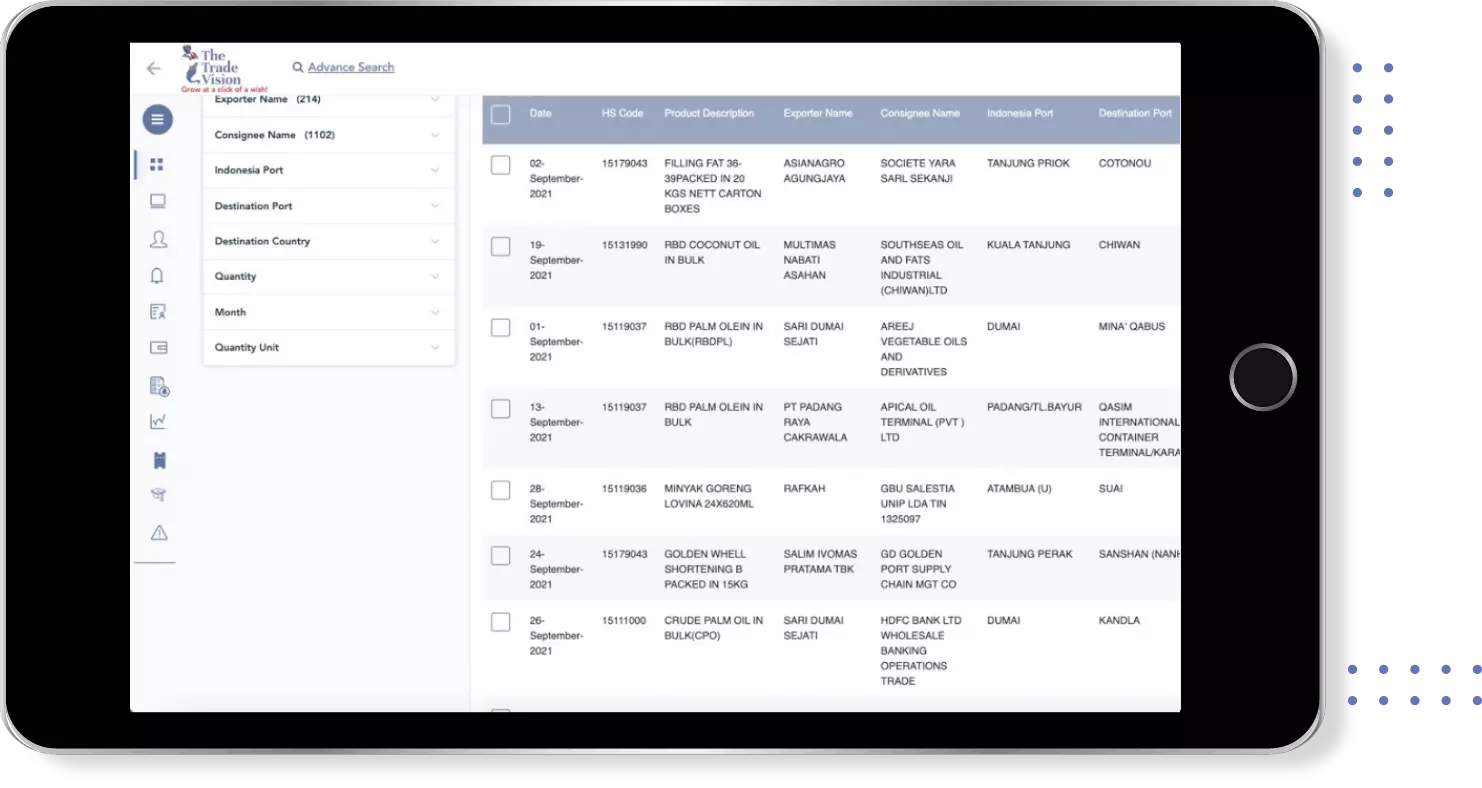
Find verified buyers and sellers of ibuprofen 800mg in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- ibuprofen 800mg import export data
Overview
- The top 3 supplier countries for ibuprofen 800mg are “ECUADOR” , “PERU” , “TURKEY” ,
- The top 3 Buyer countries for ibuprofen 800mg are “ECUADOR” , “UZBEKISTAN” , “PANAMA” , .
- Top 3 Product Categories for ibuprofen 800mg are 300490, 310510, 482110,
The above summary is based on TTV’s Global Export Import data of ibuprofen 800mg, compiled from 180+ countries export import shipments updated till .
Global Buyers of Ibuprofen 800mg
OOO PHARMAXI

UZBEKISTAN
HS-Code
Products
3004900002
1) BRUFEN RETARD 800MG 2X1 0 TAB NO. 20 (2X1 0) (BLISTERS) INN: (IBUPROFEN / IBUPROFEN) QUANTITY - 360UP SERIES: 45551 PC EXPIRY UNTIL: 01 . 08. 2025 NOT INTENDED FOR VETERINARY USE PRODUCED
3004900002
1) BRUFEN RETARD 800MG 2X1 0 TAB NO20 (2X1 0) (BLISTERS) INN: (IBUPROFEN / IBUPROFEN) QUANTITY: 420 PU SERIES: 37391 PC EXPIRY UNTIL: 01 . 1 2. 2024 NOT INTENDED FOR VETERINARY USE PRODUCED
3004900002
1) BRUFEN RETARD 800MG 2X1 0 TAB NO20 (2X1 0) (BLISTERS) INN: (IBUPROFEN/IBUPROFEN) QUANTITY - 420 PU PACKED IN CARDBONS WITH SOLD SERIES: 37391 PC EXPIRY UNTIL: 01 . 1 2. 2024 NOT INTENDED
TOO INKAR

KAZAKHSTAN
HS-Code
Products
3004900002
1 . INTRAFEN 800MG/8ML IV INF. 1 0 FLAKON (INTRAFEN (IBUPROFEN) SOLUTION FOR INTRAVENOUS INTRODUCTION 800MG / 8ML 4 ML OF THE DRUG IN A GLASS BOTTLE 10 VIALS IN A CARDBOARD BOX) SERIES 21 1 20241 GOOD 1 1. 24.
3004900002
1 . INTRAFEN 800MG/8ML IV INF. 1 0 FLAKON (INTRAFEN (IBUPROFEN) SOLUTION FOR INTRAVENOUS INTRODUCTION 800MG / 8ML 4 ML OF THE DRUG IN A GLASS BOTTLE 10 VIALS IN A CARDBOARD BOX) SERIES 21 1 1 0235 VALUE 31. 1 0. 24.
3004900002
1 . INTRAFEN 800MG/8ML IV INF. 1 0 FLAKON (INTRAFEN (IBUPROFEN) SOLUTION FOR INTRAVENOUS INTRODUCTION 800MG / 8ML 4 ML OF THE DRUG IN A GLASS BOTTLE 1 0 VIALS IN A CARDBOARD BOX) SERIES 21 1 20240 VALID 1 1/24.
DRUG PROMOTION LLC

UZBEKISTAN
HS-Code
Products
3004900002
MEDICATIONS NOT FOR VETERINARY SECTION IN FORMS FOR RETAIL: 1) BROUFEN RETARD (IBUPROFEN) TABLETS LONG-LASTING 800MG NO. 20-21 0UP SERIES: 22346PC. MANUFACTURED: FAMAR AVE GREECE. : WEIGHT: 8.16KG; 7) EXPIRY DATE: 01 . 09. 2023; 8) INVESTPROG CODE
3004900002
MEDICINES NOT FOR VETERINARY SECTION IN FORMS FOR RETAIL: 1) BROUFEN RETARD (IBUPROFEN) TABLETS LONG-LASTING 800MG NO20-300UP SERIES: 37391 PC. MANUFACTURED: FAMAR AVE GREECE. : WEIGHT: 1 1.65 KG; 7) EXPIRY DATE: 01 . 1 2. 2024; 8) INVESTPRO CODE
3004900002
MEDICATIONS NOT FOR VETERINARY SECTION IN FORMS FOR RETAIL: 1) BROUFEN RETARD (IBUPROFEN) TABLETS LONG-LASTING 800MG NO. 20-21 0UP SERIES: 22346PC. MANUFACTURED: FAMAR AVE GREECE. : WEIGHT: 8.16KG; 7) EXPIRY DATE: 01 . 09. 2023; 8) INVESTPROG CODE
LLC AERO PHARM GROUP

UZBEKISTAN
HS-Code
Products
3004900002
1 . BROUFEN RETARD TABLETS 800MG NO20 - 360UP. INN: IBUPROFEN - WEIGHT: 1 6.48 KG 2. 2 KART COR PALLET 8.000 1 1 . 02
