Find verified buyers and sellers of s ibuprofen in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- s ibuprofen buyers in peru
- COD Peru
Peru Buyers of s ibuprofen
SANOFI AVENTIS DEL PERU S A

PERU
HS-Code
Products
3004902900
IBUPROFENO 1 00MG/5ML S/M S/M
3004902900
IBUPROFENO 600MG S / M S / M
3004902900
IBUPROFENO 400 MG S / M S / M
BONAPHARM SAC

PERU
HS-Code
Products
3004902900
DOLOCHECK PLUS 400 MG X 100 CA DOLOCHECK S / M LOTE: S212659 F.FAB. JUL 2021 F. VENC. JUN 2024 FOR HUMAN CONSUMPTION SOFT CAPSULES DOLOCHECK PLUS 400 MG X 100 SOFT CAP (IBUPROFEN 400 MG CAPSULES) = 120 BOXES
3004902900
DOLOCHECK PLUS 400 MG X 100 CA DOLOCHECK S / M LOTE: S212553 S212554 S212555 S212556 F.FAB. JUN 2021 F. VENC. MAY 2024 FOR HUMAN CONSUMPTION SOFT CAPSULES DOLOCHECK PLUS 400 MG X 100 SOFT CAP (IBUPROFEN 400 MG CAPSULES) = 390 BOXES
3004902900
PLIDOCHECK PLUS 400 CAPSULES PLIDOCHECK PLUS S/M LOT: S2220531 F. FAB. MAY 2022 EXPIRATION APR 2025 FOR HUMAN CONSUMPTION SOFT CAPSULE IN ALUMINUM/PVC/PVDC BLISTER PACKAGING PLIDOCHECK PLUS 400 MG X 100 CAPSULES (IBUPROFEN 400 MG CAPSULES) = 10 BOXES
FARMINDUSTRIA SA

PERU
HS-Code
Products
2916390000
IBUPROFEN RELATED COMPOUND S / M S / M
2916390000
IBUPROFEN USP S / M REFERENCE STANDARD CERTIFIED FOR LABORATORY USE IBUPROFEN (750 MG) 1335508
2916390000
IBUPROFEN S/M S/M LOT: C100-2210219M DUE 10.27.2027 INPUTS FOR USE IN THE PHARMACEUTICAL INDUSTRY IBUPROFEN USP 90002910
LABORATORIOS BAGO DEL PERU SA

PERU
HS-Code
Products
3004902900
ANAFLEX WOMAN NF 200MG ANAFLEX S / M LOT: 57160 F.PROD. 04/2021 F.VCTO. 04/2023 PHARMACEUTICAL USE PACKAGES COMMON NAME OF THE ACTIVE INGREDIENT: IBUPROFENO ANAFLEX WOMAN NF 200MG / HEALTH REGISTRY EE-00851
3004902900
ANAFLEX WOMAN NF 200MG ANAFLEX S / M LOT: 57502 F.PROD. 09/2021 F.VCTO. 09/2023 PHARMACEUTICAL USE PACKAGES COMMON NAME OF THE ACTIVE INGREDIENT: IBUPROFENO ANAFLEX WOMAN NF 200MG / HEALTH REGISTRY EE-00851
3004902900
ANAFLEX WOMAN NF 200MG ANAFLEX S / M LOT: 57503 F.PROD. 09/2021 F.VCTO. 09/2023 PHARMACEUTICAL USE PACKAGES COMMON NAME OF THE ACTIVE INGREDIENT: IBUPROFENO ANAFLEX WOMAN NF 200MG / HEALTH REGISTRY EE-00851
PERU LIFE ASSOCIATION

PERU
HS-Code
Products
3004902900
IBUPROFENO TEVA S / M LOT: L006010 F / V: 05/31/2022 100MG / 5ML. BOTTLE X 473 ML.
3004902900
IBUPROFENO S / M S / M LOT: OFK0834 FV: 05/31/2022 IBUPROFENO 100 MG / 5 ML SUSPENSION. BOTTLE X 240 ML. BOX X 24 BOTTLES
3004902900
IBUPROFENO S / M S / M LOT: L006010 FV: 05/31/2022 IBUPROFENO 100 MG / 5 ML SUSPENSION. BOTTLE X 473 ML. BOX X 12 BOTTLES
DRESDEN LAB SAC

PERU
HS-Code
Products
2916390000
IBUPROFEN POWDER S / MS / M MFG: 12/2020 EXP: 11/2025 BATCH: 4002/1101/20 / A-0165B // 0155B BATCH: 4002/1101/20 / A-0156B // 0158B IBUPROFEN USP 43 (60-80 MICRONS) PACKED IN 50 KGS
2916390000
IBUPROFEN POWDER S/MS/M BATCH NO:4002/1201/21/A-0195B 4002/1201/21/A-0196B 4002/1201/21/A-0197B 4002/1201/21/A-270 SUPPLIES FOR THE PHARMACEUTICAL INDUSTRY IBUPROFEN USP 43 (60-80 MICRONS) PACKED IN 50KGS NEW EXPORTWORTHY HDPE DRUMS
2916390000
IBUPROFEN POWDER 60-80 MICRONS - ICPL S/M S/M BATCH NO: 4002/1101/21/A-0145B 4002/1101/21/A-0146B 4002/1101/21/A-2188 INPUT FOR THE PHARMACEUTICAL INDUSTRY IBUPROFEN USP 43 (60-80 MICRONS) - IBUPROFEN POWDER 60-80 MICRONS - ICPL MFG DATE: AUG.- 2021 / RETEST DATE: JUL.-2026
HERSIL SA LABORATORIOS INDUSTRIALES PHARMACEUTICAL

PERU
HS-Code
Products
3822003000
IBUPROFENE IMPURITY B EP. MODEL: S / M REFERENCE STANDARD FOR LABORATORY ANALYSIS IBUPROFEN IMPURITY B BRAND: EP B1220000
2916390000
COMPOSITE IBUPROFEN RELATED USP S / M LOT: R09260 REFERENCE STANDARD FOR LABORATORY ANALYSIS COMPOSITE IBUPROFENE RELATED J ITEM NO: 1335596
2916390000
IBUPROFEN IMPURITY B BP. YE IBUPROFEN IMPURITY B REFERENCE STANDARD FOR LABORATORY ANALYSIS 880 UNITS
HC LEGD GROUP SAC

PERU
HS-Code
Products
3822009000
IBUPROFENO US PHARMACOPEIAL S / M IBUPROFEN COMPOUND RELATED C (BLISTER X 0.2 ML) REFERENCE STANDARD CERTIFICATES FOR LABORATORY TESTS IBUPROFEN RELATED COMPOUND C (0.2 ML / AMPULE; 3AMPULES) QA
3822009000
IBUPROFENO US PHARMACOPEIAL S / M
3822009000
IBUPROFENO CRJ US PHARMACOPEIAL S / M IBUPROFENO CRJ (VIAL X 25MG) REFERENCE STANDARD CERTIFICATES FOR LABORATORY TESTS IBUPROFEN RELATED COMPOUND J (25 MG)(2-(4-ISOBUTYRYLPHENYL)PROPANOIC ACID) QA
MERCK PERUANA SA

PERU
HS-Code
Products
2916390000
IBUPROFEN SIGMA ALDRICH S/M I4883-1G BATCH: SLBCH8939 RESEARCH 1 X 1G IBUPROFEN
2916390000
IBUPROFEN SIGMA ALDRICH S / M
2916390000
IBUPROFEN SIGMA ALDRICH S / M
LABORATORIOS PORTUGAL SRL

PERU
HS-Code
Products
2916390000
IBUPROFEN S/M S/M 100 DRUMS OF 50 KG EACH INPUTS FOR THE PHARMACEUTICAL INDUSTRY IBUPROFEN USP 43 BATCH: 4002/1201/23/A-0066B 4002/1201/23/A-0067B 4002/1201/23/A-0071B 4002/1201/23/A-0075B 4002/1201
2916390000
IBUPROFEN S/M S/M BATCH: 23057349 DATE OF MANUFACTURING: 2023-05-09 EXPITY DATE: 2027-05 INPUTS FOR THE PHARMACEUTICAL INDUSTRY IBUPROFEN IN 20 DRUMS WITH 25KG EACH
2916390000
IBUPROFEN S/M S/M BATCHS:2303742 2303747 2303776 2303777 MFG.DATE:2023-03-11-12-14 EXPIRY DATE: 2027-03 INPUTS FOR THE PHARMACEUTICAL INDUSTRY IBUPROFEN USP2022 IN 200 DRUMS WITH 25KG EACH
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
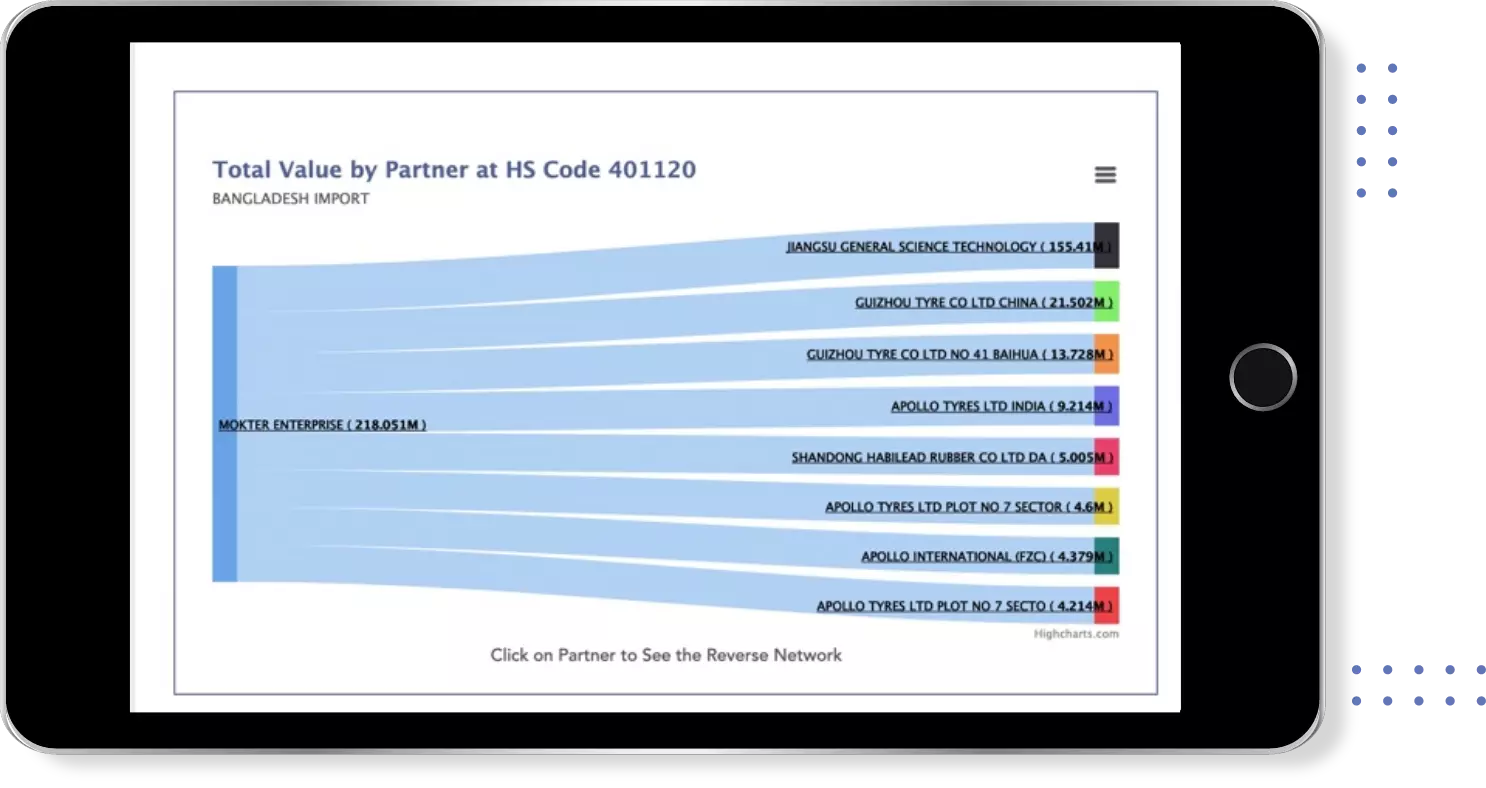
Get most profitable Buyers & Suppliers
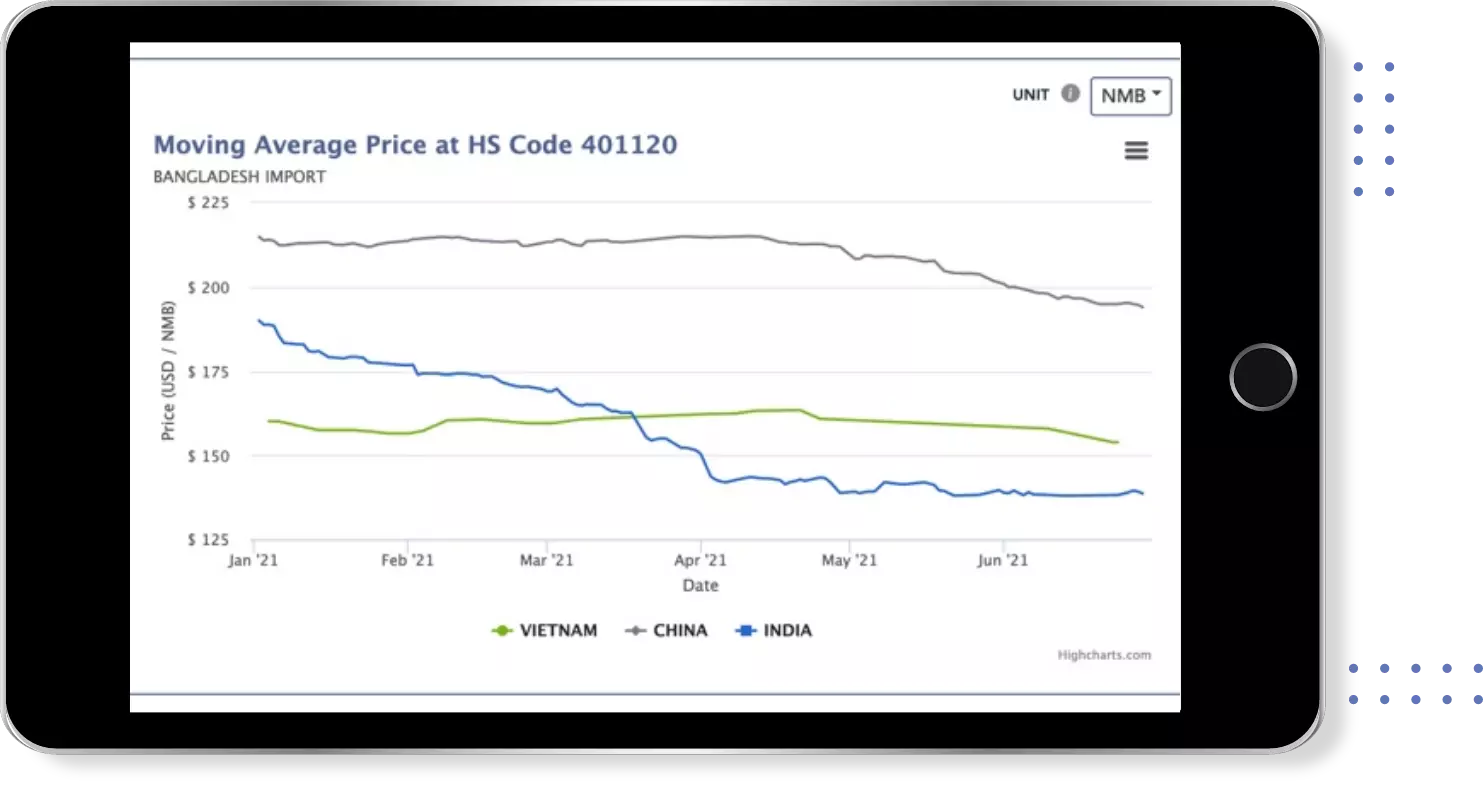
Instant insights on Price , Quantity & Value trends
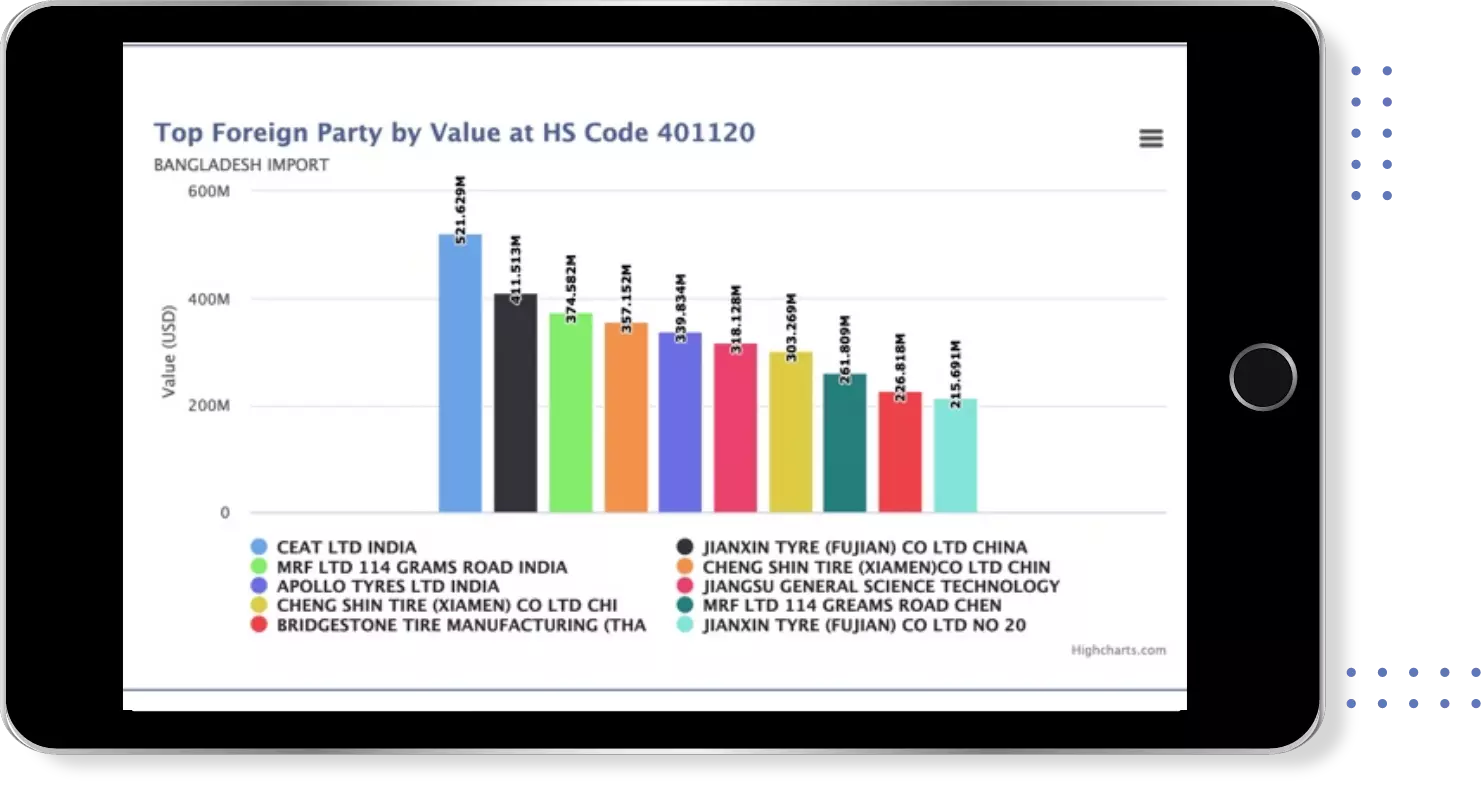
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
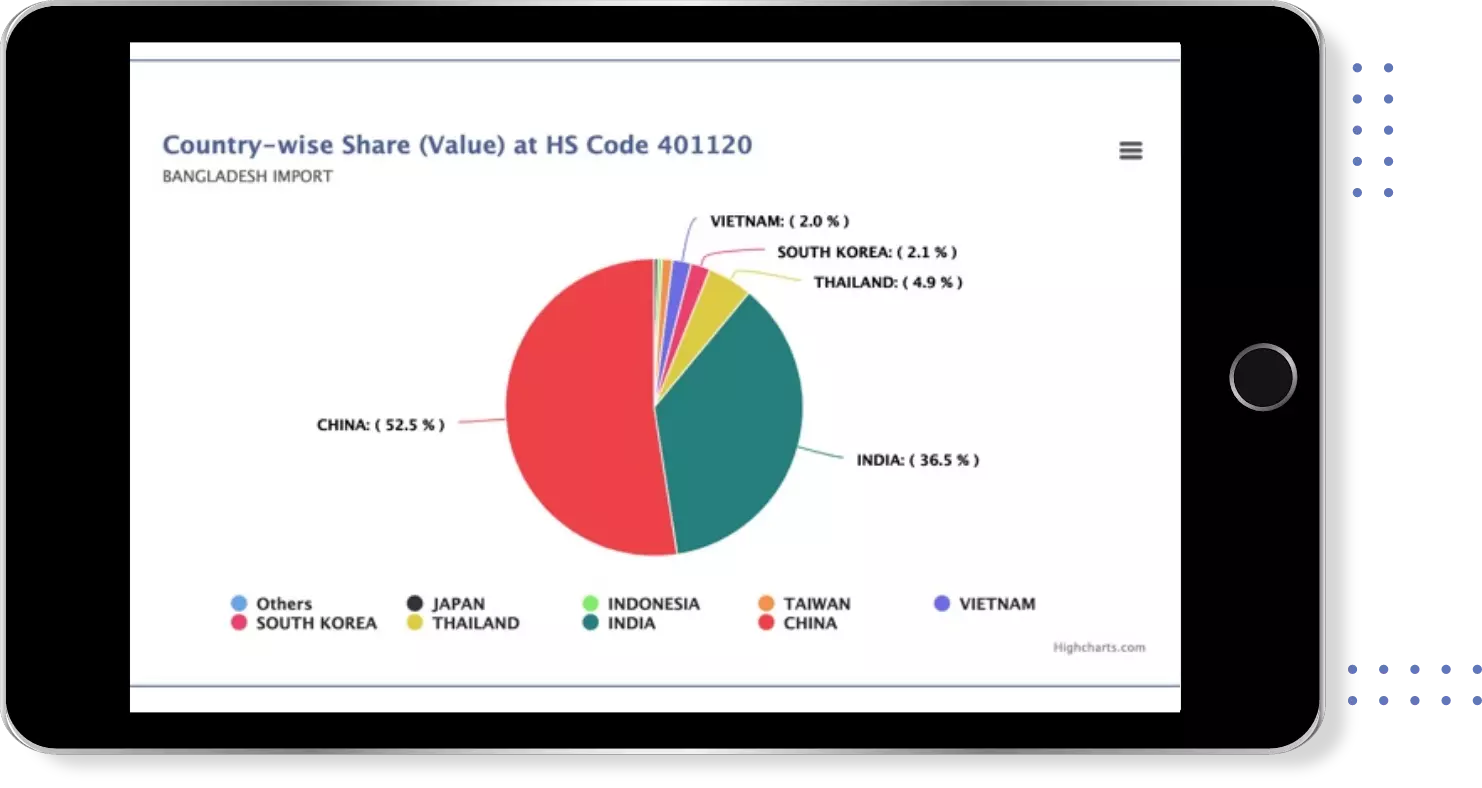
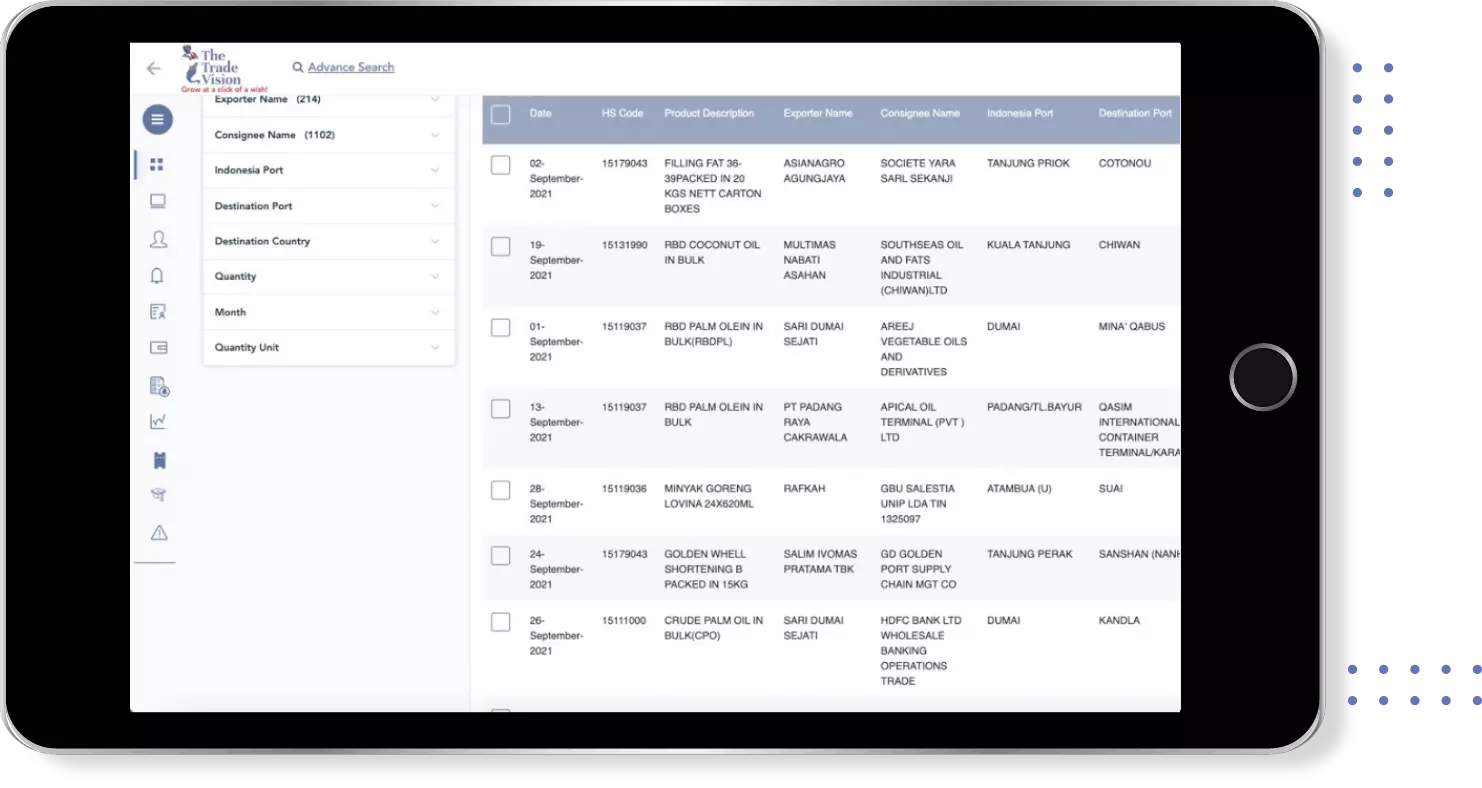
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.