Find verified buyers and sellers of s pantoprazole sodium in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- s pantoprazole sodium buyers in vietnam
- COD Vietnam
Vietnam Buyers of s pantoprazole sodium
BRANCH OF ZUELLIG PHARMA VIETNAM LTD

VIETNAM
HS-Code
Products
3004909900
NEW. DRUG: PANTOLOC IV (PANTOPRAZOLE (AS PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG). BOX OF 1 VIAL OF LYOPHILIZED POWDER FOR INJECTION. LOT: 535250. NSX: JUNE 23 2022. NHH: DECEMBER 23 2023. VN-18467-14. NSX: TAKEDA GMBH
3004909900
NEW DRUG: PANTOLOC 40MG (PANTOPRAZOLE (AS PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG). BOX OF 1 BLISTER OF 7 TABLETS ENTERIC COATED TABLETS. LOT: 544676. NSX: MARCH 21 2023. NHH: MARCH 21 2026. VN-18402-14.
3004909900
NEW MEDICINE: PANTOLOC 40MG (PANTOPRAZOLE (PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG). BOX OF 1 BLISTER OF 7 ENTERIC-COATED TABLETS. LOT: 553084. MANUFACTURER: NOVEMBER 1 2023. NHH: NOVEMBER 1 2026. (VN-18402-14) 400110081723.
CAMEL INTERNATIONAL IMPORT EXPORT COMPANY LTD

VIETNAM
HS-Code
Products
300490990000
MEDICINE TO TREAT STOMACH DISEASES PANLOZ 40 (SODIUM PANTOPRAZOLE SESQUIHYDRATE 40MG PANTOPRAZOLE). REGISTRATION NUMBER: VN-16080-12. BOX OF 3 BLISTERS X 10 ENTERIC-COATED TABLETS. LOT: BT67K001. MANUFACTURER: JUNE 24 2023. HD:JUNE 22 2025. 100% NEW.
3004909900
STOMACH TREATMENT MEDICINE: TAVOMAC DR 40 (PANTOPRAZOLE (AS PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40MG). BOX OF 3 BLISTERS X 10 ENTERIC-COATED TABLETS. REGISTRATION NUMBER: VN-20309-17. LOT: EPB723004A MANUFACTURER: 07/25/23 HD: 07/24/26.
3004909900
PANTO-DENK 20 (PANTOPRAZOLE (AS PANTOPRAZOLE SODIUM SESQUIHYDRATE) 20MG. BOX OF 2 BLISTERS X 14 ENTERIC-COATED TABLETS. REGISTRATION NUMBER:VN-19143-15. HSX: ADVANCE PHARMA GMBH. LOT NUMBER MANUFACTURER HD: FILE ATTACH.
NAM DONG TRADING COMPANY LTD

VIETNAM
HS-Code
Products
3004909900
STOMACH MEDICINE NOLPAZA 20MG (PANTOPRAZOLE SODIUM SESQUIHYDRATE 20MG) BOX OF 2 BLISTERS X 14 TABLETS.VN-22133-19. BATCH NO: DD1319 MANUFACTURED IN AUGUST 2023 EXPIRED IN AUGUST 2026 100% NEW FOC PRODUCT.
3004909900
STOMACH MEDICINE NOLPAZA 20MG (PANTOPRAZOLE SODIUM SESQUIHYDRATE 20MG) BOX OF 2 BLISTERS X 14 TABLETS.VN-22133-19 MANUFACTURED IN AUGUST 2023 EXPIRY DATE IN AUGUST 2026 BATCH NO: DD1319 100% NEW PRODUCT.
3004909900
STOMACH MEDICINE NOLPAZA 20MG (PANTOPRAZOLE SODIUM SESQUIHYDRATE 20MG) BOX OF 2 BLISTERS X 14 TABLETS.VN-22133-19 MANUFACTURED IN AUGUST 2023 EXPIRY DATE IN AUGUST 2026 BATCH NO: DD1319 100% NEW FOC PRODUCT.
BRANCH OF ZUELLIG PHARMA VIETNAM CO LTD

VIETNAM
HS-Code
Products
3004909900
WESTERN MEDICINE: PANTOLOC I.V (PANTOPRAZOLE (PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG). BOX OF 1 BOTTLE OF FREE DRIED POWDER INJECTING. LOT: 532254. NSX: MARCH 16 2022. NHH: SEPTEMBER 16 2023. VN-18467-14. NSX: TAKEDA GMBH
3004909900
NEW DRUG: PANTOLOC IV (PANTOPRAZOLE (AS PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG). BOX OF 1 VIAL OF LYOPHILIZED POWDER FOR INJECTION. LOT: 533366. NSX: APRIL 26 2022. NHH: OCTOBER 26 2023. VN-18467-14. NSX: TAKEDA GMBH
3004909900
WESTERN MEDICINE: PANTOLOC I.V (PANTOPRAZOLE (PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40 MG). BOX OF 1 BOTTLE OF FREE DRIED POWDER INJECTING. LOT: 527518. NSX: 07/04/2022. NHH: OCTOBER 7 2023. VN-18467-14. NSX: TAKEDA GMBH
CTY TNHH DAVI

VIETNAM
HS-Code
Products
2930909000
PHARMACEUTICAL - PHARMACY: PANTOPRAZOLE SODIUM SESQUIHYDRATE - IN HOUSE BATCH NO: PAN / 0091021 MFG DATE: 10/2021 EXP DATE: 09/2024 NSX: PARTH OVERSEAS- INDIA.
2922490000
PHARMACEUTICAL - PHARMACY: PANTOPRAZOLE SODIUM SESQUIHYDRATE - IN HOUSE BATCH NO: PAN / 0091021 MFG DATE: 10/2021 EXP DATE: 09/2024 NSX: PARTH OVERSEAS- INDIA.
2922490000
PHARMACEUTICAL - PHARMACY: PANTOPRAZOLE SODIUM SESQUIHYDRATE - USP 43 BATCH NO: PPS / E-004/22 MFG DATE: 01/2022 EXP DATE: 12/2025 NSX: EVEREST ORGANICS LIMITED - INDIA. GPLH NUMBER: VD-28477-17
DAT VI PHU PHARMACEUTICAL JOINT STOCK COMPANY

VIETNAM
HS-Code
Products
2930909000
PHARMACEUTICAL INGREDIENTS - PHARMACEUTICAL INGREDIENTS: PANTOPRAZOLE SODIUM SESQUIHYDRATE - USP43 BATCH NO: PPS/E-003/23 MFG DATE: 01/2023 EXP DATE: 12/2026 NSX: EVEREST ORGANICS LIMITED- INDIA. GPLH NUMBER: VD-28477-17
2930909000
PHARMACEUTICAL RAW MATERIALS - PHARMACEUTICAL SUBSTANCE: PANTOPRAZOLE SODIUM SESQUIHYDRATE - USP44 BATCH NO: PPS/E-050/23 MFG DATE: 11/2023 EXP DATE: 10/2028 MANUFACTURER: EVEREST ORGANICS LIMITED- INDIA. LICENSE NUMBER: VD-28477-17
2930909000
PHA.RMACEUTICAL INGREDIENTS: PANTOPRAZOLE SODIUM SESQUIHYDRATE - USP43 BATCH NO: PPS/E-035/22 MFG DATE: 10/2022 EXP DATE: 09/2026 NSX:EVEREST ORGANICS LIMITED- INDIA. GPLH: VD-28477-17
DOMESCO

VIETNAM
HS-Code
Products
2930909000
PANTOPRAZOLE SODIUM USP43 LOT: BPZLUSP / 2107055 SX: 07/2021 HD: 06/2025 NSX: VASUDHA PHARMA CHEM LIMITED - INDIA.
2930909000
DRUG MATERIALS: PANTOPRAZOLE SODIUM USP43 LOT: BPZLUSP / 2112097 SX: 12/2021 HD: 11/2025 NSX: VASUDHA PHARMA CHEM LIMITED - INDIA.
2930909000
PANTOPRAZOLE SODIUM USP43 LOT: BPZLUSP / 2101005 SX: 01/2021 HD: 12/2024 NSX: VASUDHA PHARMA CHEM LIMITED - INDIA.
TV PHARM

VIETNAM
HS-Code
Products
2930909000
RAW MATERIALS FOR SX TAN PHARMACEUTICAL: PANTOPRAZOLE SODIUM SESQUIHYDRATE USP 43 NSX: METROCHEM API PRIVATE LTD LOT NUMBER: PSS / 2112063 SX: 11/2021 HD: 10/2026 SDK DRUGS: VD-33128-19
2930909000
RAW MATERIALS FOR TAN PHARMACEUTICAL: PANTOPRAZOLE SODIUM UPS USP43 NSX: VASUDHA PHARMA CHEM LIMITED LOT NUMBER: BPZLUSP / 212097 SX: 12/2021 HD: 11/2025 SDK: VD-34147-20 100% NEW GOODS
2930909000
RAW MATERIALS FOR SX TAN PHARMACY: PANTOPRAZOLE SODIUM SESQUIHYDRATE USP42. NSX: METROCHEM API PRIVATE LTD. LOT NUMBER: PSS / 2106041 NSX: 6/2021 HSD: 5/2026 SDK MEDICINE: VD-33128-19
DOMESCO MEDICAL IMPORT EXPORT JOINT STOCK CORPORATION

VIETNAM
HS-Code
Products
2930909000
RAW MATERIALS USED FOR DRUG PRODUCTION: PANTOPRAZOLE SODIUM USP43 LOT:PAS/2302120 SX: 02/2023 HD: 01/2028 NSX: METROCHEM API PRIVATE LIMITED-INDIA.
2930909000
INGREDIENTS: PANTOPRAZOLE SODIUM USP43 LOT: BPZLUSP/2206037 SX:06/2022 HD: 05/2026 NSX: VASUDHA PHARMA CHEM LIMITED - INDIA.
2930909000
RAW MATERIALS USED FOR DRUG PRODUCTION: PANTOPRAZOLE SODIUM USP43 LOT:PAS/2303193 PAS/2304246 SX: 03.04/2023 HD: 02.03/2028 NSX: METROCHEM API PRIVATE LIMITED-INDIA.
PHAM NGUYEN TRADING CO LTD

VIETNAM
HS-Code
Products
3822009000
Y0000835 PANTOPRAZOLE SODIUM SESQUIHYDRATE STANDARD REAGENT CRS (CAS: 164579-32-2) (20MG/VIAL) (FOR LABORATORY CHROMATOGRAPHY ANALYSIS NOT FOR MEDICAL USE)
3822009000
1494895 PANTOPRAZOLE SODIUM STANDARD CHEMICAL (CAS: 164579-32-2) (250MG/ VIAL) (FOR ANALYSIS ON THE CHROMATOGRAPH IN THE LABORATORY NOT FOR MEDICAL USE)
3822009000
149.4895 STANDARD CHEMICAL PANTOPRAZOLE SODIUM (CAS:164579-32-2) (250MG/VIAL) (NOT FOR MEDICAL USE AND PHARMACEUTICAL ONLY FOR LABORATORY USE 100% BRAND NEW)
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
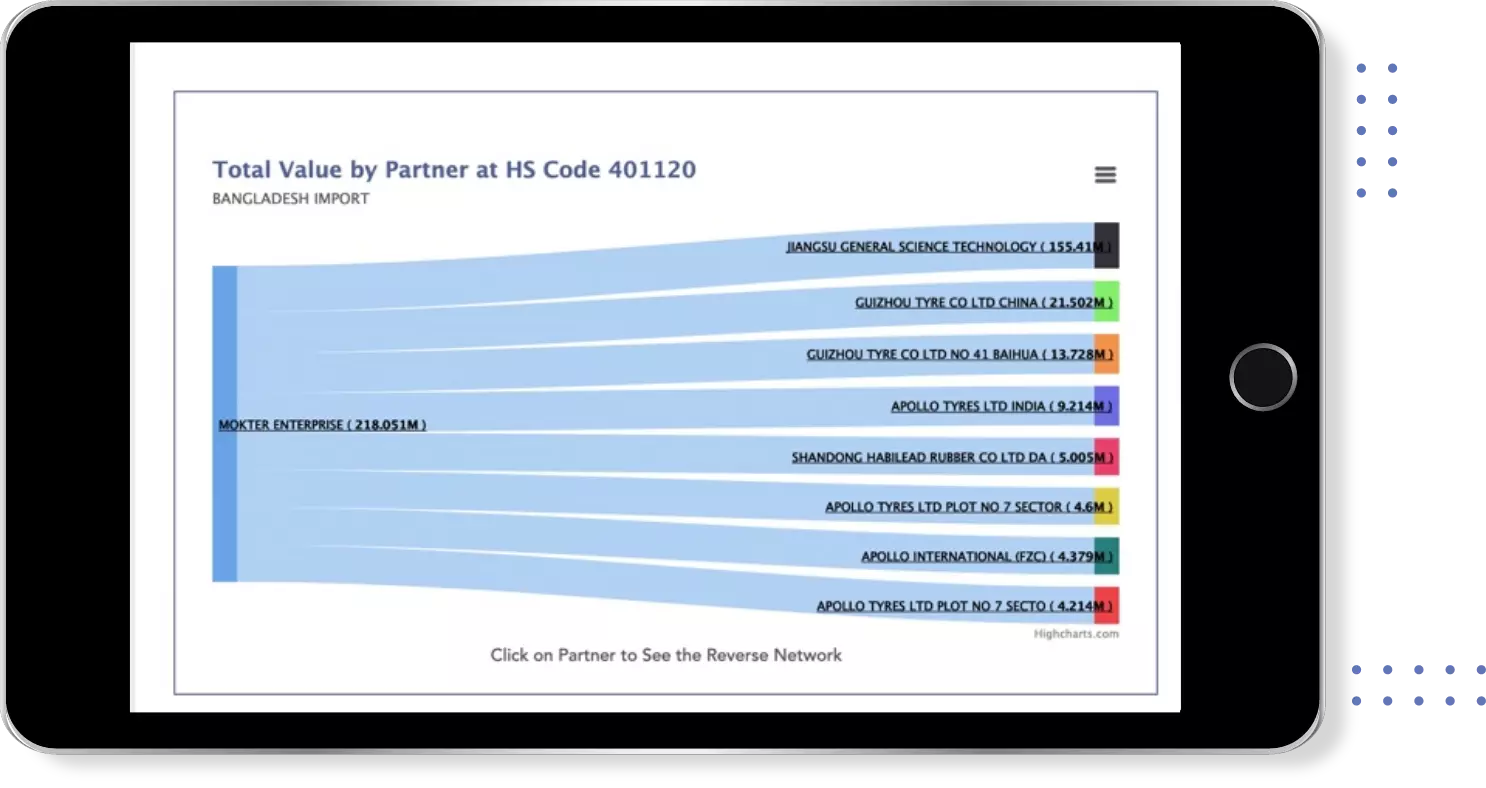
Get most profitable Buyers & Suppliers
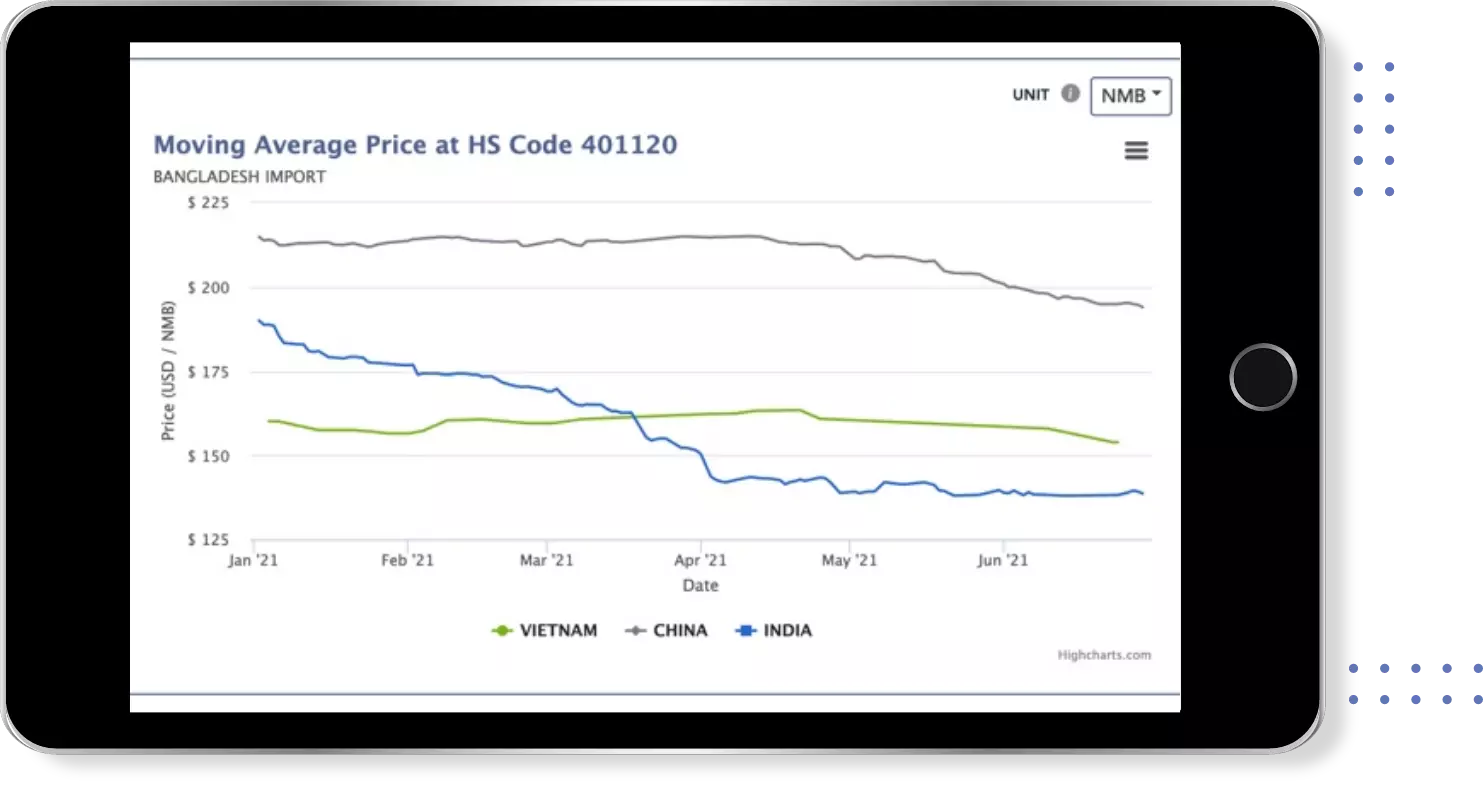
Instant insights on Price , Quantity & Value trends
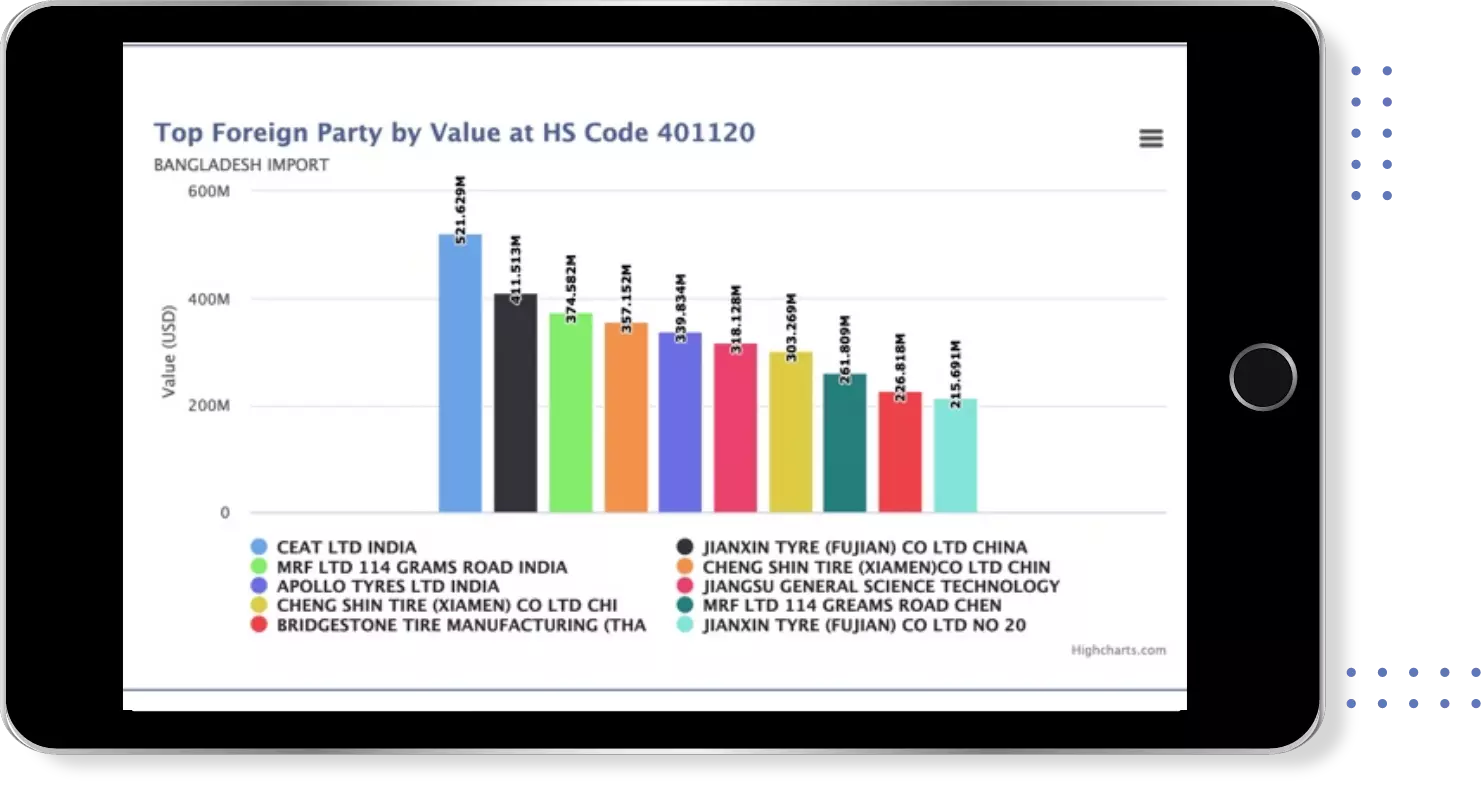
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
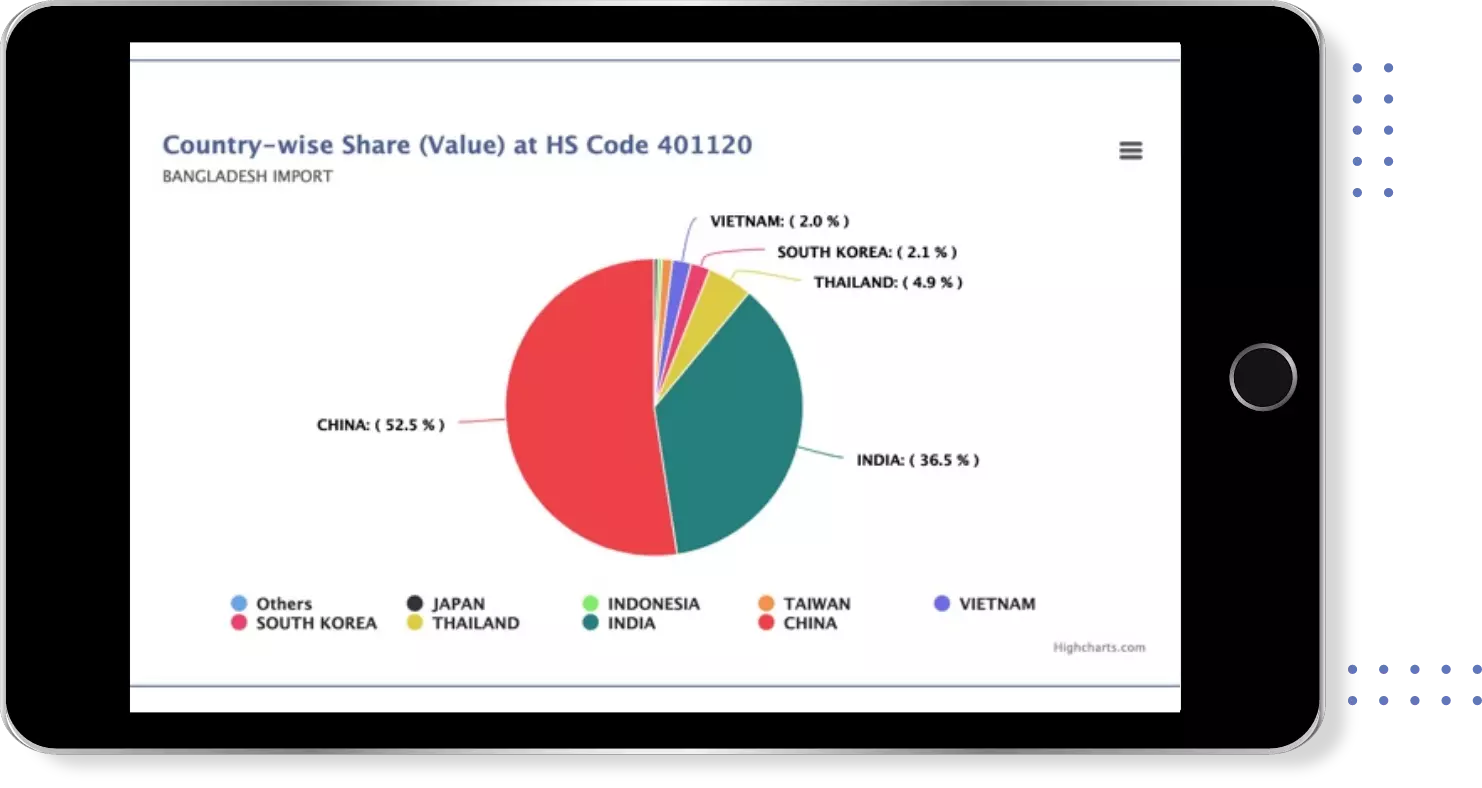
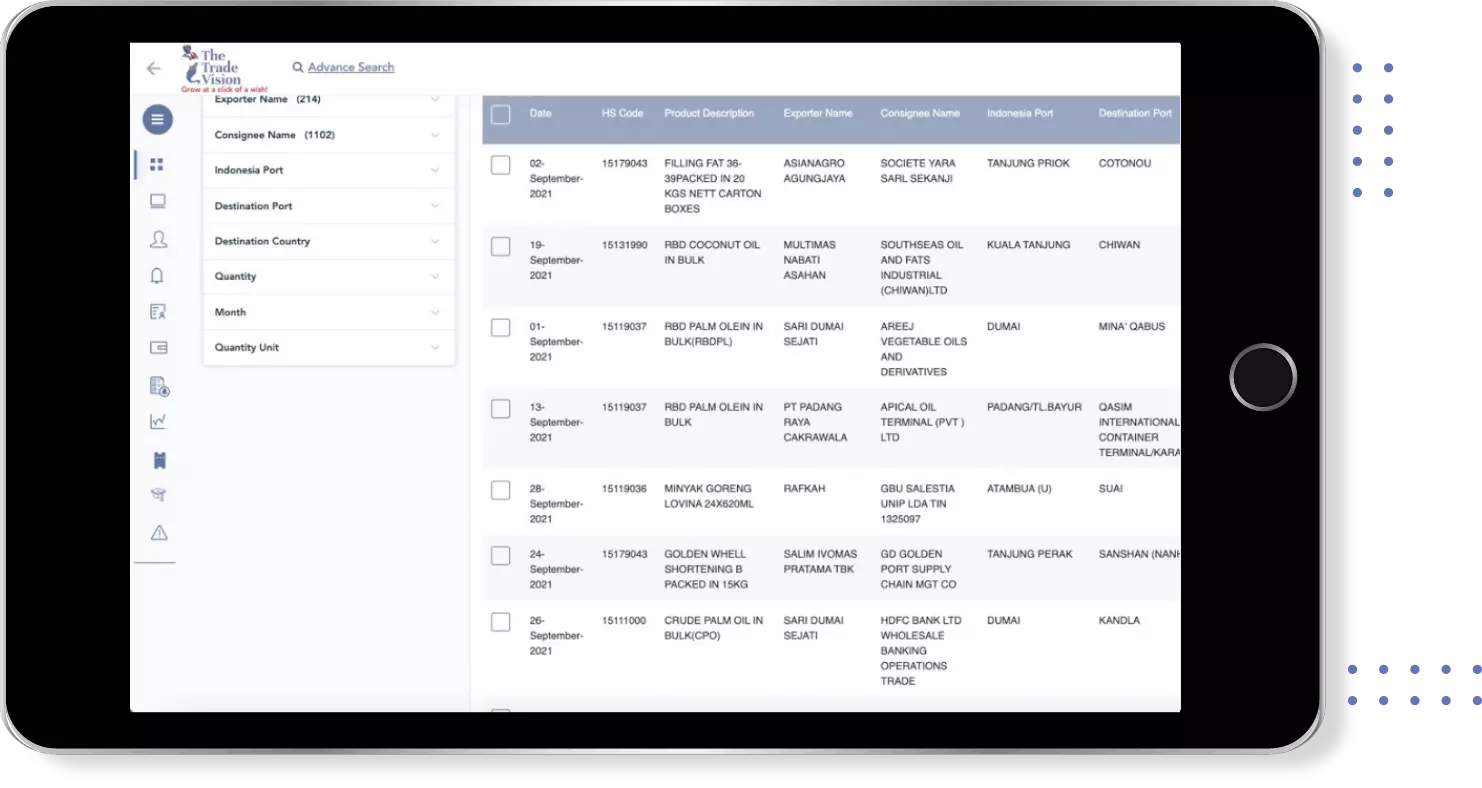
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.