Find verified buyers and sellers of quantitative pcr in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- quantitative pcr import data uzbekistan
Applied Filters
- COD Uzbekistan
Import Data by Country
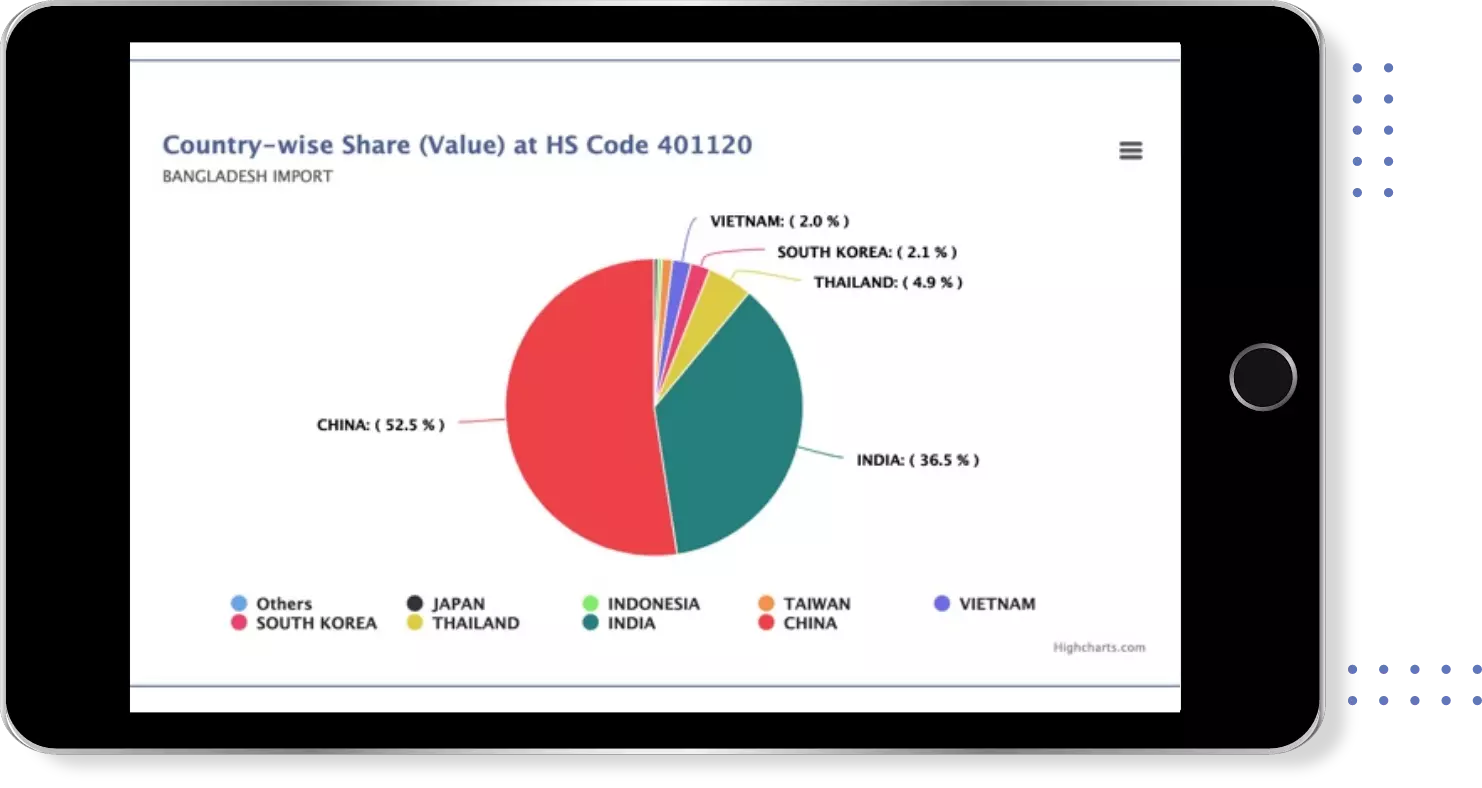
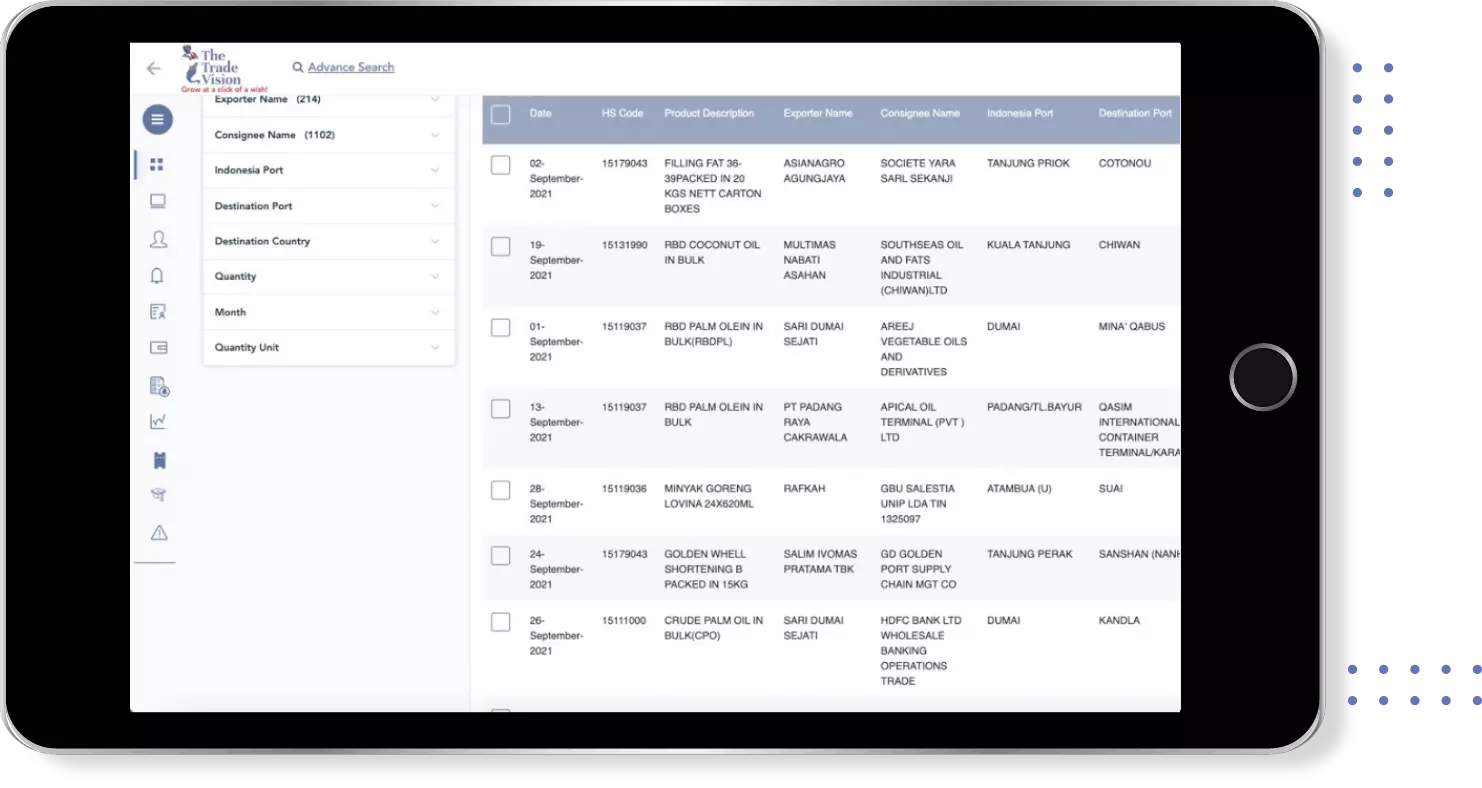
Uzbekistan Import Trade Data for quantitative pcr
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 UZBEKISTAN UZBEKISTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
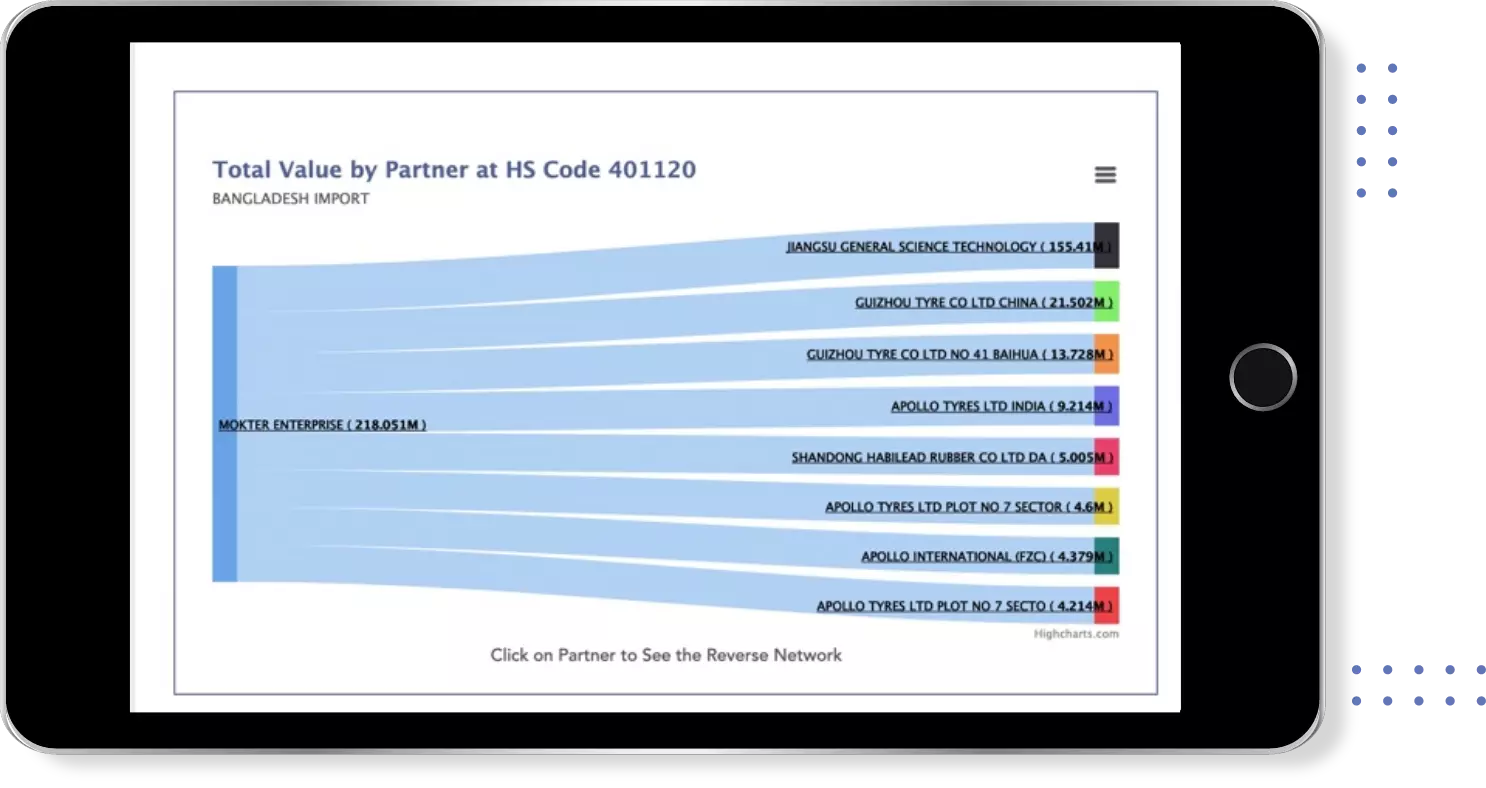
Get most profitable Buyers & Suppliers
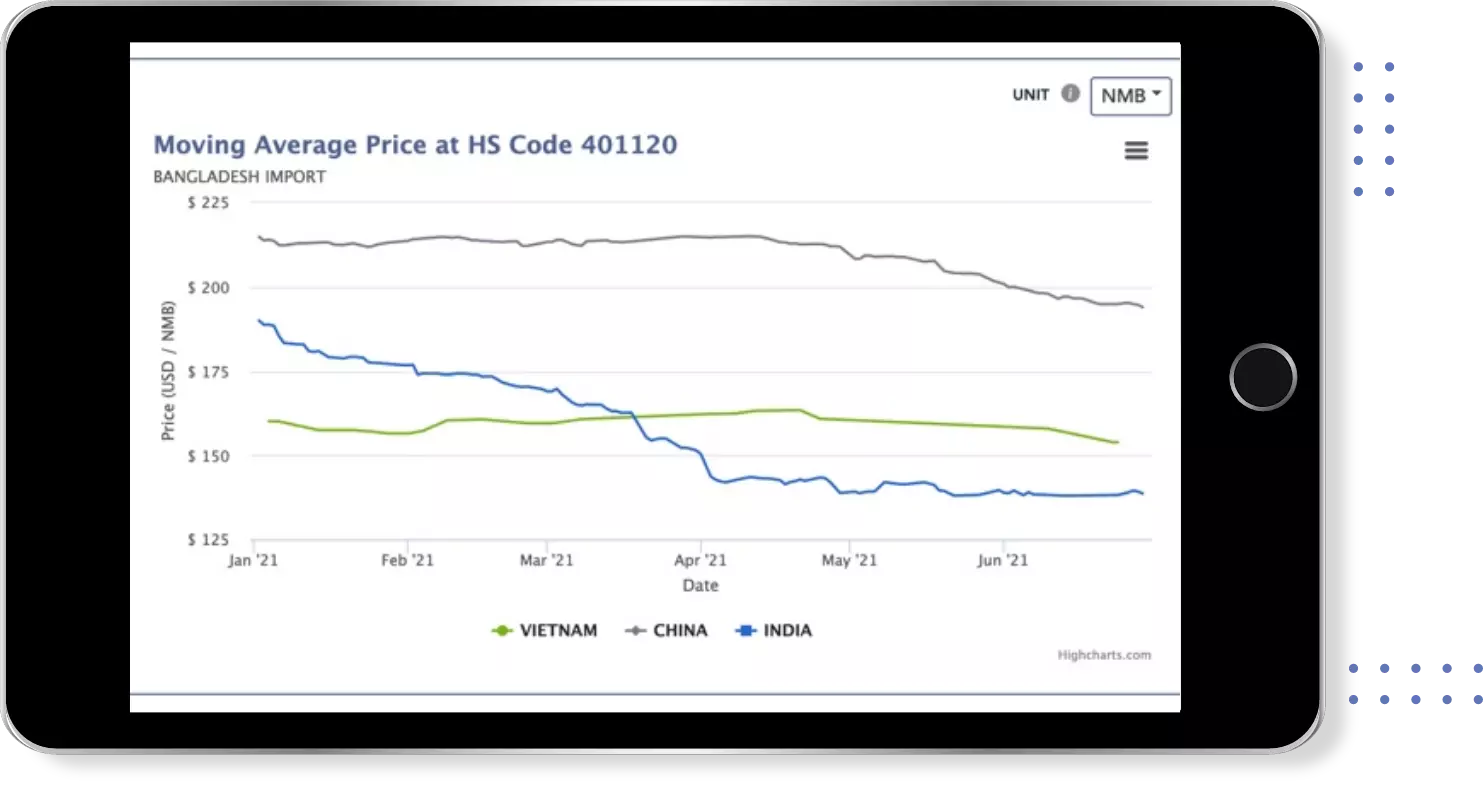
Instant insights on Price , Quantity & Value trends
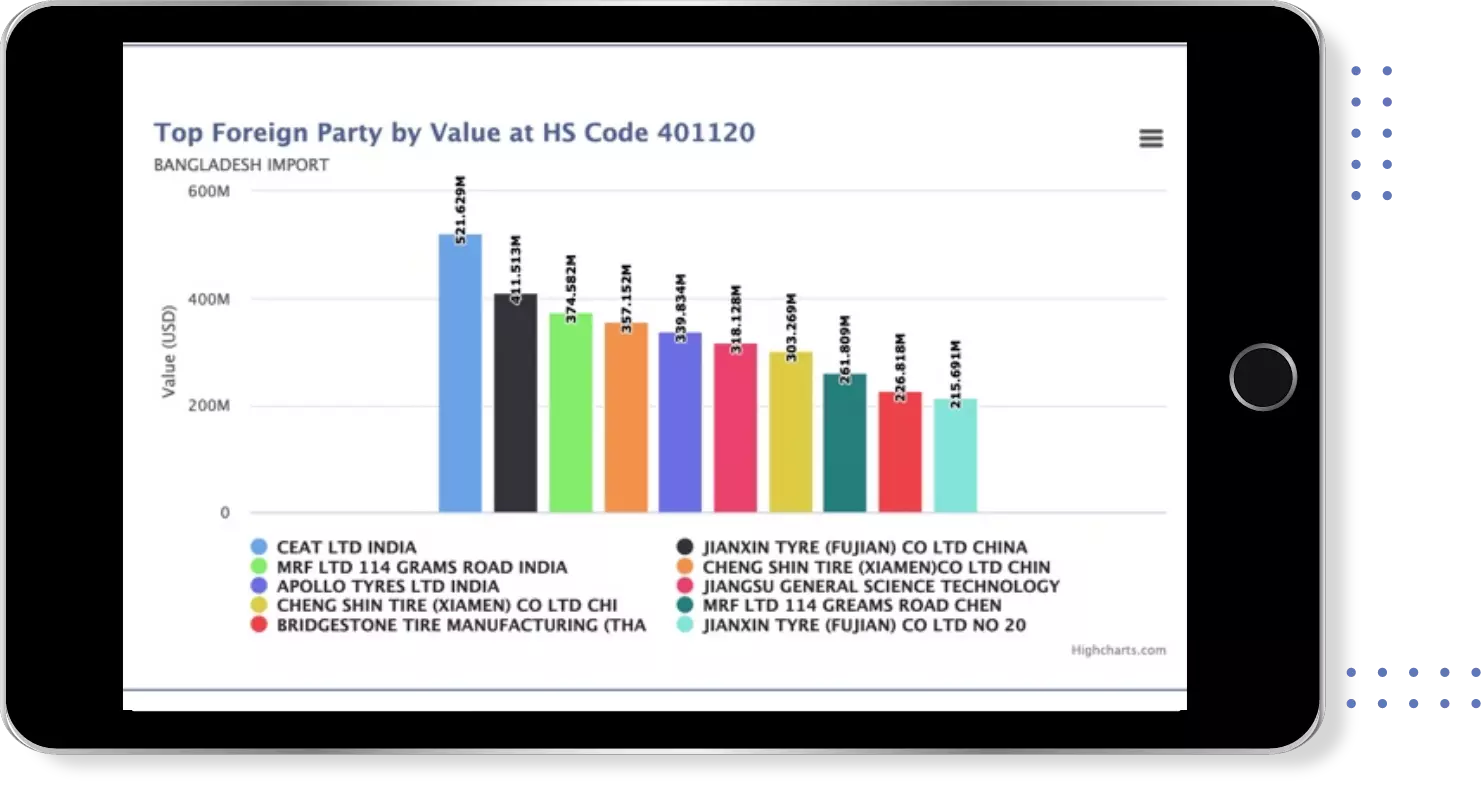
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.
Enquiry