3004900002
1 . BROOFEN RETARD CAPS. 800MG NO. 20 - 268UP. SERIES 37391 PC TO 01 . 1 2. 2024 INN: IBUPROFEN MANUFACTURER: GMBH ABBOTT LABORATORIES GERMANY - WEIGHT: 1 2.58 KG 2. 2 KART COR 7. 01 . 1 2. 2024 8.000 1 1 . 02
Global Suppliers of Ibuprofen 800mg
STRIDES PHARMA GLOBAL PTE LTD

SINGAPORE
HS-Code
Products
300490990000
IBUPROFEN TABLETS USP 800MG (500S) - (5900505A) 940 TABLETS -(FOR R & D TESTING) (FOC)
300490990000
IBUPROFEN TABLETS USP 800MG (500S) - (5900593A) 940 TABLETS -(FOR R & D TESTING) (FOC)
300490990000
IBUPROFEN TABLETS USP 800MG (500S) - (5900871A) 970 TABLETS -(FOR R & D TESTING) (FOC)
CORPORACION BONIMA SA DE CV

EL SALVADOR
HS-Code
Products
300490990000
DRUGS FOR INTERNAL USE (IBUPROFEN MK400 MG IBUPROFEN MK 600 MG LORATADINE MK 1 0MG METFORMIN MK 850 MG TRIMETHOPRIM SULFAMETHOXAZOLE MK FORTE 1 60MG/800MG
300490990000
MEDICINES FOR INTERNAL USE (FLUCONAZOLE MK 1 50MG IBUPROFEN MK600MG LORATADINE MK1 0MG LOSARTAN MK50MG SILDENAFIL MK1 00MG TETRACYCLINE MK 500MG TRIMETHOPRIME SULFAMETOXAZOLE MKFORTE 1 60MG/800MG))
300490990099
MEDICINES FOR HUMAN USE (ENALAPRIL MK 20MG FLUCONAZOLE MK 1 50MG IBUPROFENE MK 400MG) LORATADINE MK 1 0MG TRIPETOPRIMA SULFAMETHOXAZOLE MK FORTE 1 60MG/800MG
BONIMA CORPORATION SA DE CV

EL SALVADOR
HS-Code
Products
300490990099
MEDICINES FOR HUMAN USE (ACETAMINOFEN MK 500MG IBUPROFEN MK 400 MG CAPTOPRIL MK 25 MG TRIMETHOPRIM SULFAMETHOXAZOLE MK FORTE 1 60 MG/800MG)
300490990099
MED. FOR HUMAN USE (ACETAMINOFEN 500MG IBUPROFEN 400MG CAPTOPRIL 25MG TRIMETHOPRIM SULFAMETHOXAZOLE 1 60 MG/800MG AMLODIPINE 5MG FUROSEMIDE 40MG LORATADINE 1 0MG METFORMIN 850MG)
300490990099
MED. FOR HUMAN USE (FLUCONAZOLE MK 1 50MG IBUPROFEN MK 400 MG IBUPROFEN MK 600MG LORATADINE MK 1 0MG TRIMETHOPRIME SULFAMETHOXAZOLE MK FORTE 1 60MG/800MG SILDENAFIL MK 1 00MG)
ESPEE BIOPHARMA AND FINE CHEM LLC

UNITED STATES OF AMERICA
HS-Code
Products
300490990000
DUEXIS- IBUPROFEN AND FAMOTIDINE 800MG/26.6 MG TABLET COATED(PACK: 90 S X 2 BTLS = 180 TABLETS) FOR RESEARCH PURPOSE ON
300490990000
DUEXIS-IBUPROFEN AND FAMOTIDINE TABLET COATED-DOSE 800MG/26.6MG/90 IN 1 BOTTLE-QTY 2-R&D-TU/IV-RD/507/2020 DT:30.4.2021
300490990000
DUEXIS IBUPROFEN & FAMOTIDINE TABLETS 800MG / 26.6MG (PKG SIZE 90 TABS)
Global Export Import Trade Data for ibuprofen 800mg
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 GUATEMALA GUATEMALA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
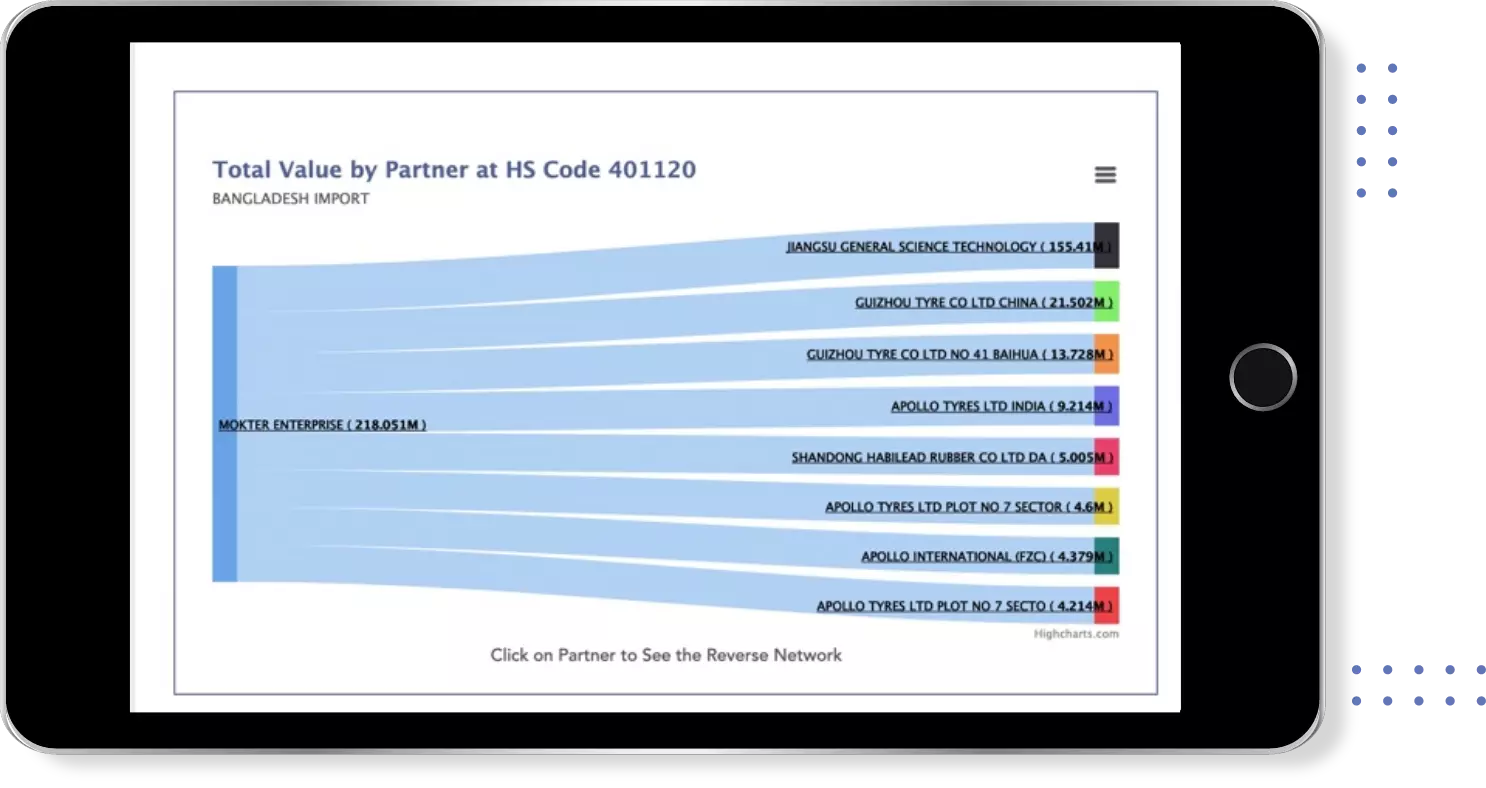
Get most profitable Buyers & Suppliers
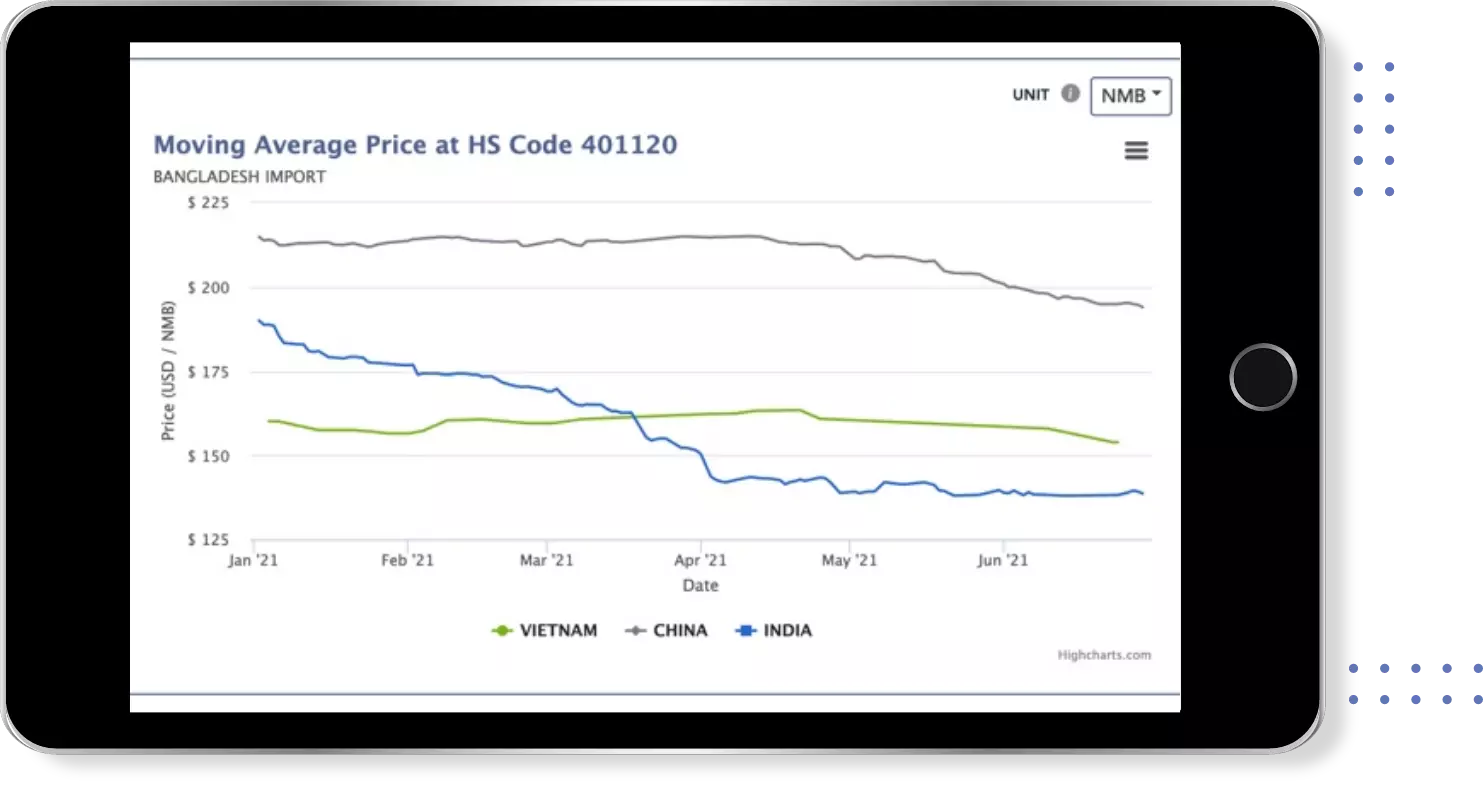
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries