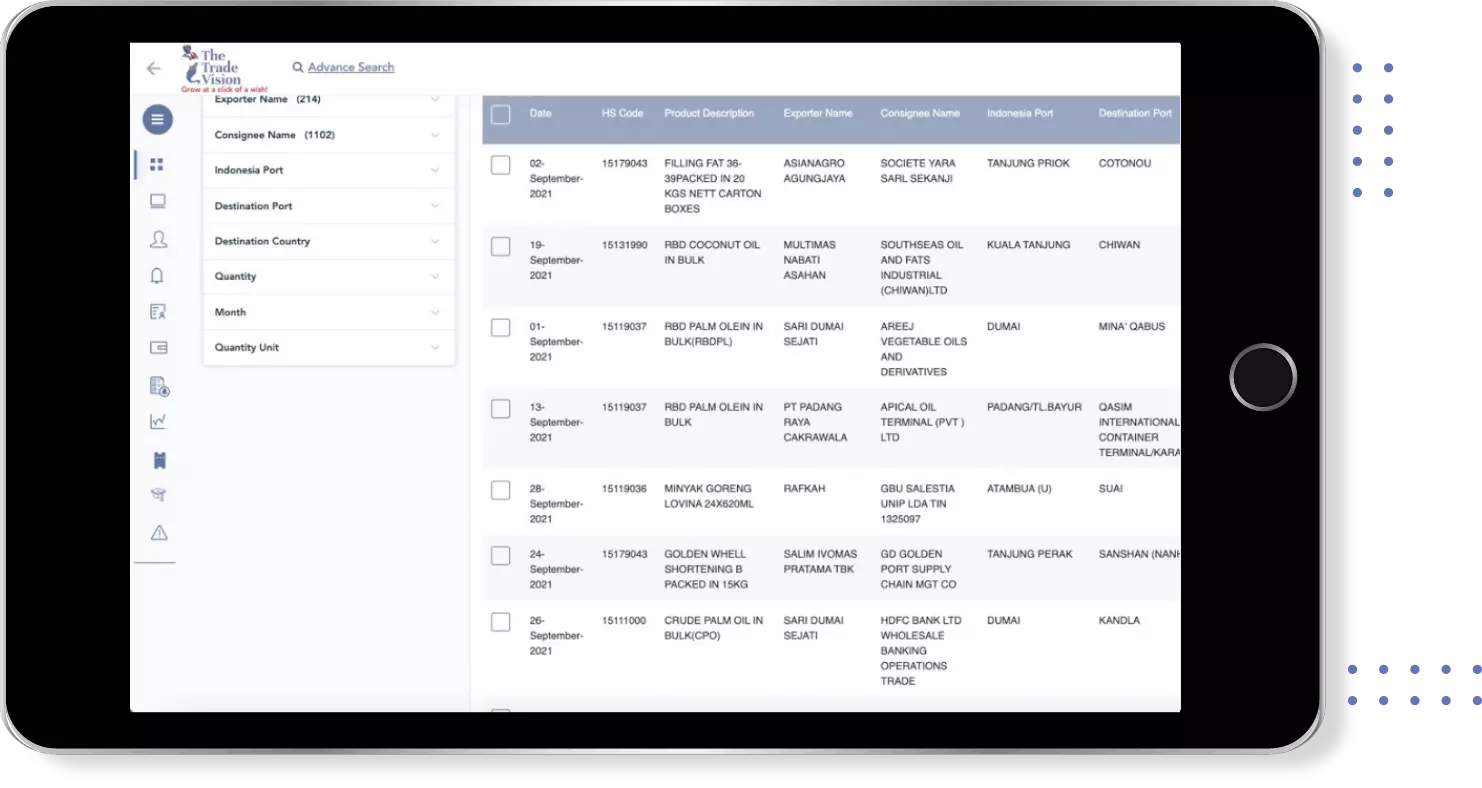
Global Export Import Trade Data for k gold under HS-Code 300490
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UZBEKISTAN UZBEKISTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SINGAPORE SINGAPORE | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED ARAB EMIRATES UNITED ARAB EMIRATES | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 MALAYSIA MALAYSIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
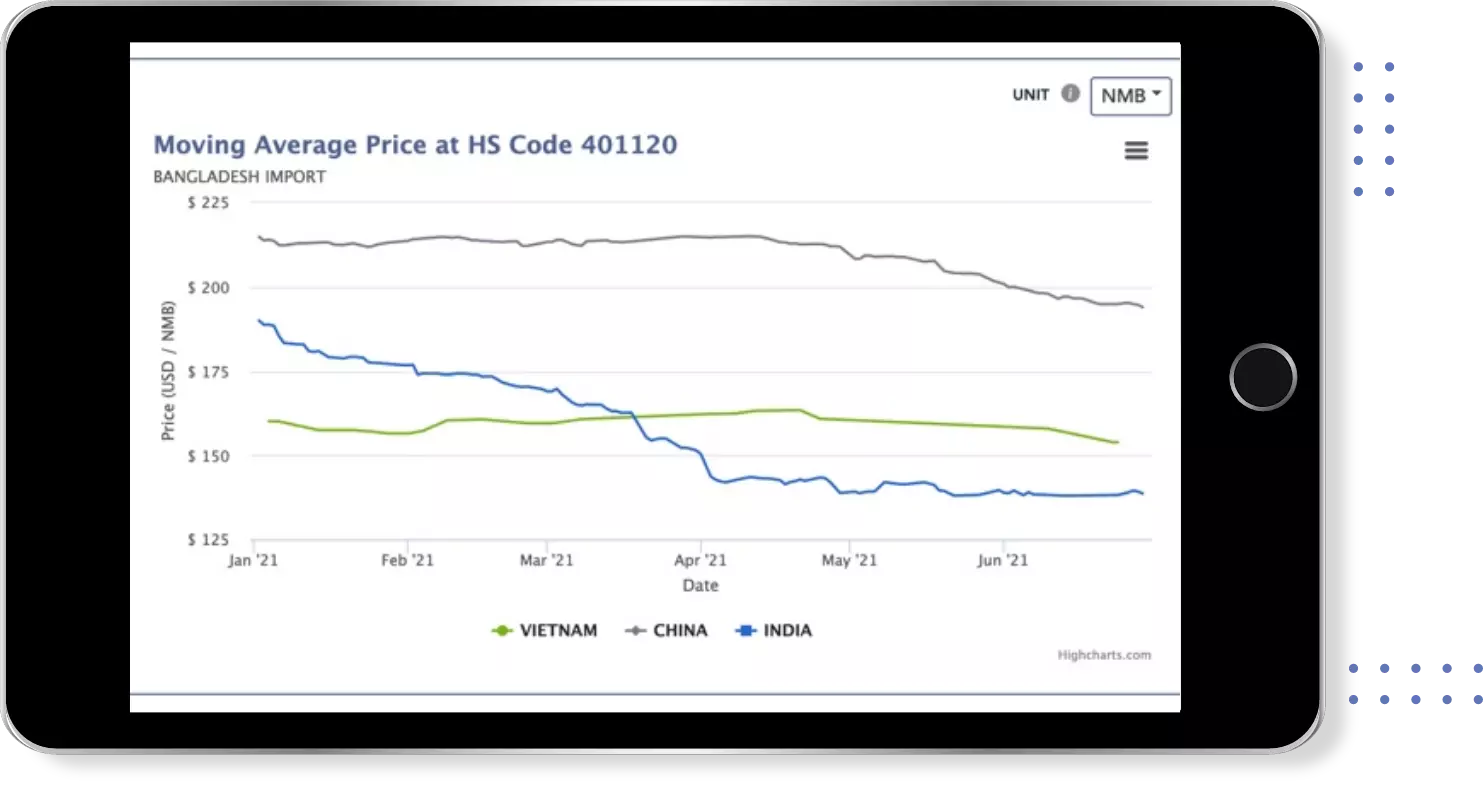
Instant insights on Price , Quantity & Value trends
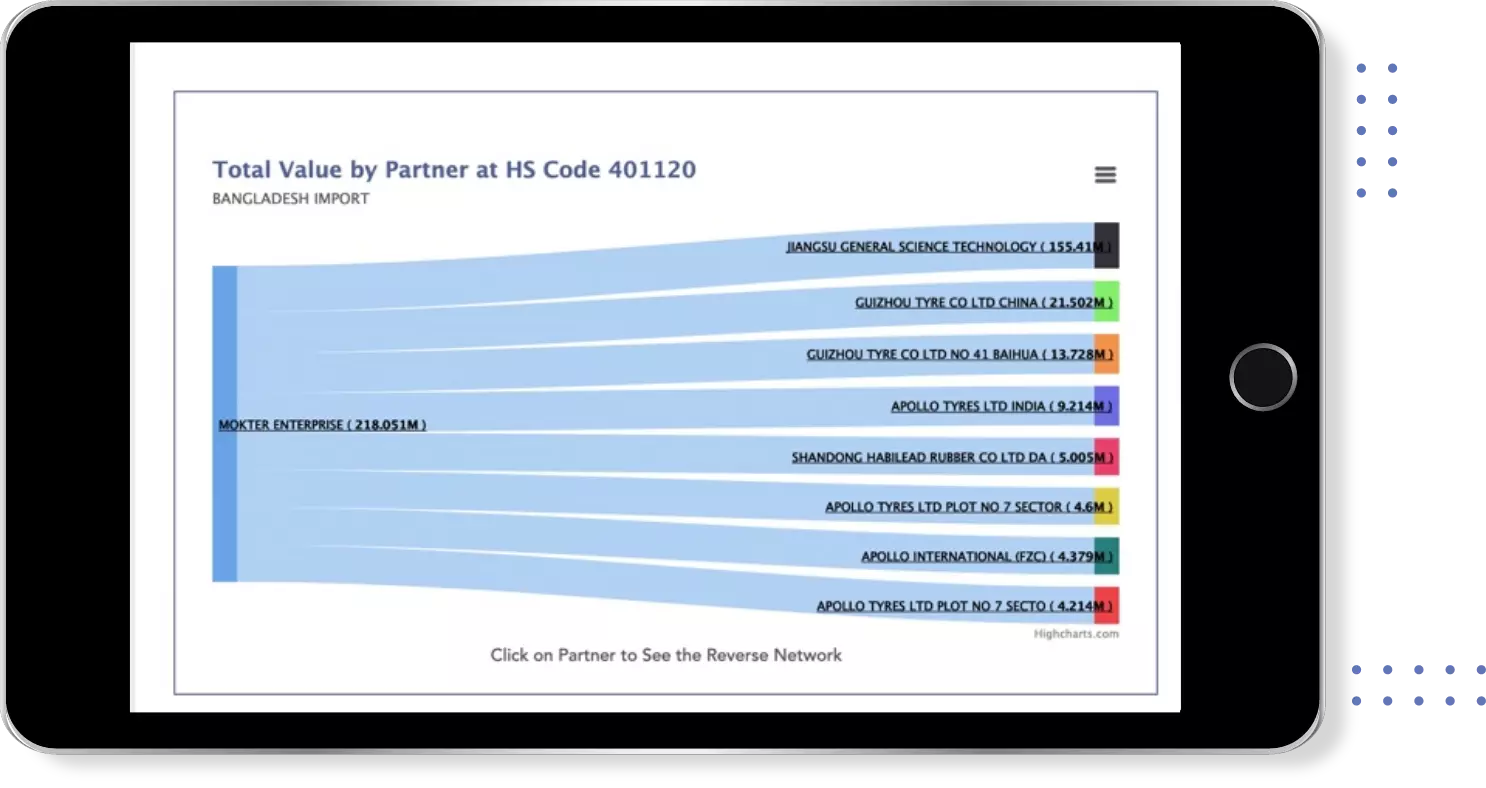
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries