Find verified buyers and sellers of each tablet in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- each tablet buyers in ukraine
- COD Ukraine
Ukraine Buyers of each tablet
BADM LTD LIABILITY COMPANY

UKRAINE
HS-Code
Products
3004200000
1. MEDICINES FOR PEOPLE CONTAINING ANTIBIOTICS PACKAGED FOR RETAIL SALE NOT IN AEROSOL PACKAGING: SORCEF® FILM-COATED TABLETS 400 MG 7 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX / 1 TABLET FILM-COATED CONTAINING CEFIXIME 400 MG (AS CEFIXIME TRIHYDRATE 447 630 MG) MANUFACTURER ALKALOID AD-SKOPJE REPUBLIC OF MACEDONIA SORCEF ® FILM-COATED TABLETS 400 MG 5 TABLETS IN A BLISTER 2 BLISTERS IN EACH BOX / 1 FILM-COATED TABLET CONTAINING CEFIXIME 400 MG (AS CEFIXIME TRIHYDRATE 447 630 MG); MANUFACTURER ALKALOID AD-SKOPJE REPUBLIC OF MACEDONIA;
3004200000
1. MEDICINES FOR PEOPLE CONTAINING ANTIBIOTICS PACKAGED FOR RETAIL SALE NOT IN AEROSOL PACKAGING: SORCEF® FILM-COATED TABLETS 400 MG 7 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX / 1 TABLET FILM-COATED CONTAINING CEFIXIME 400 MG (AS CEFIXIME TRIHYDRATE 447 630 MG) MANUFACTURER ALKALOID AD-SKOPJE REPUBLIC OF MACEDONIA SORCEF ® FILM-COATED TABLETS 400 MG 5 TABLETS IN A BLISTER 2 BLISTERS IN EACH BOX / 1 FILM-COATED TABLET CONTAINING CEFIXIME 400 MG (AS CEFIXIME TRIHYDRATE 447 630 MG); MANUFACTURER ALKALOID AD-SKOPJE REPUBLIC OF MACEDONIA;
3006600000
1. MEDICINES FOR HUMANS: HORMONE-BASED CONTRACEPTIVES PACKAGED FOR RETAIL NOT IN AEROSOL PACKAGING: BELARA ® FILM-COATED TABLETS 2 MG / 0.03 MG 21 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD PACKAGE / 1 FILM-COATED TABLET CONTAINS: CHLORMADINONE ACETATE 2 MG ETHINYL ESTRADIOL 0.03 MG MANUFACTURER GEDEON RICHTER HUNGARY BELARA ® FILM-COATED TABLETS 2 MG / 0.03 MG 21 TABLETS IN A BLISTER 3 BLISTERS IN A CARTON / 1 FILM-COATED TABLET CONTAINING: CHLORMADINONE ACETATE 2 MG ETHINYL ESTRADIOL 0.03 MG MANUFACTURER GEDEON RICHTER HUNGARY VENDIOL FILM-COATED TABLETS 0.06 MG / 0.015 28 FILM-COATED TABLETS IN A BLISTER (24 YELLOW ACTIVE TABLETS AND 4 GREEN PLACEBO TABLETS); 1 BLISTER EACH BLISTER - IN A LAMINATED SACHET WITH A FLAT CARDBOARD CASE FOR STORING THE BLISTER AND A WEEKLY CALENDAR-STICKER IN A CARDBOARD PACKAGE / 1 ACTIVE TABLET CONTAINS 0.060 MG OF GESTODENE (MICRONIZED) AND 0.015 MG OF ETHINYL ESTRADIOL (MICRONIZED; MANUFACTURER OJSC RICHTER GETERTER HUNGARY LACTINET ®-RICHTER FILM-COATED TABLETS 0.075 MG 28 TABLETS IN A BLISTER 1 BLISTER IN A PACKAGE OF LAMINATED ALUMINUM FOIL 1 PACKAGE WITH A CARDBOARD CASE FOR STORING THE BLISTER IN A CARDBOARD PACKAGE / 1 FILM-COATED CONTAINING 0.075 MG DESOGESTREL MANUFACTURER OF OJSC GIDEON RICHTER HUNGARY LINDINET 20 COATED TABLETS 21 TABLETS IN A BLISTER 1 BLISTER TOGETHER WITH A CARDBOARD CASE FOR STORING THE BLISTER IN A PACKAGE / 1 TABLET CONTAINS 02 MG AND GESTODENE 0.075 MG MANUFACTURER OJSC GIDEON RICHTER HUNGARY NOVINET COATED TABLETS 0.15 MG / 0.02 MG 21 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD PACKAGE / 1 FILM-COATED TABLET CONTAINING 0.15 MG DESOGESTREL AND 0.02 MG ETHINYL ESTRADIOL MANUFACTURER GEDEON RICHTER HUNGARY RIGEVIDON FILM-COATED TABLETS 21 TABLETS IN A BLISTER 1 BLISTER / PACK IN A BLISTER FILM-COATED TABLET CONTAINS: LEVONORGESTREL 0.15 MG AND ETHINYL ESTRADIOL 0.03 MG MANUFACTURER GEDEON RICHTER HUNGARY SILHUET ® FILM-COATED TABLETS 21 TABLETS IN A BLISTER 1 BLISTER TOGETHER WITH A CARDBOARD CASE FOR STORAGE OF THE BLISTER IN A PACK / 1 FILM-COATED TABLET CONTAINING DIENOGEST 2 MG AND ETHINYL ESTRADIOL 0.03 MG MANUFACTURER OJSC GIDEON RICHTER HUNGARY;
JV OPTIMA PHARM LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES H8C015A: NIFUROXAZIDE RICHTER COATED TABLETS 100 MG 24 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX -14280 PACK - (ACTIVE SUBSTANCE) : NIFUROXAZIDE RICHTER COATED TABLETS 100 MG 24 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX -4200 PCS. (ACTIVE INGREDIENT - NIFUROXAZIDE) MANUFACTURER: QUALITY CONTROL PERMISSION TO RELEASE A BATCH PRODUCTION OF UNPACKAGED SECONDARY PACKAGING: LLC GIDEON RICHTER POLAND PL. SERIES H87051C: SENTOR® FILM-COATED TABLETS 100 MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD PACKAGE - 84 UNITS (ACTIVE SUBSTANCE - LOSARTAN POTASSIUM) SERIES H88007B: SENTOR® FILM-COATED TABLETS 50 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD PACKAGE -72 PACK (ACTIVE SUBSTANCE - LOSARTAN POTASSIUM) MANUFACTURER: GEDEON RICHTER POLAND LLC PL. SERIES H8C025A: SINGLON® CHEWABLE TABLETS 4 EACH MG N
3006600000
1. MEDICINAL PRODUCTS FOR HUMAN TREATMENT CHEMICAL CONTRACEPTIVES PACKAGED FOR RETAIL SALE BASED ON HORMONAL COMPOUNDS. CHEMICAL CONTRACEPTIVES. SERIES U9B006C: VENDIOL FILM-COATED TABLETS 0.06 MG / 0.015 MG; 28 TABLETS IN A BLISTER ( 24 YELLOW ACTIVE TABLETS AND 4 GREEN PLACEBO TABLETS) 1 BLISTER (EACH BLISTER-IN A LAMINATED SACHET WITH A FLAT CARDBOARD CASE FOR STORING THE BLISTER AND A WEEKLY CALENDAR-STICKER) IN A CARDBOARD PACKAGE - 132 PCS. MANUFACTURER: JSC GIDEON RICHTER HU.
3004500000
1. MEDICINES FOR PEOPLE CONTAINING VITAMINS PACKAGED FOR RETAIL TRADE. SERIES 0000137534: BRONCHIPRET® SYRUP ON 100 ML IN A BOTTLE; ON 1 BOTTLE IN A CARDBOARD BOX - 640 UNITARY ENTERPRISE. SERIES 0000132825: BRONCHIPRET ON A COVER IN T® 20 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX -165 PACK SERIES 0000133766: BRONHIPRET® TP COATED TABLETS 25 TABLETS IN A BLISTER 2 BLISTERS IN A CARDBOARD BOX -300 PACK SERIES 0000129513: IMU ORAL 100 ML IN A BOTTLE 1 BOTTLE IN A CARDBOARD BOX -240 PACK SERIES 0000131667: IMUPRET® COATED TABLETS 25 TABLETS IN A BLISTER 2 BLISTERS IN A CARDBOARD BOX -600 PACK SERIES 00001344PRE ORAL DROPS 100 ML IN A VIAL WITH A DOSING DROPPER ON TOP WITH A SCREW CAP AND AN OPENING CONTROL RING 1 VIAL IN A CARDBOARD BOX -4040 PACK SERIES 0000134286: SINUPRET® SYRUP 100 ML PER VIAL 1 VIAL EACH MEASURING CAP IN A CARDBOARD BOX -2520 UNITARY ENTERPRISE SERIES 0000131815 :
LLC SIMEDIKA UA

UKRAINE
HS-Code
Products
3004900000
1. VETERINARY DRUGS PACKAGED FOR RETAIL TRADE: HARTMEDIN (R) 10MG TABLETS BLISTER OF POLYVINYL VINYL / POLYETHYLENE / POLYVINYL CHLORIDE CLOSED WITH ALUMINUM FILM EACH BOX CONTAINS 3 BLISTERS OF 10 TABLETS SERIES SERIES 1883210 - 200MP CONTAINS 200MP. ) TRADEMARK: HARTMEDIN. MANUFACTURER: SIMEDICA SEX. S.R.O.COUNTRY OF PRODUCTION: CZ.
