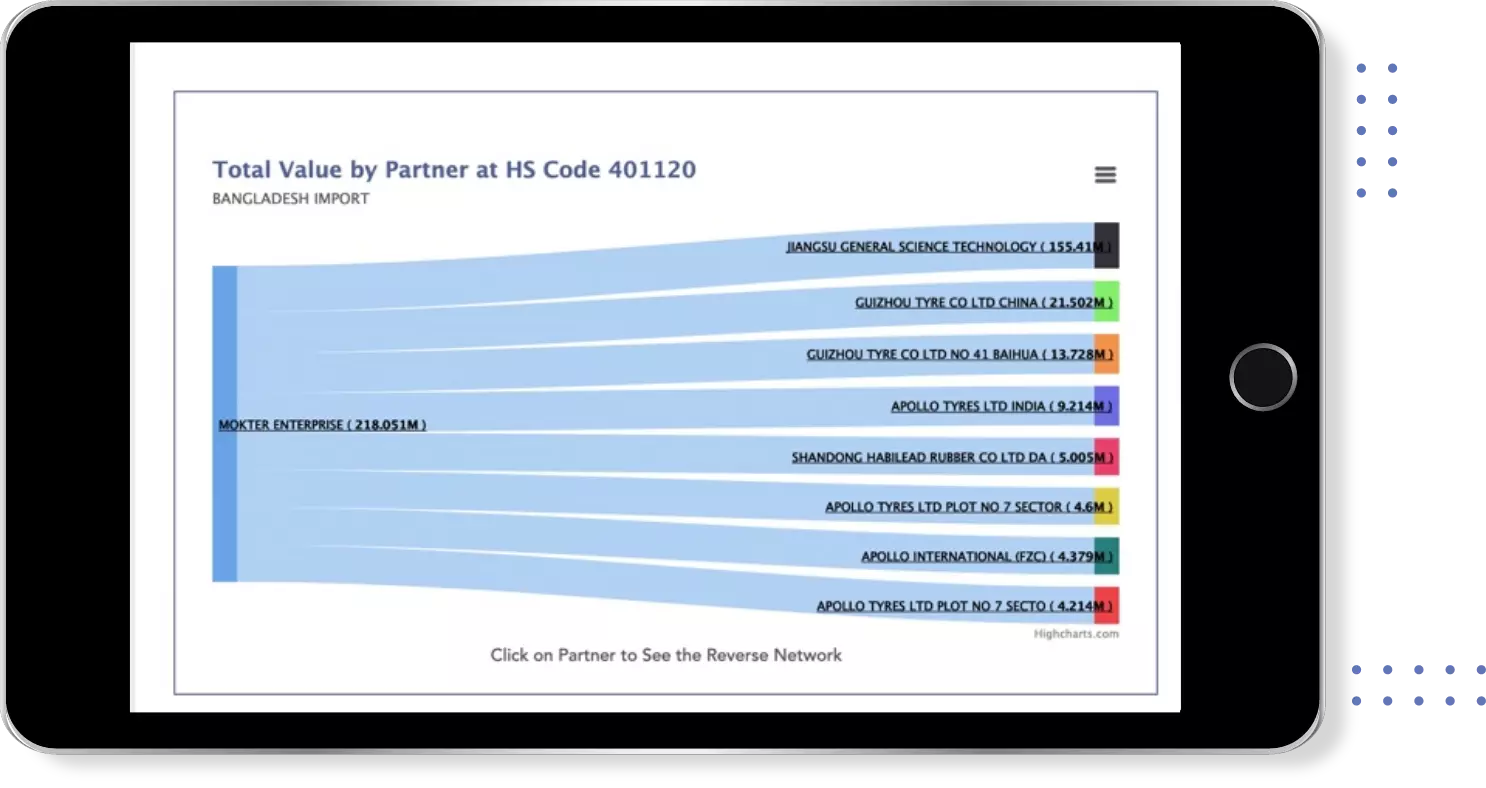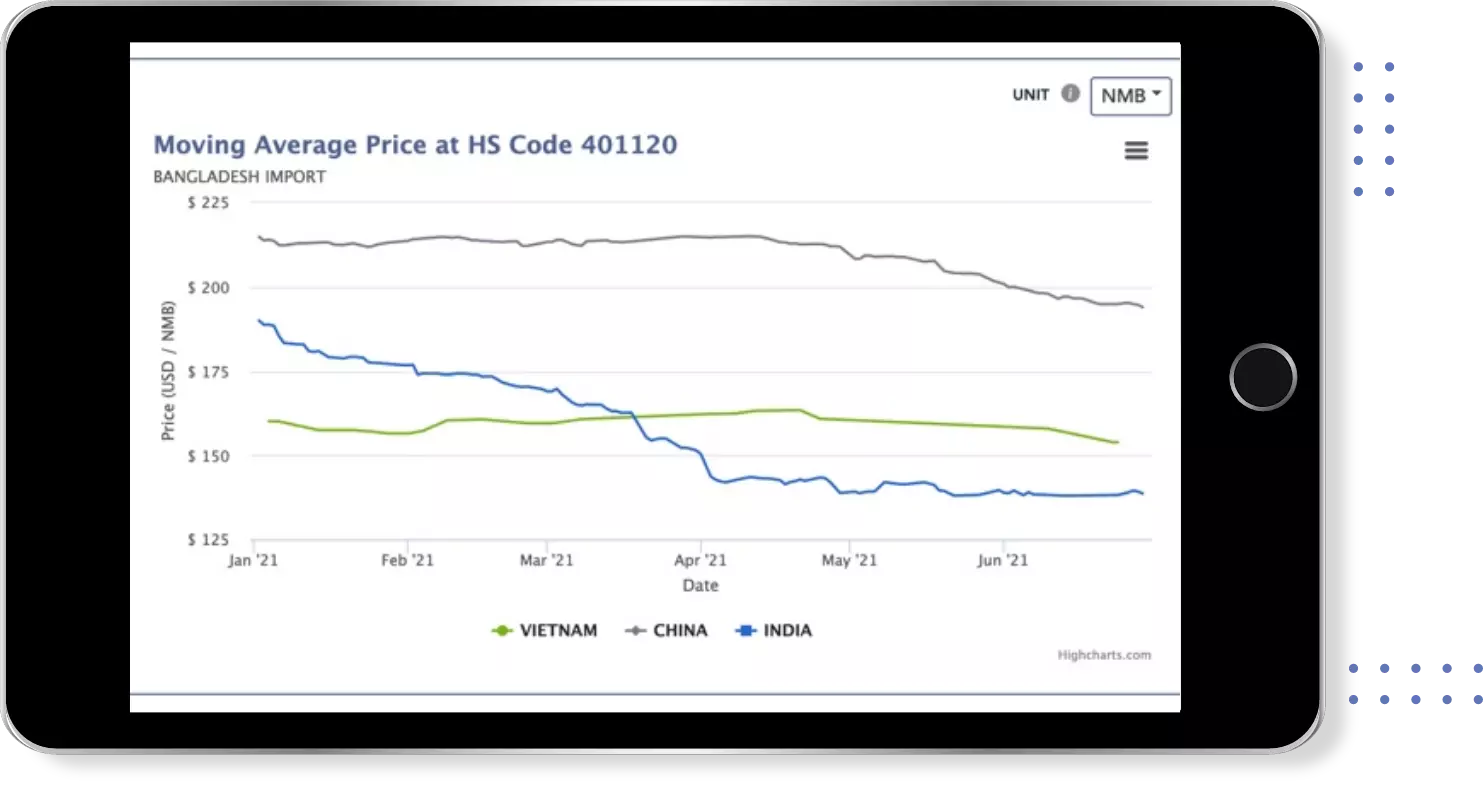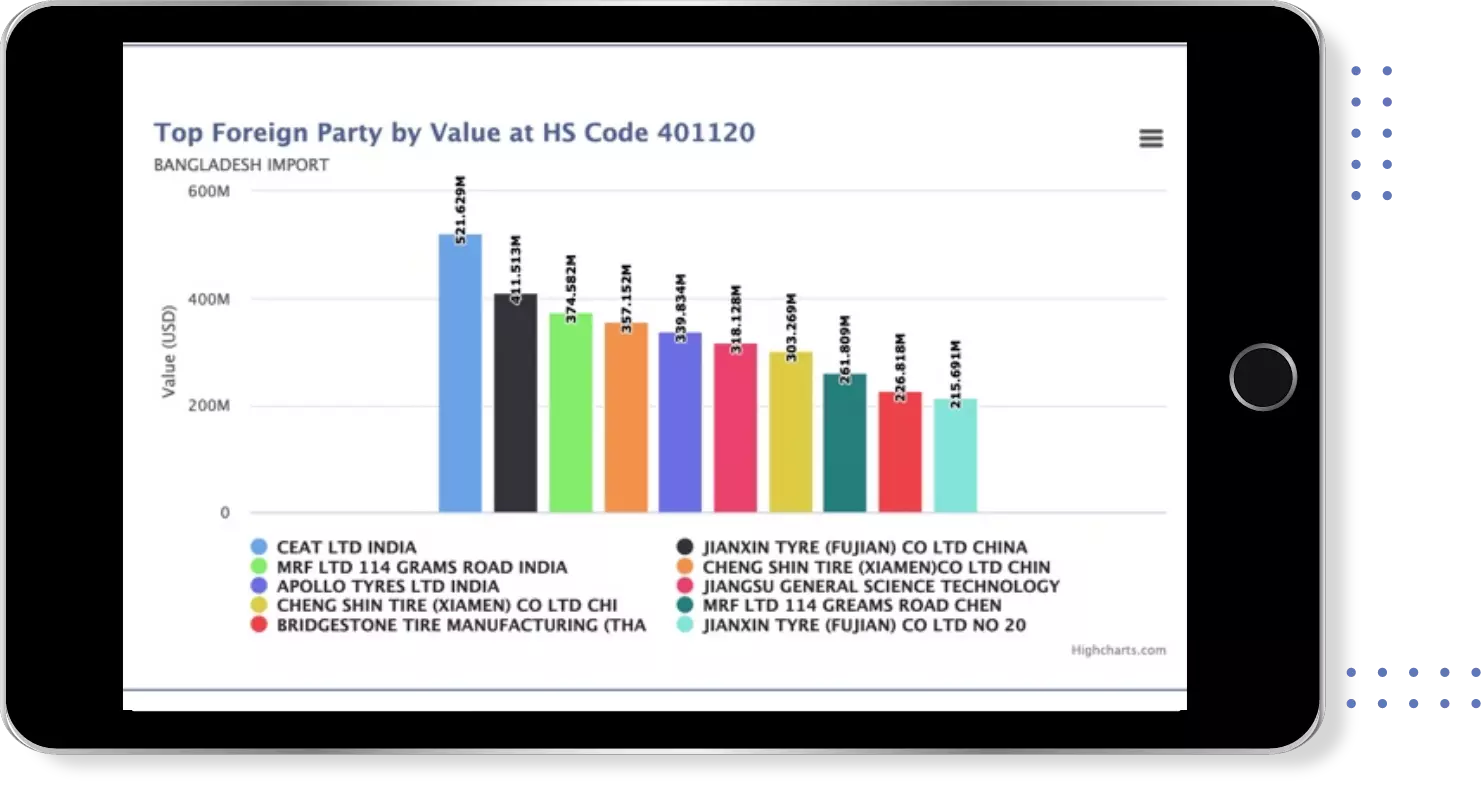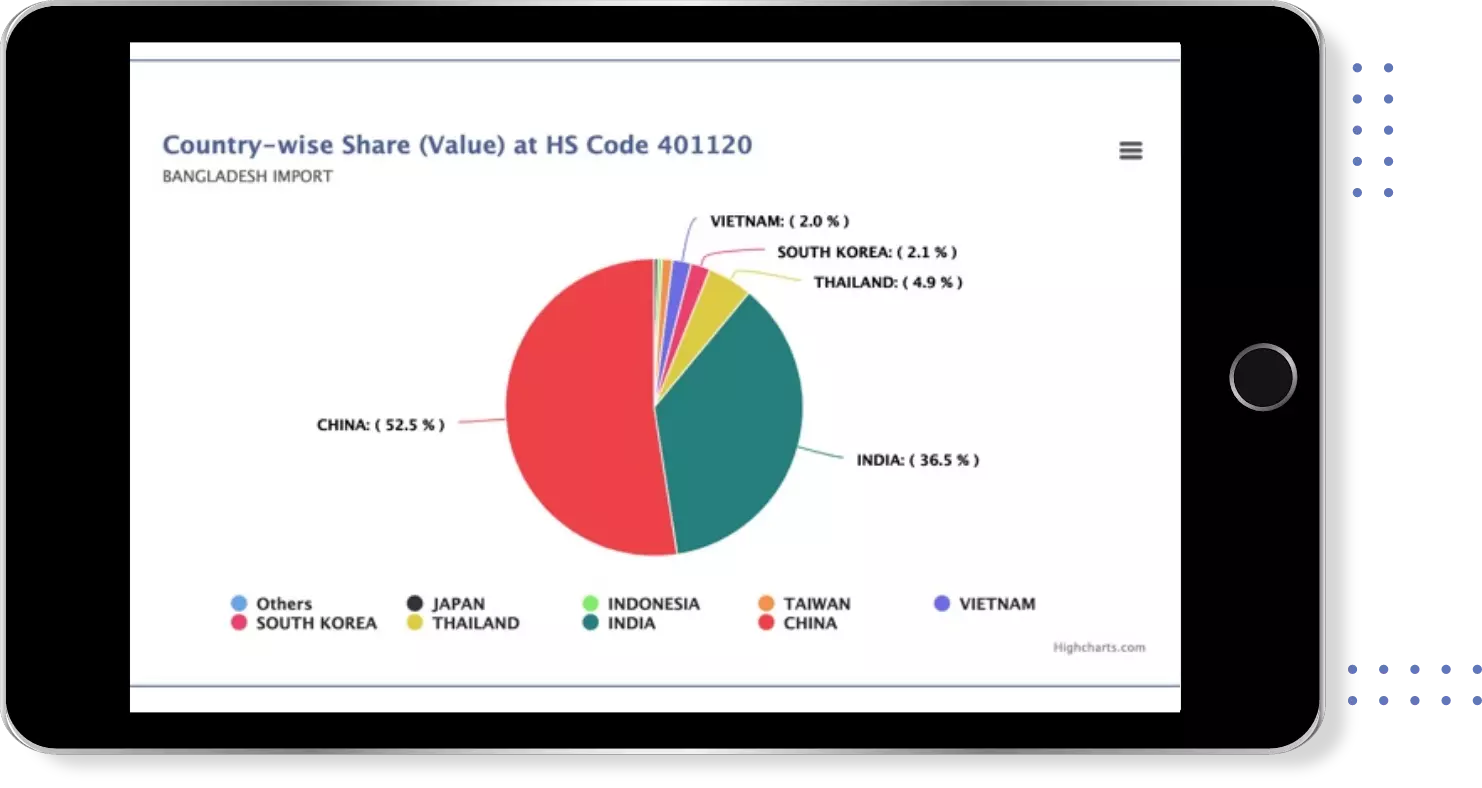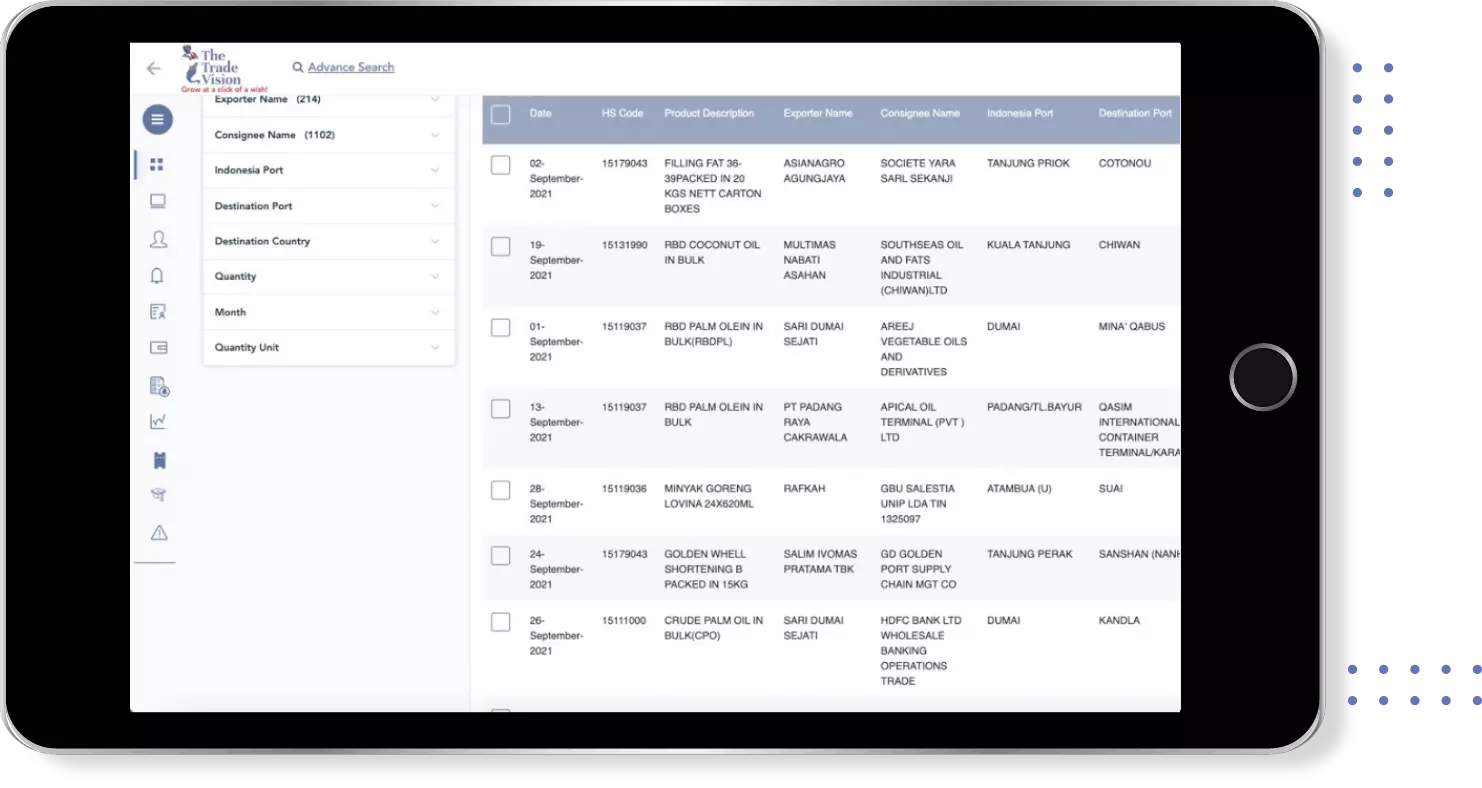
Find verified buyers and sellers of each capsule in 180+ countries along with their valid phone numbers and email ids.
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.
Enquiry