Find verified buyers and sellers of i pill tablets in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- i pill tablets suppliers in ukraine
- COO Ukraine
Ukraine Suppliers of i pill tablets
GALAXY PHARM LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINAL PRODUCTS FOR PEOPLE DISPOSED FOR RETAIL TRADE: DONORMIL TABLETS COVERED BY 1 5MG BY 30 TABLES BY 1 TUBE BY 1 TUBE TO 1 TUBE SERIES: X31 1 9. DONORMIL PLUGS 1 5MG 1 0 TABLES IN A TUBE OF 2 TUBES IN A PAPER BOX-480UP. SERIES: X1 658. REFAL RELATED EFFALGANS FOR 80MG ?1 0 (5X2) IN BLISTERS - 360UP. SERIES: T7980. REDACTAL EFFERALGAN RECTAL FOR 300MG 5 SUPPLIERS IN A BLISTER 2 BLISTERS IN A CARDBOARD-1 89UP. SERIES: T9609. ETHERALGAN 500MG EFFERVESCENT TABLETS 4 PILLS PER STRIP; 4 STRIPS IN A CARTON BOX-500UP. SERIES: X3271 T8400. EFFERALGAN ORAL SOLUTION 3% 90 ML IN VIALS # 1 WITH A MEASURING BED IN A CARDBOARD BOX-800UP. SERIES: X3423. MANUFACTURER: 'UPSA SAS' FRANCE
3004900000
1. MEDICINAL PRODUCTS FOR PEOPLE DISPOSED FOR RETAIL TRADE: DONORMIL TABLETS COVERED 1 5MG 30 TABLES 1 TUBE -1 SERIES: X31 1 8. DONORMIL TABLETS COVERED 1 5MG 1 0 TABLES IN A TUBE 1 TUBE IN A PAPER BOX -4050UP. SERIES: X1 582. UPSARIN UPSA 500MG PILLETS PLUSPY ON 500MG 4 TABLETS IN THE STRIP 4 STRIPS IN THE CARDBOX-5000UP. SERIES: X2229. MANUFACTURER: 'UPSA SAS' FRANCE
3004900000
1. MEDICINAL PRODUCTS FOR PEOPLE DISPOSED FOR RETAIL TRADE: DONORMIL TABLETS COVERED BY 1 5MG 1 0 TABLES IN A TUB BY 160 TUBES SERIES: X1 582. DONORMIL TABLETS COVERED BY 1 5MG 30 TABLES IN A TUBE 1 TUBE IN A PAPER BOX -900UP. SERIES: X31 1 9. ETHERALGAN SUPPOSITORIES RECTAL ON 1 50MG ?1 0 (5X2) IN BLISTERS-600UP. SERIES: T8506. ETHERALGAN 500MG EFFERVESCENT TABLETS 4 PILLS PER STRIP; FOR 4 STRIKS IN A CARTON BOX-300UP. SERIES: X3271. EFFERALGAN ORAL SOLUTION 3% 90 ML IN VIALS # 1 WITH A MEASURING BED IN A CARDBOARD BOX-400UP. SERIES: X3423. MANUFACTURER: 'UPSA SAS' FRANCE.
BANNERMAN WOLD CLASS PHARM UKRAINE LTD

UKRAINE
HS-Code
Products
3004500000
1. MEDICINES FOR PEOPLE CONTAINING VITAMINS FOR THERAPEUTIC USE PACKAGED FOR RETAIL SALE. BRONHIPRET ® DROPS FOR INGESTION OF 50 ML IN A VIAL NO. 1 IN A CARDBOARD BOX SERIES 0000124763 - 210 PACKAGES. IMUPRET ® DROPS. FOR INGESTION OF 50 ML NO. 1 IN A VIAL 1 VIAL IN A CARTON BOX SERIES 0000128907 - 350 PACKAGES. IMUPRET® PILLS NO. 50 (25X2) IN BLISTERS IN A CARTON BOX SERIES 0000129444 - 150 PACKS. ® TP COATED TABLETS 20 TABLETS IN A BLISTER 1 BLISTER IN A CARTON SERIES 0000134696 - 450 PACKS. SYNUPRET ® SYRUP 100 ML NO. 1 IN A VIAL 0000134250 SERIES - 324 PACKS. FOR INGESTION OF 100 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A CARTON BOX SERIES 0000133791 - 250 PACKAGES. SINUPRET® JELLY ? 50 (25X2) IN BLISTERS CA.
3004500000
1. MEDICINES FOR PEOPLE CONTAINING VITAMINS INTENDED FOR THERAPEUTIC USE PACKAGED FOR RETAIL SALE. IMUPRET ® DROPS FOR INGESTION OF 50 ML NO. 1 IN A VIAL 1 BOTTLE IN A CARTON SERIES 0000128907 - 210 PACKAGES. IMUPRET® PILLS NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000129444 - 150 PACKS. ® TP TABLETS COATED NO. 20 IN BLISTERS IN A CARDBOARD BOX SERIES 0000134696 - 485 PACKS. SYNUPRET ® SYRUP 100 ML NO. 1 IN A VIAL SERIES 0000134250 - 120 PACKS. IMUPRET ® DROPS FOR INGESTION OF 100 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A CARTON BOX SERIES 0000133791 - 120 PACKAGES. SINUPRET® DRAGEE NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000134884 - 1350 PACKAGES. SINUPRET® EXTRACT DRAGEE NO. 20 (10X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000135360 - 450 UNITS
3004500000
1. MEDICINES FOR PEOPLE CONTAINING VITAMINS INTENDED FOR THERAPEUTIC USE PACKAGED FOR RETAIL SALE. IMUPRET ® DROPS FOR INGESTION OF 50 ML NO. 1 IN A VIAL 1 BOTTLE IN A CARTON BOX SERIES 0000128907 - 120 PACKAGES. ® PILLS NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000129444 - 150 PACKAGES. BRONHIPRET ® SYRUP LIQUID FOR INGESTION OF 50 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A BOX SERIES 0000131750 - 350 PACKAGES. DROPS FOR INTAKE OF 100 ML NO. 1 IN A VIAL IN A CARDBOARD BOX SERIES 0000133389 - 55 PACKAGES. BRONHIPRET ® DROPS FOR INGESTION OF 50 ML IN A VIAL NO. 1 IN A CARDBOARD BOX SERIES 0000134175 - 200 PACKAGES. BRONHIPRET® TP COATED TABLETS 20 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX69 SERIES 00001 600 PACKAGES. SYNUPRET ® SYRUP ON 100 ML NO. 1 IN A BOTTLE SERIES 0000134250 - 300 PACKAGES. IMUPRET ® DROPS FOR INGESTION ON 100 ML NO. 1 IN A BOTTLE
JSC PHARMAC

UKRAINE
HS-Code
Products
3004900000
1.PRESENTS FOR PEOPLE NOT IN AEROSOL PACKING: AMIZON KIDD SYRUP 10MG / ML IN VIALS 100 ML IN PACKING ?1 -108UP. SERIES60719.DYEING DROPS FOR INTERNAL USE IN VIALS 25ML IN PACKING ?1 -360UP. 0 1G IN BLISTERS OF PACKING ?10X1 -512UP. SERIES 240619.DIAZOLIN PILLS 0 05G IN BLISTERS OF PACKING ?10?2 -432UP. SERIES 310418.DIAZOLIN PILLS 0 1G IN BLISTERS IN PACKING ?10?2 -1 008??. SERIES 230519 # 390619. DIALIBON TURBO SOLUTION FOR INFUSIONS 1.2% IN 50 ML VIALS IN PACK NO. 10 -54 UNIT. SWELLING 25MG / ML IN 2 ML AMPOULES IN BLISTERS IN A PACKAGE ?5X2 -400UP. SERIES 20119. CORVALOL DROPS FOR INTERNAL USE IN 25 ML VIALS IN A PACKAGE ?1 -20 040UP. SERIES 570819. MILT NASAL SPRAY IN 10 ML VIALS IN A PACKAGE ?1 -180UP. SERIES 110619. RHEUMOXICAM SOLUTION FOR INJECTION 15MG / 1.5ML IN AMPOULES 1.5ML IN BLISTERS IN A PACKAGE ?5X1 -540UP. SERIES 210619. URONEPHRON TABLETS COATED WITH FILMS
3004900000
1. MEDICINES FOR PEOPLE NOT IN AEROSOL PACKING: AMIZON KIDD SYRUP 10MG / ML IN VIALS 100 ML IN PACKING ?1 -360UP. SERIES60719.DIAZOLIN 100 TABLETS 0 1G IN BLISTERS PACKING ?10X1 -648UP. SERIES 240619.DIAZOLIN DRAY 05G IN BLISTERS OF PACKING ?10?2 -288UP. SERIES 310418.DIAZOLIN PILLS 0 1G IN BLISTERS OF PACKING ?10?2 -2 160??. SERIES 390619.CORVALOL DROPS FOR INTERNAL USE IN 25 ML BOTTLES IN PACKING ?1 -12 000??. SERIES 580819 PECTOLVAN IVY SYRUP IN 100 ML VIALS IN PACK ?1 -216UP. SERIES 90219. RHEUMOXIC SOLUTION FOR INJECTION 15MG / 1.5ML IN AMPOULES 1.5ML IN BLISTERS IN PACKING ?5X1 -36 0UP. SERIES 210619. URONEPHRON TABLETS FILM-COATED TABLETS IN BLISTERS IN PACKING ?10X6 -168UP. SERIES 150619. HEPILOR ORAL RINSE SOLUTION IN BOTTLES 100 ML IN PACK ?1 -108UP. SERIES 70619. HEPILOR ORAL MUCOSA IN VIALS OF 20 ML IN PACKING ?1 -210UP. SERIES 170619. EUCASOLIN AQUA SPRAY NASAL 1 MG / G IN VIALS WITH DISPENSER 10G IN UPA
3004900000
1.CHECKS FOR PEOPLE NOT IN AEROSOL PACKING: VALIDOL TABLETS BILINGUAL0.06G IN BLISTERS ?10 -31 000UP. SERIES1590819.KEVER PILLS COVERED WITH A 25MG SHELL IN BLISTERS IN A PACKAGE ?10X1 -300UP. PECTOLVAN IVY SYRUP IN 100 ML VIALS IN PACKAGE ?1 -2 096UP. SERIES 90219. RHEUMATIC AGENTS SOLUTION FOR INJECTION 15MG / 1.5ML IN AMPOULES 1.5IL IN BLISTERS IN PACKAGE NO. 5X1 -1 080UP. SERIES 210619. URONEPHRON TABLETS FILM-COATED TABLETS IN BLISTERS IN A PACKAGE OF ?10X6 -462UP. SERIES 150619. HE ILOR MOUTHWASH SOLUTION IN 100 ML VIALS IN PACK NO. 1 -642UP. SERIES 70619. HEPILOR SPRAY FOR ORAL MUCOUS MEMBRANE IN 20 ML VIALS IN PACK NO. 1 -3 132UP. SERIES 170619. EUCASOLIN AQUA SPRAY MG / ML G IN VIALS WITH DISPENSER 10G IN PACKING ?1 -3 380UP. SERIES 530519. MANUFACTURER: JSC PHARMAK UA MANUFACTURER COUNTRY: UA. TRADEMARK: PHARMAC.
CFC BIOKON LLC

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR HUMANS: -EXFORGE H FILM-COATED TABLETS 5MG / 160MG / 12.5MG 14 TABLETS IN A BLISTER# 2 BLISTERS IN A CARTON (ACTIVE INGREDIENTS: 1 TABLET CONTAINS: AMLODIPINE BESYLATE6.94MG EQUIVALENTS 5 EQUIV VALSARTAN 160MG IHYDROCHLOROTHIAZIDE 12.5MG SERIES: BJY56-25UP MANUFACTURER: PRODUCTION QUALITY CONTROL PACKAGING SERIES RELEASE: NOVARTIS PHARMACEUTICALS SA - GALVUS 50MG TABLETS 14X2 / GALVUS PILLS 2MG 14GGS : 1 TABLET CONTAINS: VILDAGLIPTIN 50MG) SERIES: BJM91 -45UP SERIES: BKY45 -35UPMANUFACTURER: NOVARTIS ARMASTICS SA - DIOVAN 160MG FILM-COATED TABLETS ?14 / DIOVAN 160MG TABLETS FILM-COATED FILM ?14 (IN CARTON) (ACTIVE SUBSTANCES: VALSARTAN160MG / TABLET) SERIES: BKK78 -400UP-EXFORGE H 10MG / 160MG / 12.5MG FILM-COATED TABLETS 14X2 / EXFORGE H FILM-COATED TABLETS 10MG / 160MG / 12.5MG 14X2 (ACTIVE INGREDIENTS: 1 TABLET CONTAINS: 13.87MG AMLODIPINE EQUIVALENT EQUIVALENT TO
3004900000
1. MEDICINES FOR HUMANS: -EXFORGE H FILM-COATED TABLETS 5MG / 160MG / 12.5MG 14 TABLETS IN A BLISTER# 2 BLISTERS IN A CARTON (ACTIVE INGREDIENTS: 1 TABLET CONTAINS: AMLODIPINE BESYLATE6.94MG EQUIVALENTS 5 EQUIV VALSARTAN 160MG IHYDROCHLOROTHIAZIDE 12.5MG) SERIES: BJY56-40UP MANUFACTURER: PRODUCTION QUALITY CONTROL PACKAGING SERIES RELEASE: NOVARTIS PHARMACEUTICALS SA - GALVUS 50MG TABLETS 14X2 / GALVUS PILLS2M 50G : 1 TABLET CONTAINS: VILDAGLIPTIN 50MG) SERIES: BKY45 -250UPMANUFACTURER: NOVARTIS PHARMACEUTICALS .A - DIOVAN 160MG FILM-COATED TABLETS ?14 / DIOVAN 160MG TABLETS FILM-COATED FILM ?14 (IN CARTON) (ACTIVE SUBSTANCES: VALSARTAN160MG / TABLET) SERIES: BKK78 -150UP- SERIES: BLD98 -150UP- DIOVAN 80MG FILM-COATED TABLETS ?14 / DIOVAN 80MG TABLETS FILM-COATED TABLETS ?14 (IN CARTON) (ACTIVE SUBSTANCES: WALSARTAN80MG / TABLET) SERIES: BKX97 -100UP-EXFORGE 10MG / 160MG FILM-COATED
3004900000
1.PHARMACEUTICALS: -SIRDALUD (R) 2MG TABLETS 10 TABLETS IN A BLISTER# 3 BLISTERS IN A CARTON BOX- SERIES: KL424 -100UPMANUFACTURER: NOVARTIS SAGLIK GUIDA VE TARIM URUNLERI SAN TIC. .S. TURKEYCOUNTRY OF PRODUCTION: TRTAR MARK: NOVARTIS SIRDALUD-VOLTAREN 50MG ENTERIC-COATED TABLETS ?20 / VOLTAREN 50MG TABLETS IN INTESTINAL SHELL ?20 (IN CARDBOARD PACKAGING) - SERIES: KN211-1ARUP: NOVARTIS SAGLIK GUIDES VE TARIM URUNLERI SAN VE TICAS) TURKEYCOUNTRY OF PRODUCTION: TRTRADE MARK: NOVARTIS-VOLTAREN (R) RAPID TABLETS SUGAR COATED 50MG PER 10 PILLS BLISTER# 2 BLISTERS IN A CARTON BOX- SERIES: KL777 -200UP-VOLTAREN (R) TABLETS RESISTANT TO 25MG 10 PILLS IN A BLISTER# 3 BLISTERS IN A CARTON BOX-2 SERIES: K -70UPMANUFACTURER: NOVARTIS URUNLERI TURKEYCOUNTRY OF PRODUCTION: TRTAR MARK: NOVARTIS VOLTAREN
BANNERMAN GOLD CLASS FARM UKRAINE LLC

UKRAINE
HS-Code
Products
3004400000
1. MEDICINAL PRODUCTS FOR THE PEOPLE CONTAINING ALKALOIDS INTENDED FOR THERAPEUTIC USE PACKAGED FOR RETAIL SALE. CYCLODINON ® TABLETS COATED NO. 30 IN BLISTERS IN A CARTON BOX SERIES 0000121782 - 50 PACKS. TONZIPRET® TABLETS NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000128604 - 100 PACKAGES. KANEFRON® N DROPS FOR INGESTION 100 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A CARTON BOX SERIES 0000133677 - 300 PACKAGES. KANEFRON ® H PILLS ?60 (20?3) IN BLISTERS IN A CARDBOARD BOX SERIES 0000138561 - 2000 PACKAGES . MASTODINON® TABLETS ? 60 (20X3) IN BLISTERS IN A CARDBOARD BOX SERIES 0000137574 - 200 PACKAGES. TRADE MARK - BIONORICA. MANUFACTURER - BIONORICA SE. COUNTRY OF PRODUCTION - DE. DOES NOT CONTAIN ANY DRUGS OR PRECURSORS. NOT IN AEROSOL DISPENSER.
3004500000
1. MEDICINES FOR PEOPLE CONTAINING VITAMINS INTENDED FOR THERAPEUTIC USE PACKAGED FOR RETAIL SALE. IMUPRET ® DROPS FOR INGESTION OF 50 ML NO. 1 IN A VIAL 1 BOTTLE IN A CARTON SERIES 0000128907 - 210 PACKAGES. IMUPRET® PILLS NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000131308 - 150 PACKAGES. SYNUPRET® PILLS NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000134884 - 1200 PACKAGES. 20 (10X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000135360 - 450 PACKAGES. BRONHIPRET ® SYRUP LIQUID FOR INGESTION OF 50 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A BOX SERIES 0000134830 - 490 PACKAGES. INTAKE OF 100 ML NO. 1 IN A VIAL IN A CARDBOARD BOX SERIES 0000136055 - 600 PACKAGES. BRONHIPRET® TABLETS COATED WITH NO. 20 IN BLISTERS IN A CARDBOARD THE FIRST BOX SERIES 0000137710 - 330 UPAKOVOK.SYNUPRET® FORT NUMBER 20 PILLS IN A BLISTER IN A CARDBOARD BOX SERIES 0000140222 - 990
3004500000
1. MEDICINES FOR PEOPLE CONTAINING VITAMINS INTENDED FOR THERAPEUTIC USE PACKAGED FOR RETAIL SALE. IMUPRET ® DROPS FOR INGESTION OF 50 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A CARTON SERIES 0000128907 - 60 PACKAGES. ® SYRUP 100 ML NO. 1 IN A BOTTLE SERIES 0000134250 - 200 PACKAGES. IMUPRET ® DROPS FOR INGESTION OF 100 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A CARTON SERIES 0000133791 - 200 PACKAGES. SINUPRET® PILLS NO. 50 (25X2) IN BLISTERS IN A CARDBOARD BOX SERIES 0000134884 - 800 PACKS. LIQUID FOR INGESTION OF 50 ML NO. 1 IN A BOTTLE 1 BOTTLE IN A BOX SERIES 0000134830 - 120 PACKAGES. ® TP COATED TABLETS 20 TABLETS IN A BLISTER 1 BLISTER
PHARM START LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE. -KLIVAS FILM-COATED TABLETS 10 MG.30 10 TABLETS IN A BLISTER# 3 BLISTERS IN A CARDBOARD BOX -480UP.SERIES 700319 PRODUCTION DATE 26.03.2019 SHELF LIFE 04.2021R. COMPOSITION FOR ONE TABLET OF ACTIVE SUBSTANCES: ROSUVASTATIN CALCIUM 10 4MG (IN TERMS OF ROSUVASTATIN 10MG). PRODUCTION DATE 25.06.2019 SHELF LIFE 07.2021.COMPOSITION PER TABLET OF ACTIVE SUBSTANCES: ROSUVASTATIN CALCIUM 20.8MG (IN (FOR ROSUVASTATIN 20MG) .- RISOPTAN NO. 9 10 MG TABLETS 3 TABLETS IN A BLISTER 3 BLISTERS IN A PACK OF CARDBOARD -461UP. SERIES 980619. PRODUCTION DATE 06/20/2019. SHELF LIFE 07.2022R. ACTIVE INGREDIENT PILL: RISATRIPTAN BENZOATE 14.53 MGNOT IN AEROSOL DISPENSER. WITHOUT NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS.
3004900000
1. MEDICINES FOR PEOPLE. -KLIVAS FILM-COATED TABLETS 10 MG.30 10 TABLETS IN A BLISTER# 3 BLISTERS IN A CARTON -600UP.SERIES 700319 DATE OF PRODUCTION 26.03.2019 SHELF LIFE 04.2021R. COMPOSITION FOR ONE TABLET OF ACTIVE SUBSTANCES: ROSUVASTATIN CALCIUM 10 4MG (IN TERMS OF ROSUVASTATIN 10MG). PRODUCTION DATE 25.06.2019 SHELF LIFE 07.2021.COMPOSITION PER TABLET OF ACTIVE SUBSTANCES: ROSUVASTATIN CALCIUM 20.8MG (IN (FOR ROSUVASTATIN 20MG) .- RISOPTAN NO. 9 10 MG TABLETS 3 TABLETS IN A BLISTER 3 BLISTERS IN A PACK OF CARDBOARD -300UP. SERIES 980619. PRODUCTION DATE 06/20/2019. SHELF LIFE 07.2022R. ACTIVE INGREDIENT PILL: RISATRIPTAN BENZOATE 14.53 MGNOT IN AEROSOL DISPENSER. WITHOUT NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS CONTENT. MANUFACTURER: PHARMA START LLC U.A.
3004900000
1. MEDICINES FOR PEOPLE. -KLIVAS TABLETS COVERED BY FILM SHEET 1 0MG. # 30 PO1 0 TABLETS IN THE BLISTER; 3 BLISTERS IN A CARDBOARD PACK -300UP. SERIES 70031 9 PRODUCTION DATE 26. 03. 201 9P. EXPIRY DATE 04. 2021 R. COMPOSITION PER ONE TABLET OF ACTIVE SUBSTANCES: ROSUVATATIN CALCIUM 1 0 4MG (IN RELATION TO ROSUVATATIN 1 0MG). -KLIVAS TABLETS COVERED 20MG. # 30 BY 1 0TABLETS IN THE BLISTER; 3 BLISTERS IN A CARDBOARD PACK -300UP. SERIES 03071 9 PRODUCTION DATE 25. 06. 201 9P. EXPIRY DATE 07. 2021 R. COMPOSITION PER ONE TABLE OF ACTIVE SUBSTANCES: ROSUVATATIN CALCIUM 20.8MG (IN RELATION TO ROSUVATATIN 20MG). - RIZOPTAN ?9 1 MG TABLETS. 3 PILLS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX -400UP. 98061 SERIES 9. PRODUCTION DATE 20. 06. 201 9. EXPIRY DATE 07. 2022 COMPOSITION ON ONE TABLET OF ACTIVE SUBSTANCES: RISATRIPTANE BENZOATE 1 4.53 MNE IN AEROSOL PACKAGING. WITHOUT CONTENT OF DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS. MANUFACTURER: PHARM START LTD. UA. TRADE MARK: PHARM
TOB GALAFARM

UKRAINE
HS-Code
Products
2106909890
1. SPECIAL FOOD PRODUKTYBEZ CONTENT OF DAIRY FAT ISOGLUCOSE GLUCOSE COMPONENTS ANIMALORIGIN WITH ABOTSUKROZY STARCH CONTENT OF MORE THAN 5% NOT CONTAINING ALCOHOL A PRIMARY PACKAGING: TABLETS PILLS PECHAYEVSKI PECHIYI?20 WITH TASTE RASPBERRY
3004900000
1. MEDICINES IN FINISHED FORMS PACKAGED FOR RETAIL SALE FOR THE TREATMENT SPETSYFICHNYHNEDUH OR ILLNESS NOT CONTAINING NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL PACKAGING MOTION SICKNESS PILLS 50 MG AND IN NUDOTYTABLETKY ?1 0 - 1 00 UE. TABLETS OF STORAGE AND NUTOTABLETS OF 50MG ?25 - 20 UE. TRADEMARK: PHARMASCIENCE. MANUFACTURER: PHARMASCIENCE INC. MANUFACTURING COUNTRY: CA.
3004900000
1. READY-TO-USE PHARMACEUTICALS PACKAGED FOR THE TREATMENT OF SPECIFIC AILMENTS OR DISEASES FREE FROM NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL DISPENSERS: L-LYSINE ESCINATE SOLUTION FOR INJECTION 1MG / ML AMPOULES ?10 - 50 UNIT. TRADE MARK: ARTERIUM . MANUFACTURER: PJSC GALICHFARM # AMIGREN CAPSULES OF 100 MG ?3 (3X1) - 70 UNITS. UNITARY ENTERPRISE NORMASON TABLETS ON 7 5MG NO. 10 - 700 UNITARY ENTERPRISE. : LLC ASTRAFARM# GLUTARGIN PILLS OF 0.25G ?30 (10X3) - 40 UNIT. TRADEMARK: FC HEALTH. DENTAGEL GUM FOR GUMS ON 20 G TUBES - 150 UNIT. DERMASAN SKIN FLUID FOR 50 ML IN BOTTLES - 600 UNIT. TRADE MARK: PHYTOFARM . MANUFACTURER: PJSC FITOFARM # UNITARY COILS NIFUROXAZIDDABLETS COATED WITH FILM SHEATH 200MG ?20 (10X2) - 400
ARTERIUM CORPORATION

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR RETAIL TRADE FOR PEOPLE CONSISTING OF MIXED OR UNMIXED PRODUCTS NOT IN AEROSOL PACKAGING AND NOT RELATED TO DRUGS OR PREKURSORIVKSANTYNOLU NICOTINATE 50MG 1 PILL IN BLISTERS IN PACK ?1 0H6 SERIES 231 741 - 6 201 UP. IN 21 CARTON BOXES. LOPERAMIDE TABLETS 2MG IN BLISTERS IN PACKING NO.1 0X2 SERIES 2271 81 - 3331 UE. IN 1 4 CARTON BOXES. FLUXEN CAPSULES 20MG IN BLISTERS IN PACKING ?1 0X1 SERIES 241 396 - 1 2000 UE. IN 1 00 CARTON BOXES. MANUFACTURER: PJSC KYIVMEDPREPAT. MANUFACTURING COUNTRY - UA.
3004900000
1. MEDICINES FOR RETAIL TRADE FOR PEOPLE CONSISTING OF MIXED OR UNMIXED PRODUCTS NOT IN AEROSOL PACKAGING AND NOT RELATED TO DRUGS OR PREKURSORIVKSANTYNOLU NICOTINATE 50MG 1 PILL IN BLISTERS IN PACK ?1 0H6 SERIES 231 741 23 841 6 - 3072UP. IN 33 CARTON BOXES. 2MG LOPERAMIDE TABLETS IN BLISTERS IN PACKAGING ?1 0X2 SERIES 2271 81 - 21 60UP. IN 9 CARTON BOXES. HERPREVIR OIL FOR OUTDOOR APPLICATION 2.5% IN TUBES 1 5G IN PACKING ?1 SERIES 237494 - 201 6UP. IN 1 2 CARTON BOXES. HERPREVIR OIL FOR OUTDOOR APPLICATION 2.5% IN 5G TUBES IN PACKING # 1 SERIES 237496 - 4000UP. IN 25 CARTON BOXES. MANUFACTURER: PJSC KYIVMEDPREPAT. MANUFACTURING COUNTRY - UA.
3004900000
1. MEDICINAL PRODUCTS FOR RETAIL SALE TO PERSONS CONSISTING OF MIXED OR UNMIXED PRODUCTS NOT IN AEROSOL DISPENSERS AND NOT RELATED TO NARCOTIC SUBSTANCES OR PRECURSORS. PILLS OF 50 TABLETS IN CONTOURED CELLULAR PACKAGING SERIES 790919 801019 811019 811019 811019 81101 IN 294 CARTONS.L-LYSINE ESCINAT® DISTRICT FOR INTRAVENOUS ADMINISTRATION OF 1 MG / ML TO 5 ML IN AMPOULES NO. 10 SERIES 490919 - 15000 UNITS IN 500 CARTONS.DIMEXIDE SOLUTION FOR EXTERNAL USE 50 ML IN A VIAL SERIES 80919 - 29740 UNITS IN 744 CARTONS. YIN FOR RECEIVING IN THE MIDDLE IN A 25 ML VIAL WITH A PLASTIC LID SERIES 171019 - 26000 UNITS IN 520 CARTONS. MANUFACTURER: PJSC GALICHFARM. COUNTRY OF PRODUCTION - UA.
PVT ENTERPRISE TD OLKAR

UKRAINE
HS-Code
Products
3004900000
1.VETERINARY MEDICINAL PRODUCT: PRAZITABS ANTHELMINTIC AGENT IN THE FORM OF FLAT-PILL CYLINDRICAL TABLETS WITH A WHITE AND YELLOW COLOR WITH A SPECIFIC ODOR. COMPOSITION 1 TABLET (0.8 G) CONTAINS ACTIVE SUBSTANCES: PRAZIQUANTEL 50 MG# EXCIPIENTS: POWDERED SUGAR MAGNESIUM STEARATE POLYVINYLPYRROLIDONE CARBOXYMETHYL CELLULOSE SODIUM SALT FLAVORING CALCIUM CARBONATE. NEMATODES.PRIMARY PACKAGING NOT IN AEROSOL PACKAGING. PACKED FOR RETAIL SALE IN PACKAGES (1 PACK-10 BLISTERS 100 TABLETS) QUANTITY 1504 PACKS. COUNTRY OF MANUFACTURE UKRAINE.UA.TRADEMARK NO DATA.MANUFACTURER AGROZOOVET-SERVICE .
3004200000
1. VETERINARY MEDICINAL PRODUCT: METRIOX IN THE FORM OF TABLETS. COMPOSITION: 1 PILL CONTAINS ACTIVE INGREDIENT: CHLORTETRACYCLINE HYDROCHLORIDE (ANTIBIOTIC). THE USE IS USED FOR THE TREATMENT OF AFTER-ORDINAL INFECTIOUS INFECTIONS THE ORIGINAL PACKAGING IS NOT IN THE AEROSOL PACK. PACKAGED FOR RETAIL TRADE IN PACKAGING (1 PACKAGING 1 TABLE) NUMBER 6000 PACKAGING. PRODUCTION COUNTRY UKRAINE. UA. TRADEMARK NO DATA. MANUFACTURER'S PRIVATE ENTERPRISE OL KAR-AGROZOOVET-SERVICE.
3004900000
1.VETERINARY MEDICINAL PRODUCT: PRAZITABS ANTHELMINTIC AGENT IN THE FORM OF FLAT-PILL CYLINDRICAL TABLETS WITH A WHITE AND YELLOW COLOR WITH A SPECIFIC ODOR. COMPOSITION 1 TABLET (0.8 G) CONTAINS ACTIVE SUBSTANCES: PRAZIQUANTEL 50 MG# EXCIPIENTS: POWDERED SUGAR MAGNESIUM STEARATE POLYVINYLPYRROLIDONE CARBOXYMETHYL CELLULOSE SODIUM SALT FLAVORING CALCIUM CARBONATE. NEMATODES.PRIMARY PACKAGING NOT IN AEROSOL PACKAGING. PACKED FOR RETAIL SALE IN PACKAGES (1 PACK-10 BLISTERS 100 TABLETS) QUANTITY 1504 PACKS. COUNTRY OF MANUFACTURE UKRAINE.UA.TRADEMARK NO DATA.MANUFACTURER AGROZOOVET-SERVICE .
PVT JOINT STOCK COMPANY LEKHIM

UKRAINE
HS-Code
Products
3004900000
1.PREPARATIONS FOR THERAPEUTIC PROPHYLACTIC USE PACKAGED FOR RETAIL SALE NOT IN AEROSOL DISPENSERS PACKAGED IN INDIVIDUAL PAPER PACKS: -LEXACIN 500MG TABLETS NO. 10-1360UP. 80919MD SHELF LIFE. -TABLETS NITROGLYCERIN 0 5MG ?40 - 14040PACK. 311019MD SERIES SHELF LIFE TILL 01/04/2022 MANUFACTURER OF PJSC TECHNOLOGIST .- PAPAVERINE SOLUTION FOR OTHER 20 MG / ML 2 ML ? 10 - 4048PACKING SERIES 91534008 SHELF LIFE UP TO 01/11/2021 - RENALGAN SOLUTION FOR OTHER 500MG 2MG 0 02MG / 1ML 5ML ?5 - 2040 PACKING SERIES 91565010 SHELF LIFE UP TO 01. - PILLS RENALGAN 500MG 5MG 0.1MG ?10X2 - 5040PACK. SERIES 93525016 SHELF LIFE TILL 01.10.2021.MANUFACTURER OF PJSC LEKHIM-KHARKIV. TRADEMARK: NO DATA. COUNTRY OF MANUFACTURE-UA .
3004900000
1. MEDICINAL PRODUCTS FOR HUMANS: -AMITRIPTILIN TABLETS COVERED BY 25MG IN BLISTER PACKAGING ?50 (5X1 0) -1 0080UP. SERIES: 1 4081 9UZ. PLACE: 84 MANUFACTURED: 08. 201 9P 3 YEARS AVAILABLE. -CARBAMAZEPINE PILLS 200 MG EACH IN BLISTER PACK NO. 50
3004900000
1. MEDICINAL PRODUCTS FOR HUMANS: -AMITRIPTILIN PILLETS P / O 25MG 1 0 TABLES IN PACKAGING 5 PACKAGES IN A PACK OF PAPER -2040 SERIES: 1 81 01 9AM MANUFACTURED: 1 0. 201 9P. FITNESS THREE YEARS. -NITROGLYCERINE PILLOWS TABLETS 0.5 MG TO 40 TABLES IN A POLYMER CONTAINER WITH PACKAGING -6048UP. SERIES 31 1 01 9AM MANUFACTURED: 1 0. 1 9P. FITNESS THREE YEARS. -NIFEDIPINE TABLETS COVERED 1 0 MG ON 1 0 TABLES IN A BLISTER 5 BLISTERS IN A PACK OF PAPER -5040UP. SERIES 1 91 1 1 9AM MANUFACTURED: 1 1. 201 9P. FITNESS THREE YEARS. MANUFACTURER: PROT TECHNOLOGIST. COUNTRY MANUFACTURING UA.
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
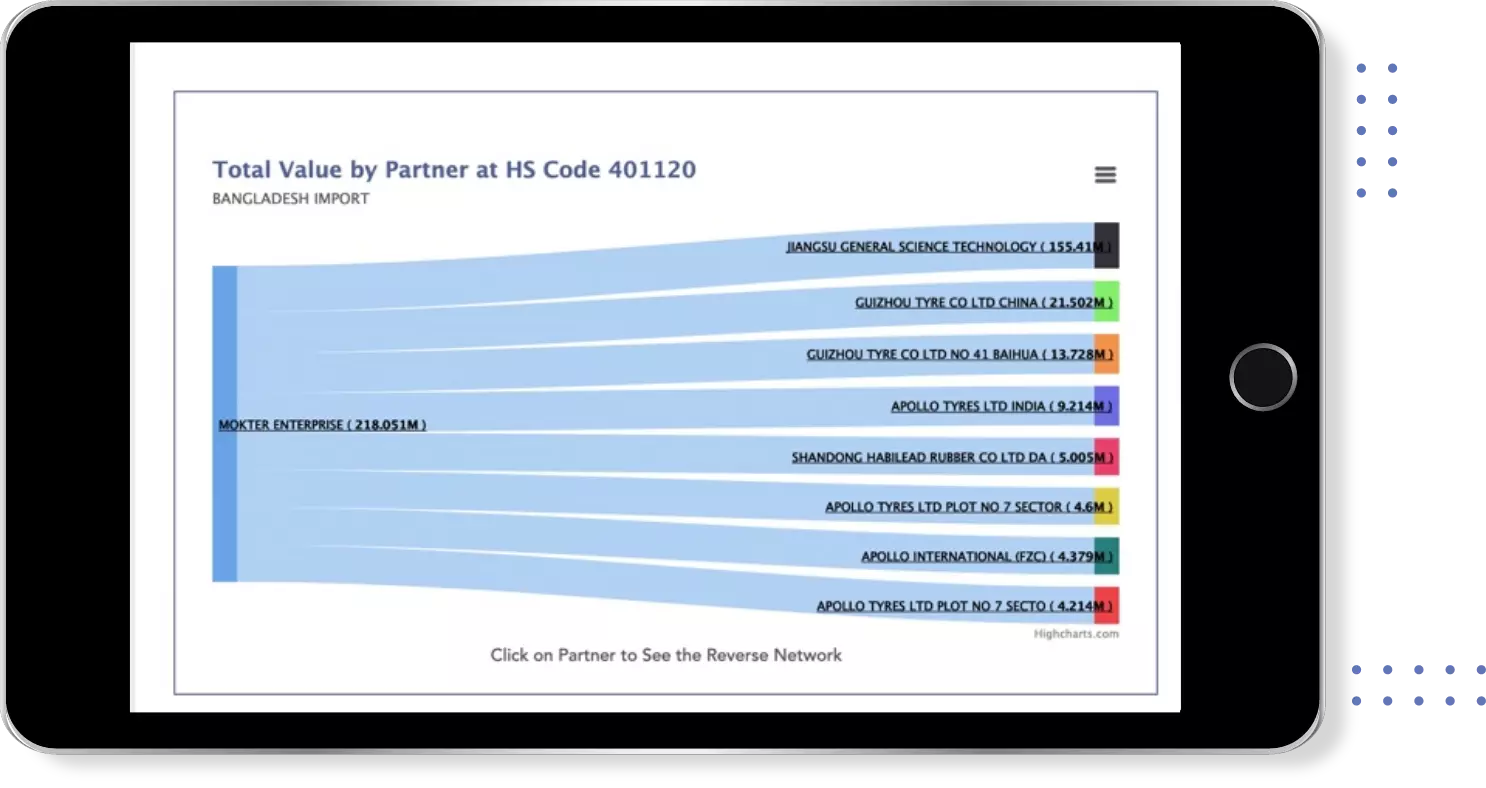
Get most profitable Buyers & Suppliers
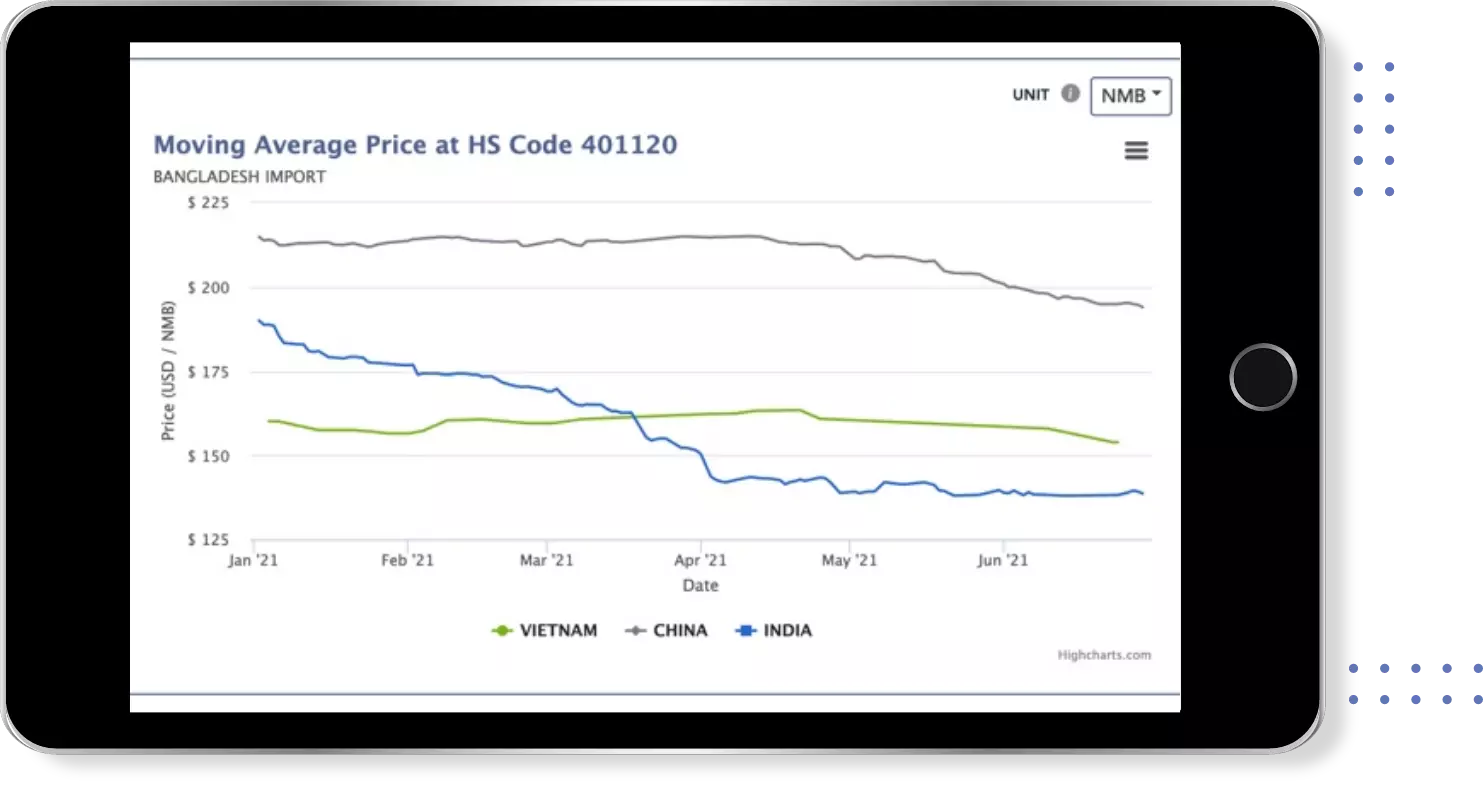
Instant insights on Price , Quantity & Value trends
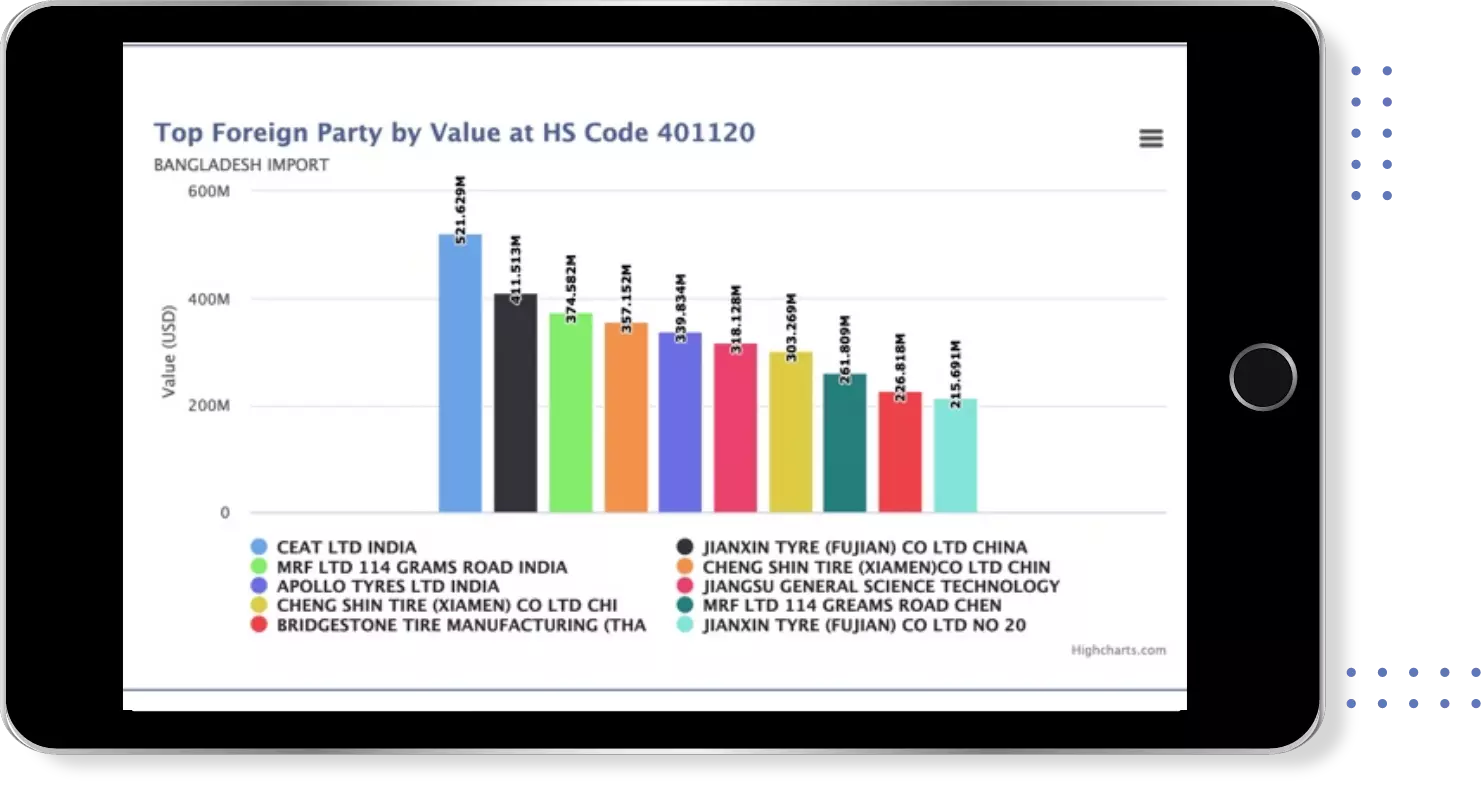
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
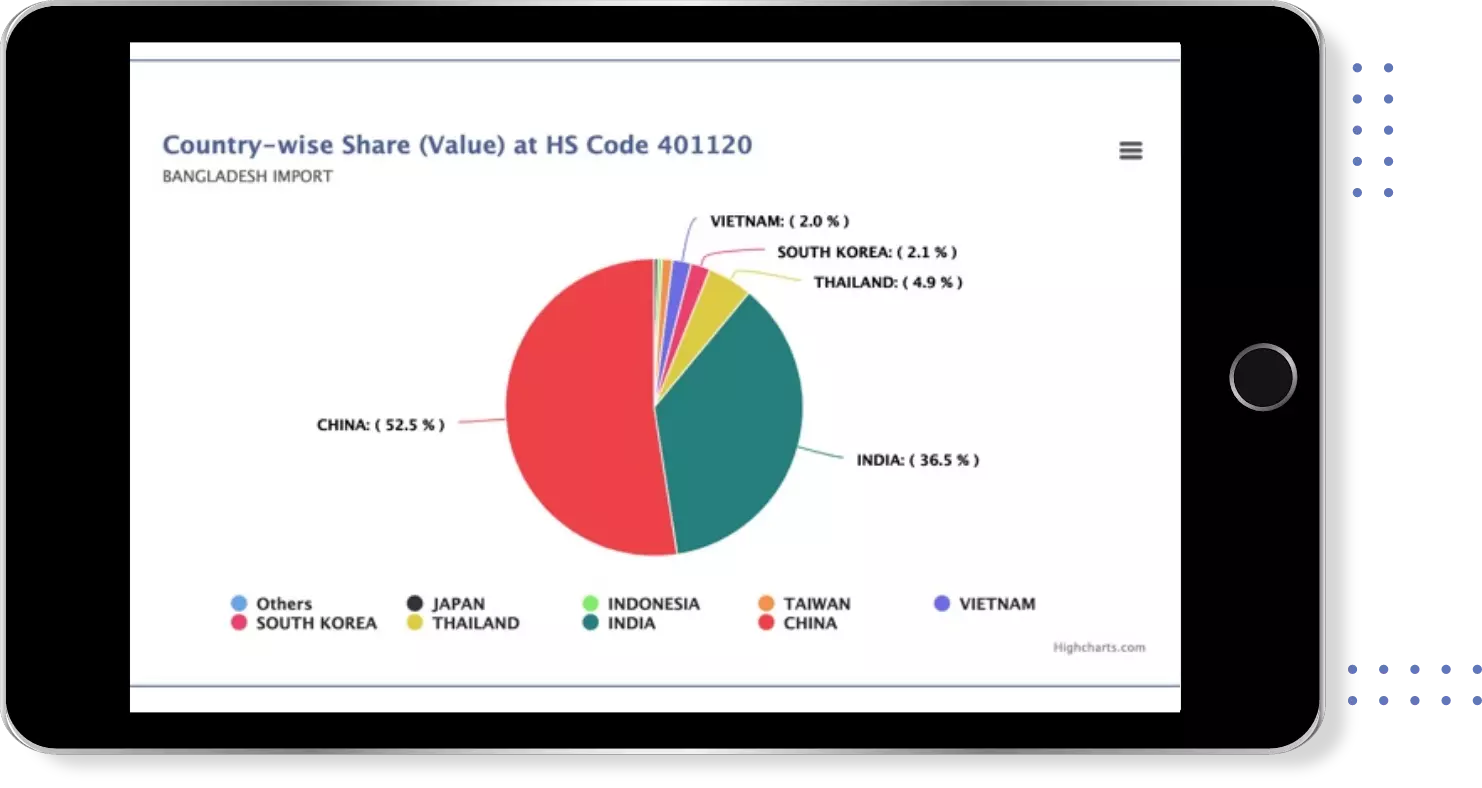
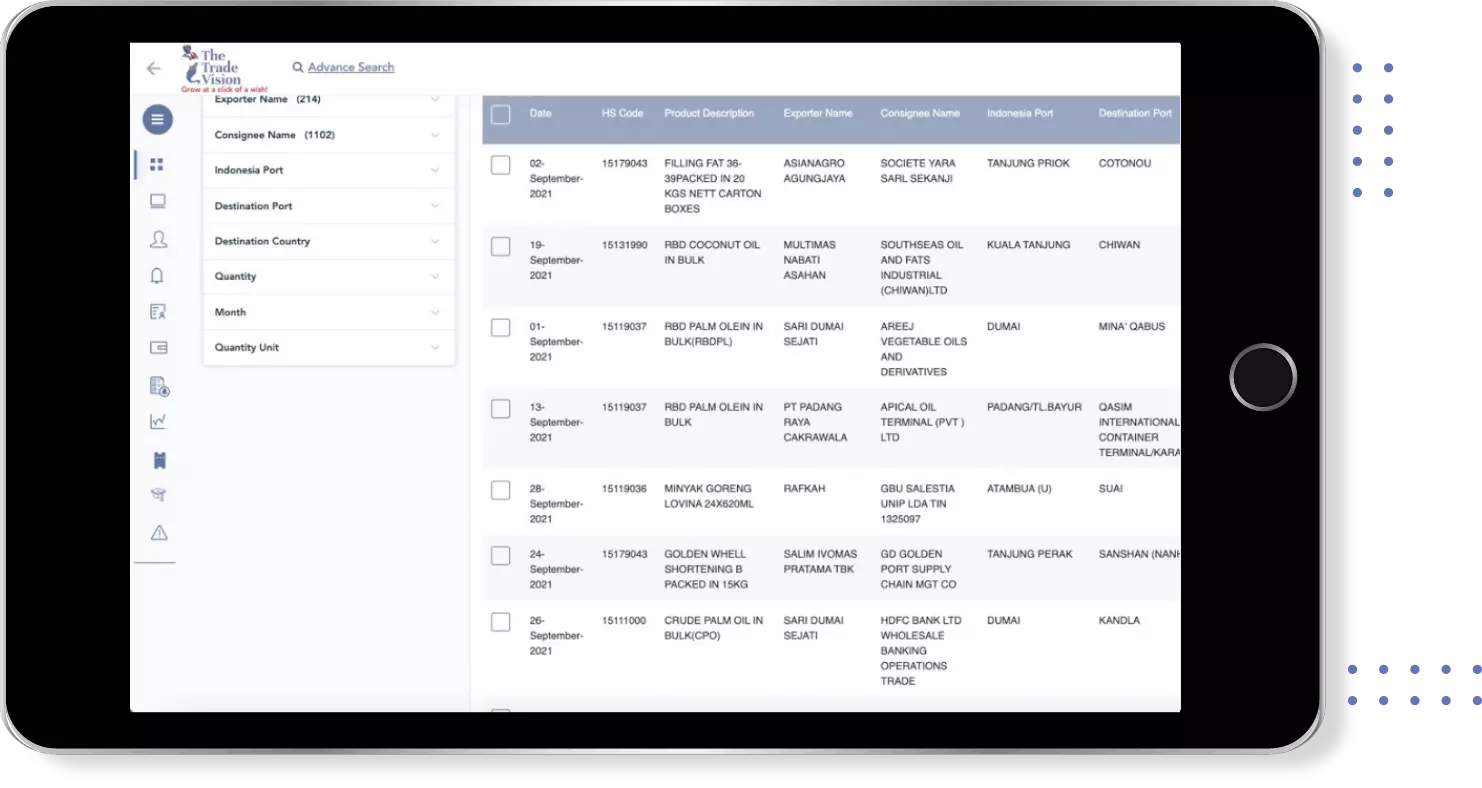
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.