Find verified buyers and sellers of each tablet in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- each tablet suppliers in ukraine
- COO Ukraine
Ukraine Suppliers of each tablet
PJSC NPC BORSHCHAGIV CFC

UKRAINE
HS-Code
Products
3004900000
1.PREPARATIONS FOR HUMANS (MEDICINES) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM PUT UP FOR RETAIL SALE CONTAINING NO NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL DISPENSER. NITROXOLINE TABLETS 50 X 50 X 10 X 5 -740 UNITS MANUFACTURER: PJSC NPC BORSCHAGOVSKY CFC SERIES EXPIRY DATE ARE INDICATED ON EACH BOX. COUNTRY OF PRODUCTION - UA.
3004900000
1. DRUGS FOR PEOPLE (DRUGS) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE A METERED DOSE FORM PACKAGED FOR RETAIL NOT CONTAINING NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL PACKAGING. PARACETAMOL FOR CHILDREN SYRUP 1 20MG / 5ML IN 1 00ML VIALS IN PACKING # 1 -1 320UP. SEPTEFRYL TABLETS 0.2MG IN BLISTERS IN PACKING ?1 0X5 -1 1 090UP. MANUFACTURER: PJSC NPC BORSHCHAGIV CFC. SERIES SHELF LIFE DESIGNED ON EACH VESSEL. MANUFACTURING COUNTRY - UA.
3004900000
1. DRUGS FOR PEOPLE (DRUGS) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE A METERED DOSE FORM CONTAINING ALKALOIDS OR DERIVATIVES THEREOF PUT UP FOR RETAIL SALE NOT CONTAINING NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL PACKAGING. ALOPURINOL TABLETS 1 00MG IN BLISTERS IN PACKING # 1 0X5 -1 008UP. PARACETAMOL FOR CHILDREN SYRUP 1 20MG / 5ML IN 1 00ML VIALS IN PACKING # 1 -1 440UP. SEPTEFRYL TABLETS 0.2MG IN BLISTERS IN PACKING ?1 0X5 -5520UP. COAL ACTIVATED TABLETS 250MG IN COURTESE COUNTRY BATCH NO.1 0 -84200UP. MANUFACTURER: PJSC NPC BORSCHAGOV CFC SERIES EXPIRY DATE ON EACH PACKAGING. MANUFACTURING COUNTRY - UA.
IMPEROV FOODS LTD

UKRAINE
HS-Code
Products
0407210000
1. FRESH EGGS IN SCALOUP TABLE CHICKEN TABLETS CATEGORIES S (C2) AVERAGE WEIGHT 50-55GR BROWN IN BOXES OF 360 PCS IN EACH 449 ALL 449 EACH PCS COMPANY MANUFACTURER: BRANCH OF BARYSHOVSKY STV INTERBIZNES. TRADEMARK: NO DATA. MANUFACTURING COUNTRY: UKRAINE UA.
0407210000
1. FRESH EGGS TABLETS TABLETS PIGS PACKAGING BOXES OF 360PCS EACH: CATEGORIES M (C1) BROWN AVERAGE WEIGHT 55-60GR. SIZE 3- 832 BOXES; CATEGORIES M (C1) WHITE AVERAGE WEIGHT 55-60GR. SIZE 3 - 41 6 BOXES TOTAL - 449 280 THOUSANDS. PCS COMPANY MANUFACTURER: BRANCH OF BARYSHOVSKY STV INTERBIZNES. TRADEMARK: NO DATA. MANUFACTURING COUNTRY: UKRAINE UA.
0407210000
1. FRESH EGGS TABLETS TABLETS PURPLE CHICKEN BROWN PACKAGING BOXES OF 360 EACH CATEGORY L (C0) AVERAGE WEIGHT 65-70GR SIZE 65-70GR SIZE CATEGORIES M (C1) AVERAGE WEIGHT 60-65GR SIZE 2-1 50 BOXES; CATEGORIES M (C1) AVERAGE WEIGHT 55-60GR SIZE 3-600 BOXES; CATEGORIES S (C2) AVERAGE WEIGHT 50-55GR SIZE 4-1 50 BOXES. TOTAL: 388 800 THOUSANDS. PCS COMPANY MANUFACTURER: BRANCH OF BARYSHOVSKY STV INTERBIZNES. TRADEMARK: NO DATA. MANUFACTURING COUNTRY: UKRAINE UA.
PJSC NVTS BORSCHAGIVSK HFZ

UKRAINE
HS-Code
Products
3004400000
1.PREPARATIONS FOR HUMANS (MEDICINES) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM CONTAINING ALKALOIDS OR DERIVATIVES THEREOF PUT UP FOR RETAIL SALE NOT IN AEROSOL DISPENSERS NOT CONTAINING PRODUCTS OF ANIMAL ORIGIN DO NOT CONTAIN NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS .: KRATAL TAB. 10 TABLETS IN A CONTOURED CELL PACK 2 CONTOUR PACKS IN A CARDBOARD PACK - 322UP. STARTED ON EACH BOX. COUNTRY OF PRODUCTION - UA.
3004200000
1.PHARMACEUTICALS (MEDICINES) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN A DOSAGE FORM CONTAINING OTHER ANTIBIOTICS PACKAGED FOR RETAIL SALE NOT IN AEROSOL DISPENSERS NOT CONTAINING PRODUCTS OF ANIMAL ORIGIN DO NOT CONTAIN NARCOTIC PSYCHOTROPIC SUBSTANCE OF PRECURSORS NOT FOR VETERINARY MEDICINE: NYSTATIN TABLET IN / ABOUT 500000OD ?20-2150UP.GRISOFULVIN TABLE 125MG ?20?2- 129UP.DOXYCYCLINE CAPS. 250MG ?10X2- 860UP TETRACYCLINE G / HL TABLE IN / O. 100MG ?20- 8600UP.TSEFAZOLIN-BCHFP.D FOR OTHER 1000MG ?1 VIAL - 10500UP.MANUFACTURER: PJSC NPC BORSCHAGOVSKY HFZ SERIES EXPIRY DATE ARE INDICATED ON EACH CARTON. COUNTRY OF MANUFACTURE - UA.PACKAGED IN 343 CARTONS BOXES ON 5 PALLETS.
3004900000
1.PHARMACEUTICALS CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM PUT UP FOR RETAIL SALE NOT IN AEROSOL DISPENSERS CONTAINING NO ANIMAL PRODUCTS DRUG-FREE PSYCHOTROPIC SUBSTANCES AND PRECURSORS .: ALLOPURINOL TABLETS IN CONTOUR CELLULOSE PACK 100MG?10X5- 420UP. 80MG ?10X5- 2142UP CLOTRIMAZOLE TABL OTHER 100MG ?10- 440UP.IBUPROFEN TABLET V / O 200MG ?10X5- 720UP.ETAMBUTOL TABLE 400MG ?10X5- 108UP.KETOTIFEN SYRUP 1MG / 5ML 100ML VIAL-960UP.CLOTRIMAZOL OINTMENT 1% 25MG TUBULAR R / N. 1% 25ML BOTTLE - 1800UP.INDOVENOL GEL 40G TUBA - 144UP.AMBROXOL G / HL. SERIES EXPIRY DATE ARE INDICATED ON EACH BOX. COUNTRY IN
PJSC NPC BORSCHAGOVSKY CFC

UKRAINE
HS-Code
Products
3004400000
1. DRUGS FOR PEOPLE (DRUGS) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE A METERED DOSE FORM CONTAINING ALKALOIDS OR DERIVATIVES THEREOF PUT UP FOR RETAIL SALE NOT CONTAINING NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL PACKAGING. CAFFEINE-BENZOATE SODIUM TABLETS 200MG ?1 0 -2400UP. MANUFACTURER: PJSC NPC BORSCHAGOV CFC SERIES EXPIRY DATE ON EACH PACKAGING. MANUFACTURING COUNTRY - UA.
3004900000
1. DRUGS FOR PEOPLE (DRUGS) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE A METERED DOSE FORM PACKAGED FOR RETAIL NOT CONTAINING NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL PACKAGING. ALOPURINOL TABLETS IN PACKAGING PACKAGING 1 00MG ?1 0X5 -840UP. BALSAMIC LINEN (OVER VYSHNEVE) 40G TUBE -2700UP. BRILLIANT GREEN SOLUTION 1% 20ML VIAL -1 0008UP. VERAPAMIL HYDROCHLORIDE TABLE. I / O 80MG ?1 0X5 -2520UP. ISONIAZIDE TABLE. 200MG ?1 0X5 -300UP. CLOTRIMAZOLE TABLE. VAGINAL 1 00MG ?1 0 -440UP. IBUPROFEN TABLE. I / O 200MG ?1 0X5 -324UP. CLOTRIMAZOLE OINTMENT 1% 25G TUBE -3240UP. DIGOXIN TABLE. 0 25MG ?20X2 -6300UP. NITROSORBIDE TABLE. 1 0MG ?1 0X4 -540UP. PARACETAMOL FOR CHILDREN SYRUP 1 20MG / 5ML 1 00M VIAL -240UP. TABLE TABLE. 1 50MG ?1 0 -4800UP. MANUFACTURER: PJSC NPC BORSCHAGOV CFC SERIES EXPIRY DATE ON EACH PACKAGING. MANUFACTURING COUNTRY - UA.
3004900000
1. DRUGS FOR PEOPLE (DRUGS) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE A METERED DOSE FORM PACKAGED FOR RETAIL NOT CONTAINING NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS NOT IN AEROSOL PACKAGING. AMBROTUSIN 3MG / ML SYRUP 1 00ML IN A VIAL 1 VIAL IN A BAG -1 5240PCS. AMBROTUSIN 30MG TABLETS 1 0 TABLETS IN A BLISTER 2 BLISTERS IN A PACK -25000PCS. MANUFACTURER: PJSC NPC BORSHCHAGIV CFC. SERIES SHELF LIFE DESIGNED ON EACH VESSEL. MANUFACTURING COUNTRY - UA.
PRAT PHARMACEUTICAL COMPANY DARNITSA

UKRAINE
HS-Code
Products
3004900000
1.PHARMACEUTICALS (MEDICINES) FOR HUMANS NOT CONTAINING DRUGS WITHOUT AEROSOL PACKING PUT UP FOR RETAIL SALE: STREPTOCIDE-DARNITSA TABLE. 300MG ?10-16000UP. DIBAZOL-DARNITSA TABLE. -DARNITSA TABLET V / O 10MG ?50-1000UP. NOVOCAINE-DARNITSA DISTRICT D / OTHER AMP 5MG / ML 5 ML ?5?2-5500UP. DIMEDROL-DARNITSA DISTRICT D / OTHER AMP 1 ML 10MG / ML ?5X2-1000UP. PIRACETAM-DARNITSA TABLE 400MG ?10X3-600UP. PROSERIN-DARNITSA DISTRICT D / OTHER AMP. SUPPLIED COMPLETE WITH INSTRUCTIONS FOR USE. MANUFACTURER - PHARMACEUTICAL COMPANY DARNITSA. RKA- DARNYTSIA PHARMACEUTICAL COMPANY. COUNTRY OF MANUFACTURE - UKRAINE (UA). THE MANUFACTURER AND COUNTRY OF ORIGIN MARKING ARE INDICATED ON EACH BOX.
3004900000
1. MEDICINAL PRODUCTS (MEDICINES) FOR HUMAN DRUG FREE WITHOUT AEROSOL PACKAGED FOR RETAIL MARKET: -NOW-MONEY 5MG / MLAMP. 2ML ?5X2 -5000UP. -DYTYLINE-DARNITSA R-H D / IN 20MG / ML 5MLAMP. ?1 0 -200UP. -DIMEDROL-DARNITSA TABLET 50MG PACKAGING ?1 0 -1 000UP. -DIBASOL-DARNITSA SOLUTIONS FOR INITIONS 1% 1 0ML ?1 0-1 0000UP. -SULFOCAMPHOCAIN-DARNITSA RN N D / IN 1 00MG / ML 2 0ML ?1 0 -3000UP. -RIBOXIN-DARNITSA RN N D / IN 20MG / ML IN AMPULES OF 1 0ML NO. 5X2 -1 0000UP. COMPLETE WITH INSTRUCTIONS FOR USE. MANUFACTURER-PHARMACEUTICAL COMPANY DARNITSA. TRADEMARK - PHARMACEUTICAL COMPANY DARNITSA. MANUFACTURING COUNTRY- UKRAINE (UA). MARKING THE MANUFACTURER AND THE COUNTRY OF ORIGINATED ON EACH SHIP.
3004400000
1. DRUGS (MEDICINE) FOR A PERSON OF MIXED SCHOSKLADAYUTSYA NEZMISHANYHPRODUKTIV OR FOR THERAPEUTIC USE ABOPROFILAKTYCHNOHO NOT MISTYATNARKOTYKIV WITHOUT AYEROZOLNOHO PACKAGING CONTAINING ALKOLOYIDY PACKAGED DLYAROZDRIBNOYI TRADE: -PENTOKSYFILIN-DARNITSA TABLETS OF 200 MG ?1 0H2 -300UP. -PLATIFILIN-DARNITSA DISTRICT. 2MG / ML VAMP. 1 ML ?5X2-1 00UP. COMPLETE INSTRUCTIONS FOR USE. MANUFACTURER-PHARMACEUTICAL COMPANY DARNITSA. TRADEMARK - PHARMACEUTICAL COMPANY DARNITSA. MANUFACTURING COUNTRY- UKRAINE (UA). MARKING THE MANUFACTURER AND THE COUNTRY OF ORIGINATED ON EACH SHIP.
PHARM START LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE. - BIFREN CAPSULES NO. 20 TABLETS 250MG EACH. BY 1 0 TABLETS IN THE BLISTER BY 2 BLISTERS IN THE PACK OF PAPER -1 2363UP. SERIES 1 1 021 9. MANUFACTURING DATE 06. 02. 201 9. EXPIRY DATE 03. 2021 R. COMPOSITION PER ACTIVE SUBST
3004900000
1. MEDICINES FOR PEOPLE. - BIFREN CAPSULES NO. 20 TABLETS 250MG EACH. BY 1 0 TABLETS IN THE BLISTER BY 2 BLISTERS IN THE PACK OF PAPER -24566UP. SERIES 91 091 9. MANUFACTURING DATE 22. 09. 201 9. EXPIRY DATE 1 0. 2021 R. COMPOSITION PER ACTIVE SUBSTANCE TABLE: FENIBUT - 250MG. - RISOPTAN TABLES 1 0MG. ?3 3 TABLETS IN THE BLISTER 1 BLISTER IN THE PACK OF PAPERBOARD-51 1 8UP. SERIES 02091 9. PRODUCTION DATE 02. 09. 201 9. SHELF LIFE 1 0. 2022P. ACTIVE SUBSTANCES: 1 TABLE CONTAINING RISATRIPTANE BENZOATE 1 4 53MG. IN TRANSFER TO RIZATRIPTAN 1 0MG. - RISOPTAN TABLES 1 0MG. ? 6 3 TABLETS IN THE BLISTER 2 TABLETS IN THE PACK OF PAPER -1 877UP. SERIES 23091 9. PRODUCTION DATE 02. 09. 201 9. EXPIRY DATE 1 0. 2022P. ACTIVE SUBSTANCES: 1 TABLE CONTAINING RISATRIPTANE BENZOATE 1 4 53MG. IN TRANSFER TO RIZATRIPTAN 1 0MG. NOT IN THE AEROSOL PACK. WITHOUT CONTENT OF DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS. MANUFACTURER: PHARM START LTD. UA. TRADE MARK: PHARM START. PRODUCTION COUNTRY UA.
3004900000
1. MEDICINES FOR HUMANS. - RIZOPTAN TABLETS 10MG.?3 3 TABLETS IN A BLISTER 1 BLISTER IN A PACK OF CARDBOARD-1800UP. SERIES 240919. DATE OF MANUFACTURE 02.09.2019. SHELF LIFE UP TO 10.2021R. ACTIVE SUBSTANCES: 1 TABLET CONTAINS RISOTRIPTAN BENZOATE 14 53MG. CALCULATED ON RISATRIPTAN 10MG.- RIZOPTAN TABLETS 10MG.?6 3 TABLETS IN A BLISTER 2 BLISTERS IN A PACK OF CARDBOARD -1800UP. SERIES 250919. ACTIVE INGREDIENTS: 1 TABLET CONTAINS 14.53MG RISOTRIPTAN BENZOATE CALCULATED ON 10MG RISATRIPTAN. 3 BLISTERS IN A CARTON -600UP: SERIES 910819 PRODUCTION DATE 30.08.2018R. SHELF LIFE UP TO 09.2021R.1 CAPSULE CONTAINS: ACTIVE SUBSTANCES: PREGABALIN 75MG.-NEOGABIN CAPSULES 150MG.30 EACH 10 TABLETS IN A BLISTER# 3 BLISTERS IN A CARTON OF -600UP: SERIES 040919 PRODUCTION DATE 02.09.2018. SHELF LIFE UP TO 10.2021R.1 CAPSULE CONTAINS: ACTIVE SUBSTANCES: PREGABALIN 150MG. TABLETS IN A BLISTER 3 BLISTERS IN A PACK OF CARDBOARD -240UP. SERIES 030919. PRODUCTION DATE 02.09.2019. SHELF LIFE UP TO 10.2021G. ACTIVE INGREDIENTS: 1 TABLET CONTAINS ARIPIPRAZOLE 15MG (IN TERMS OF 100% ARIPIPRAZOLE DRY SUBSTANCE) NOT IN AEROSOL DISPENSER. NO NARCOTIC PSYCHOTROPIC OR PRECURSOR CONTENT. TRADEMARK: FARMA START. COUNTRY OF PRODUCTION UA.
CFC BIOKON LLC

UKRAINE
HS-Code
Products
3004900000
1. MEDICINAL PRODUCTS FOR PEOPLE: -VOLTAREN (R) RAPID TABLETS CRITICAL SUGAR 50MG ON 1 0 TABLETS IN A BLISTER; 2 CARDBOARD CARTON BLISTERS EACH 3 BLISTERS VKARTONNIY BOX (ACTIVE INGREDIENT: 1 TABLET CONTAINS: 2 288MH TIZANIDINE HYDROCHLORIDE 2 MG TYZANIDYNUVIDPOVIDNO MEETS) SERIES: KL282-200UP-SIRDALUD (R) 4 MG TABLETS OF IN 0 1 TABLETS IN A BLISTER; 3 BLISTERS VKARTONNIY BOX (ACTIVE INGREDIENT: 1 TABLET CONTAINS: 4 576MH TYZANIDYNUHIDROHLORYDU THAT MEETS TIZANIDINE 4 MG RESPECTIVELY) SERIES: KJ667-1 00UPVYROBNYK: NOVARTIS SAHLIK GUIDE TARIM URUNLERI VE SAN. WE ARE TICK. A. -VOLTAREN 50MG ENTERIC-COATED TABLETS ?20 / VOLTAREN 50 MG TABLETS VKYSHKOVOROZCHYNNYY MEMBRANE ?20 (IN CARTON) (ACTIVE INGREDIENT: DICLOFENAC SODIUM 50MG / TABLET) SERIES: KN904 -270UP-VOLTAREN (R) GASTRORESISTANT TABLETS ON 25MG P
3004900000
1. MEDICINAL PRODUCTS FOR PEOPLE: -VOLTAREN (R) RAPID TABLETS CRITICAL SUGAR 50MG ON 1 0 TABLETS IN A BLISTER; 2 CARDBOARD CARTON BLISTERS EACH 3 BLISTERS VKARTONNIY BOX (ACTIVE INGREDIENT: 1 TABLET CONTAINS: 2 288MH TIZANIDINE HYDROCHLORIDE 2 MG TYZANIDYNUVIDPOVIDNO MEETS) SERIES: KM262-330UP-SIRDALUD (R) 4 MG TABLETS OF IN 0 1 TABLETS IN A BLISTER; 3 BLISTERS VKARTONNIY BOX (ACTIVE INGREDIENT: 1 TABLET CONTAINS: 4 576MH TYZANIDYNUHIDROHLORYDU THAT MEETS TIZANIDINE 4 MG RESPECTIVELY) SERIES: KJ667-500UPVYROBNYK: NOVARTIS SAHLIK GUIDE TARIM URUNLERI VE SAN. WE ARE TICK. A.S.
3004900000
1. DOCTORS FOR LICENSES 60MG) SERIES: BKK76 -588UP- SERIES: BLX01 -442UP-EXFORGE 5MG / 1 60MG FILM-COATED TABLETS?1 4 / EXFORGE TABLETS COVERED SHEET 60MG / TABLE 1MG AMLODYPIN 5MG VALSARTAN 1 60MG) SERIES: BLV65 -1 50UP-DIOVAN (R) TABLETS 1 60MG FOR 1 4 UPLISTER TABLES; 1 BY EACH BLISTER IN A BOARD WITH CARDBOARD ;
PVT JOINT STOCK COMPANY LEKHIM

UKRAINE
HS-Code
Products
3004900000
1. MEDICINAL PRODUCTS FOR HUMANS: -AMITRIPTILIN TABLETS COVERED BY 25MG IN BLISTER PACKAGING ?50 (5X1 0) -1 0080UP. SERIES: 1 4081 9UZ. PLACE: 84 MANUFACTURED: 08. 201 9P 3 YEARS AVAILABLE. -CARBAMAZEPINE PILLS 200 MG EACH IN BLISTER PACK NO. 50
3004900000
1.PREPARATIONS FOR HUMANS: -BISACODYL SUPPOSITORIES OF 0.01G TO 5 SUPPOSITORIES IN CONTOUR CELL PACKS OF 2 CONTOUR CELLULAR PACKS IN A PACK OF CARDBOARD -640UP. SERIES: 92006024 PRODUCED: 08.2019 SUITABLE: 3.2019YRS. -LORATADINE TAB. 0.01G NO. 10 IN BLISTERS -1200UP. SERIES 93022013 PRODUCED ON 07.2019 3 YEARS. . SUITABLE FOR 3 YEARS.-NO-X-HA TABLETS 0.04G IN CONTOUR CELL PACKS ?30 -3000UP. SERIES: 93013009 MADE 09.2019 SUITABLE: 5 YEARS.-BUT-X-SHA SOLUTION FOR INJECTION 20MG / ML FOR 2ML EACH AMPOULES ?5 -3040UP. SERIES: 91001013 MADE: 07.2019 SUITABLE: 3 YEARS -DICLOFENAC SODIUM SOLUTION FOR INJECTION 25MG / ML 3 ML IN A 5 AMPOULE IN A BLISTER 1 BLISTER IN A PACK OF CARDBOARD -2040UP. SERIES: 91034015 PRODUCED: 07.2019 SUITABLE: 2 YEARS. MANUFACTURER: PJSC LEKHIM-KHARKIV# PRODUCTION COUNTRY UA.
3004900000
1. MEDICINES FOR HUMAN USE PACKAGED FOR RETAIL SALE NOT IN AEROSOL DISPENSERS NOT CONTAINING NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS: - WATER FOR INJECTION SOLVENT FOR PARENTERAL USE AMPOULES OF 2 ML NO. 10 - 2024 PACKS ML PER AMPOULE 5 AMPOULES PER BLISTER 2 BLISTERS PER PACK) ATC CODE V07A B ACTIVE INGREDIENT: WATER FOR INJECTION# (SER. 91527015 - 2024 PACKS PRODUCTION DATE: NOVEMBER 2019 EXPIRY DATE) : UNTIL NOVEMBER 2023) - SUPROSTYLIN TABLETS 0 025G ?20 (2X10) (CONTOUR CELL PACKS) - 5200 PACKS (10 TABLETS EACH) K IN CONTOUR CELL PACK 2 CONTOUR CELL PACKS IN PACK) ATC CODE R06AC03 ACTIVE INGREDIENT: CHLOROPYRAMINE HYDROCHLORIDE 0.025 G# ) - TRIESEPTOL TABLETS 100MG / 20MG ?20 (2X20) (BLISTERS) - 2080 PACKS (10 TABLETS IN A BLISTER 2 BLISTERS IN A PACK) ATC CODE J01EE01 ACTIVE INGREDIENT: SULFAMETHOXAZOLE - 100 MG TRIMETHOPRIM - 20 MG #. (SER. 93516001 - 2080 PACKS PRODUCTION DATE: NOVEMBER 2019 SHELF LIFE: UNTIL NOVEMBER 2024) - TRIESEPTOL TABLETS 400MG / 80MG ?20 (2X20) (BLISTERS) - 3024 PACKS (10 TABLETS EACH IN BLISTER PACKS 2 BLISTERS PER PACK) PBX CODE J01EE01 ACTIVE INGREDIENT: SULFAMETHOXAZOLE - 400 MG TRIMETHOPRIM - 80 MG #. (SER. 93517006 - 3024 PACKAGES PRODUCTION DATE: NOVEMBER 2019 EXPIRY DATE: NOVEMBER 2024) TRADEMARK: NO DATA. MANUFACTURER: PJSC LEKHIM-KHARKIV - IBUPROFEN TABLETS COATED 200MG ?50 (5X10) (BLISTERS) - 10080 PACKS (10 TABLETS IN A BLISTER# 5 BLISTERS IN A PACK OF CARDBOARD) PBX CODE M01AE01 ACTIVE INGREDIENT: IBUPROFEN 200 MG #. (SER. 311119UZ - 10080 PACKAGES DATE OF PRODUCTION: 19.11.2019 SHELF LIFE: UNTIL NOVEMBER 2022) - NITROXOLINE TABLETS COATED 50MG ?50 (5X10) (BLISTERS) - 5040 PACKAGES (TABLETS COATED) 50 MG OF 10 TABLETS IN A BLISTER# 5 BLISTERS IN A PACK OF CARDBOARD) PBX CODE J01XX07 ACTIVE INGREDIENT: NITROXOLINE 50 MG #. (SER. 231119UZ - 5040 PACKAGES DATE OF PRODUCTION: 11.11.2019 SHELF LIFE: UNTIL NOVEMBER 2023) - RIBOXIN TABLETS COATED 200MG ?50 (5X10) (BLISTERS) - 4631 PACKAGES (TABLETS COATED) 200 MG OF 10 TABLETS IN A BLISTER# 5 BLISTERS IN A PACK OF CARDBOARD) ATC CODE C01EB ACTIVE INGREDIENT: RIBOXIN (INOSINE) - 200 MG #. (SER. 111119UZ - 4631 PACKAGES DATE OF PRODUCTION: 08.11.2019 SHELF LIFE: UNTIL NOVEMBER 2022) - FLUCONAZOL TABLETS COATED 150MG ?1 (1?1) (BLISTERS) - 5040 PACKAGES (TABLETS COATED) 150 MG 1 TABLET IN A BLISTER# 1 BLISTER IN A PACK OF CARDBOARD) PBX CODE J02AC01 ACTIVE INGREDIENT: FLUCONAZOLE 150 MG #. (SER. 30919UZP2 / 1 - 5040 PACKS DATE OF MANUFACTURE: 09/09/2019 EXPIRY DATE: SEPTEMBER 2022) - FLUCONAZOL TABLETS COATED WITH 150MG NO. 2 (1X2) (BLISTERS) - 5040 PACKS (TABLETS COATED 150 MG OF 2 TABLETS IN A BLISTER# 1 BLISTER IN A PACK OF CARDBOARD) PBX CODE J02AC01 ACTIVE INGREDIENT: FLUCONAZOLE 150 MG #. (SER. 30919UZ / 2 - 5040 PACKS DATE OF MANUFACTURE: 09/09/2019 EXPIRY DATE: SEPTEMBER 2022) - CIPROFLOXACIN TABLETS COATED WITH 250MG ?10 (1X10) (BLISTERS) - 2100 PACKS (TABLETS COATED 250 MG OF 10 TABLETS IN A BLISTER# 1 BLISTER IN A PACK OF CARDBOARD) PBX CODE J01MA02 ACTIVE INGREDIENT: CIPROFLOXACIN HYDROCHLORIDE IN THE FORM OF CIPROFLOXACIN 250 MG #. (SER. 31119UZ - 2100 PACKAGES DATE OF PRODUCTION: 20.11.2019 SHELF LIFE: UNTIL NOVEMBER 2022) - CIPROFLOXACIN TABLETS COATED 500MG ?10 (1X10) (BLISTERS) - 5040 PACKAGES (TABLETS COATED) 500 MG OF 10 TABLETS IN A BLISTER# 1 BLISTER IN A PACK OF CARDBOARD) ATC CODE J01MA02 ACTIVE INGREDIENT: CIPROFLOXACIN HYDROCHLORIDE IN THE CONVERSION TO CIPROFLOXACIN 500 MG #. (SER. 291119UZ - 5040 PACKAGES DATE OF PRODUCTION: 11/13/2019 EXPIRY DATE: NOVEMBER 2022) TRADEMARK: NO DATA . MANUFACTURER: PJSC TECHNOLOGIST COUNTRY OF PRODUCTION: UA.
CFC LLC BIOKON

UKRAINE
HS-Code
Products
3004900000
1. DRUGS FOR PEOPLE -KATAFAST POWDER FOR ORAL SOLUTION OF 50 MG TO 3 SACHETS IN A BOX MADE OF CARDBOARD (ACTIVE INGREDIENT: 1 SACHET CONTAINS: 50 MG DICLOFENAC POTASSIUM) SERIES: AMPA0CJ -1 5UPVYROBNYK 51: PRODUCTION CYCLE: MIFARM S. P A. -TRILEPTAL TABLETS COVERED WITH FILM SHEET 600MG ON 1 0 UBLISTER TABLETS; PACKAGING EACH POINTS (ACTIVE SUBSTANCES: 1 TABLETS)
FOP SEMENOVYCH NATALIA VASYLIVNA PR T KR 202753 FROM 04 06 1997

UKRAINE
HS-Code
Products
3406000000
1.PARAFFIN CANDLES: - TABLET CANDLE - 10 SETS 600 PIECES EACH (6000PCS).
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
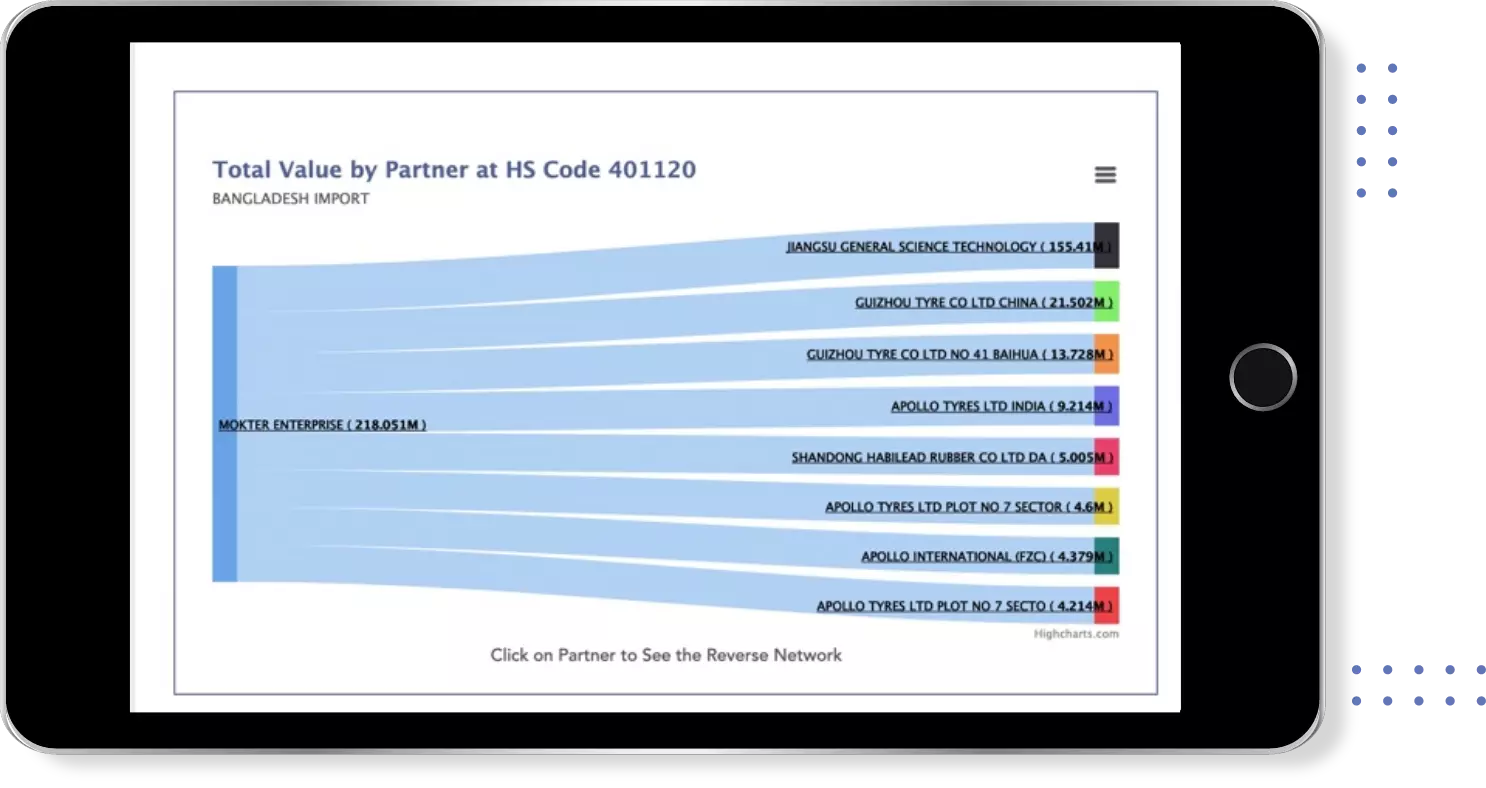
Get most profitable Buyers & Suppliers
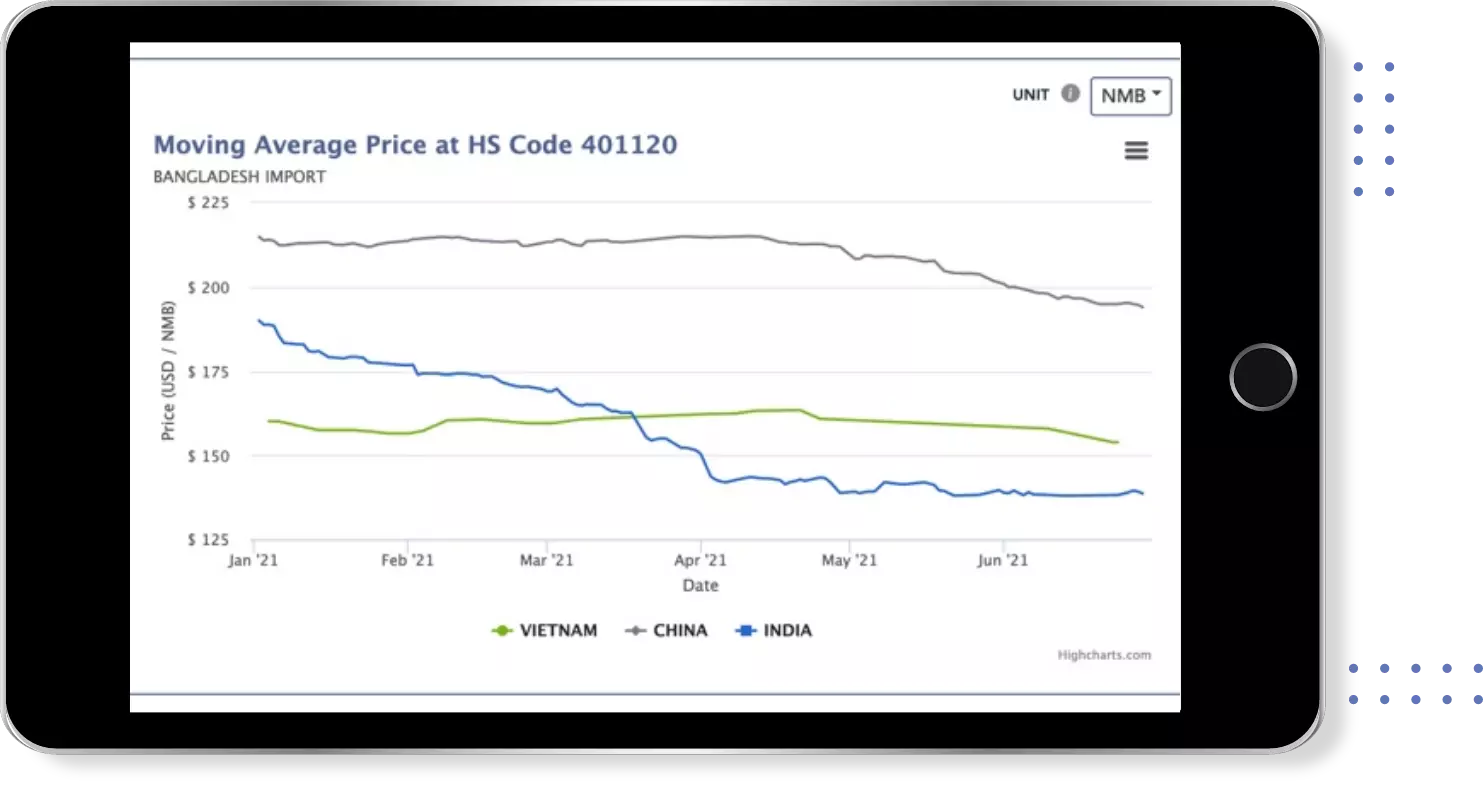
Instant insights on Price , Quantity & Value trends
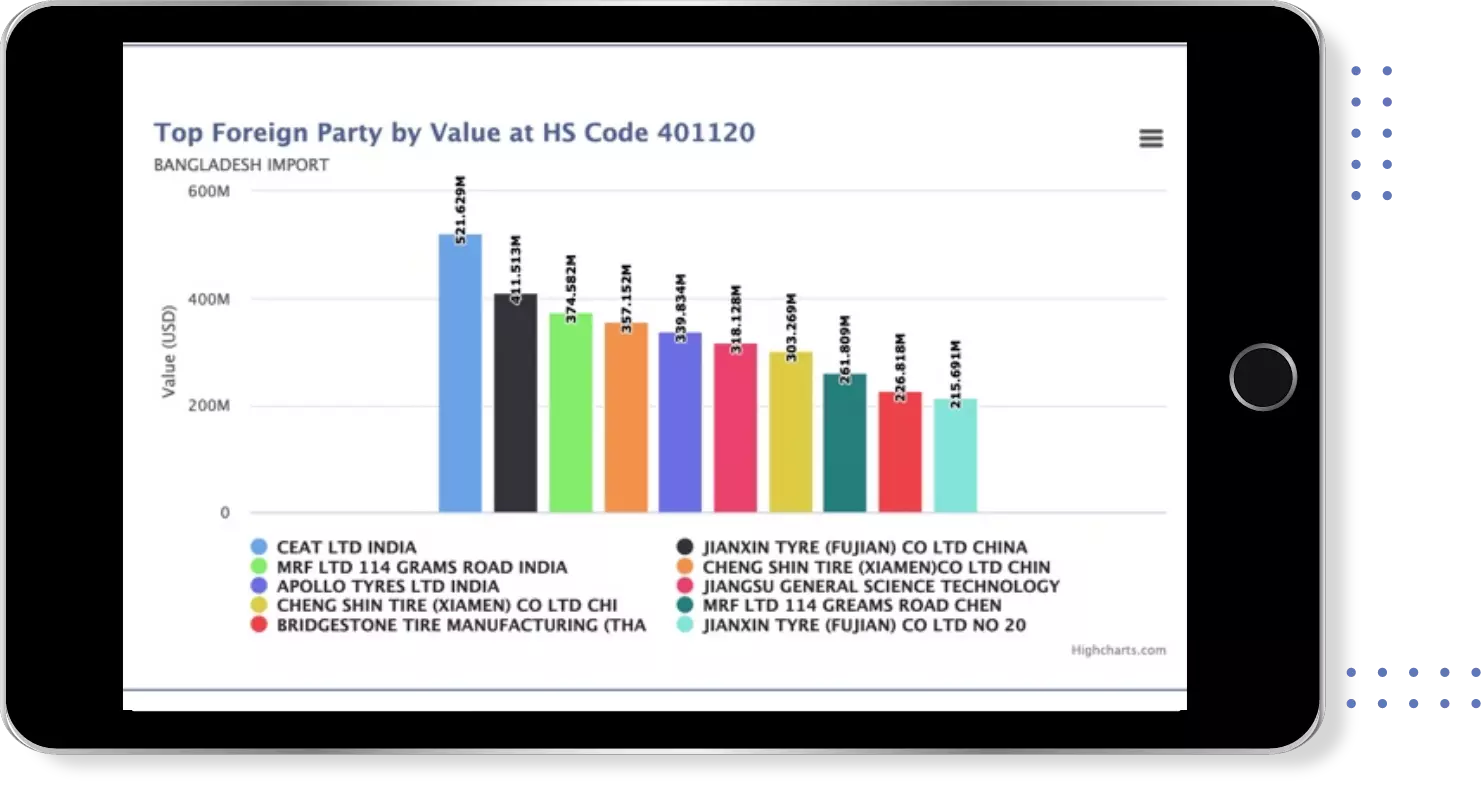
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
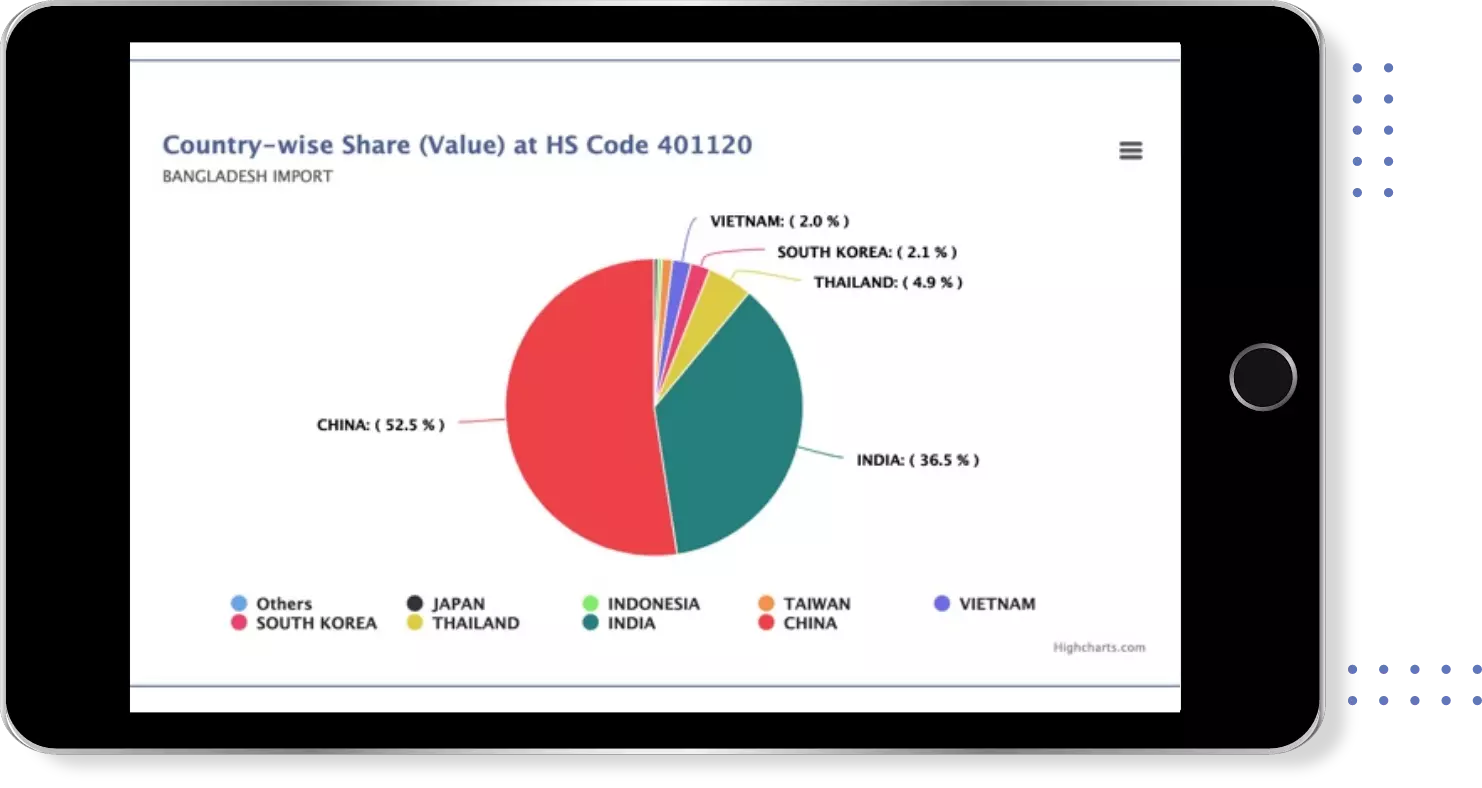
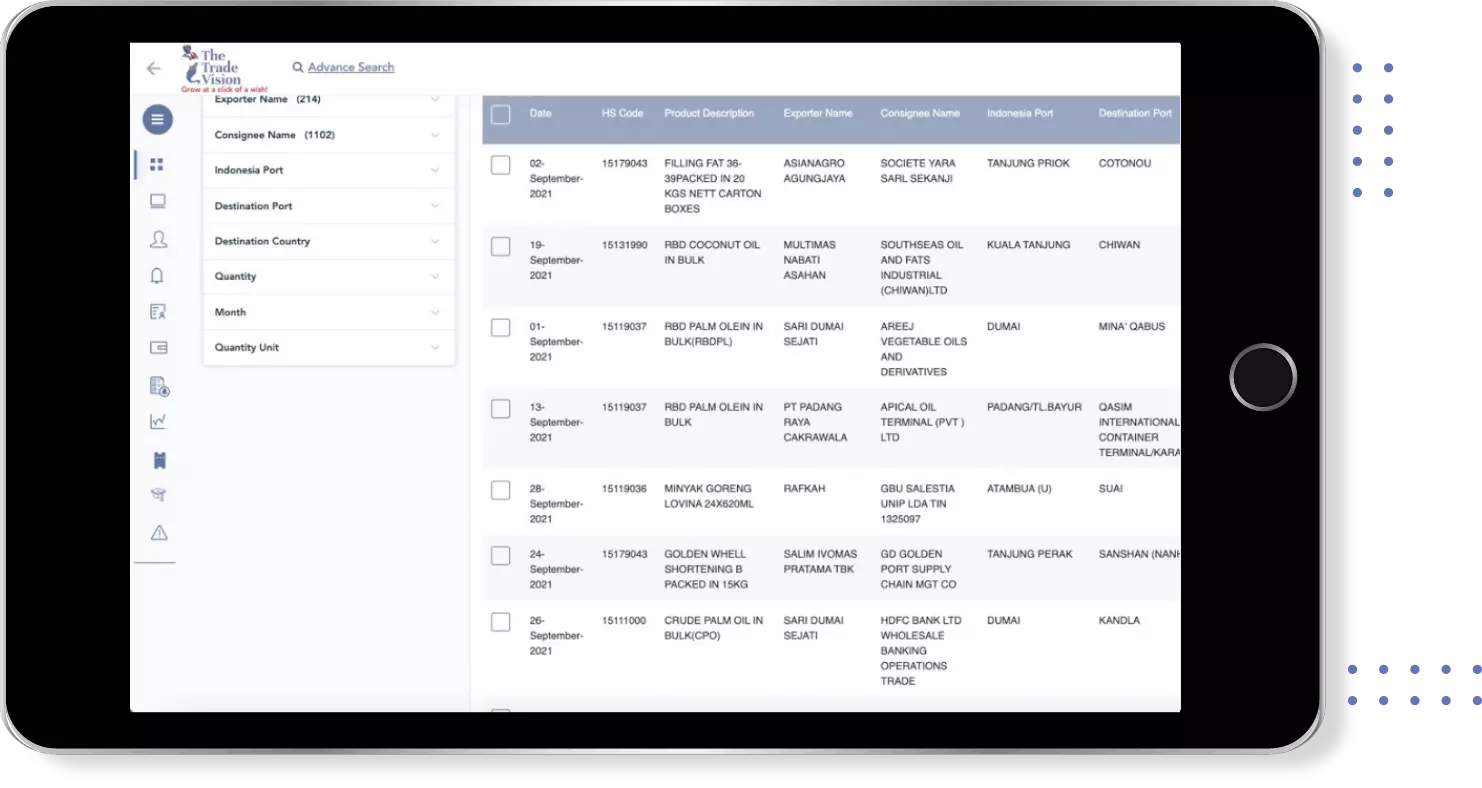
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.