3004900000
1. RAZFASOVANI VETERINARY MEDICINAL PRODUCTS FOR RETAILERS: HARTMEDIN (R) 1 0MH TABLETS BLISTER POLIHROLVINILU / POLYETHYLENE / PVC CLOSED ALUMINUM FOIL EACH BOX CONTAINS 3 BLISTER PILLS 1 0 IN SERIES SERIES 1 88 321 2 - 1 62UP. SERIES 1 88321 0 - 38UP. (1 TABLET CONTAINS: PIMOBENDAN 1 0MG) TRADEMARK: HARTMEDIN. MANUFACTURER: SEX SYMEDICS. S. R. O. COUNTRY OF PRODUCTION: CZ.
3004390000
1. VETERINARY MEDICINES PACKAGED FOR RETAIL TRADE: VETORIL 10 MG HARD CAPSULES POLYVINYL CHLORIDE BLISTERS COATED WITH ALUMINUM 10 CAPSULES EACH BOX CONTAINS 3 BLISTERS SERIES 118865 - 30 PACK (1 CAPSULE CONTAINS: TRILOSTAN 10 MG TABLETS 30 MG) POLYVINYL CHLORIDE COATED WITH ALUMINUM 10 CAPSULES EACH BOX CONTAINS 3 BLISTERS SERIES 118900 - 30UP. (1 CAPSULE CONTAINS: TRILOSTAN 30 MG) VETORIL 60 MG HARD CAPSULES POLYVINYL CHLORIDE BLISTERS ALUMINUM COATED WITH 10 CAPSULES EACH BOX CONTAINS 3 BLISTERS SERIES 119256 - 60 PACK. (1 CAPSULE CONTAINS: TRILOSTAN 60 MG) TRADEMARK: VETORIL. MANUFACTURER: DALES PHARMACEUTICAL LIMITED. COUNTRY OF MANUFACTURE: GB.
REPRESENTATIVE OFFICE JANSEN PHARMACEUTICALS NV

UKRAINE
HS-Code
Products
3923900000
1. CONCOMITANT PLASTIC CONSUMABLES FOR TRANSPORTING SAMPLES IN A CLINICAL TRIAL: KITS FOR LABORATORY TESTING (TYPE 2I) INCLUDING: DAY 2 (DAY 2): ART.3 -19PCS EACH CONTAINING: PLASTIC TEST TUBE WITH A TABLET OF BORIC ACID AND A TRANSPARENT LID 10ML -1PC PLASTIC STERILE PIPETTE WITH LABELS VOLUME 3.5ML -1PC ADDITIONAL BARCODE LABELS (SPARE) -2PCS LARGE LABEL FOR MIXED SAMPLES -1PC CONICAL TEST TUBE FOR TRANSPORTING SAMPLES WITH A PRESERVATIVE TABLET 10ML - 1PC PACKAGE FOR MOISTURE ABSORPTION -1PC CCLS APPLICATION FORM WITH A BARCODE -1PC STERILE PLASTIC CONTAINER FOR TRANSPORTATION OF URINE SAMPLES WITH A STAND -1PC DNA (DNA): ART.T-3 -16PCS EACH CONTAINS: PLASTIC TEST TUBE WITH TABLET BORIC ACID AND TRANSPARENT LID 10ML -1PC PLASTIC STERILE PIPETTE WITH LABELS VOLUME 3.5ML -1PC ADDITIONAL BARCODE LABELS (SPARE) -2PCS LARGE LABEL FOR MIXED SAMPLES -1PC CONICAL TEST TUBE FOR TRANSPORTING SAMPLES FROM PRESERVING TABLET 10ML -1PC MOISTURE ABSORPTION PACKAGE -1PC CCLS APPLICATION FORM WITH BARCODE -1PC STERILE PLASTIC CONTAINER FOR TRANSPORTING URINE SAMPLES WITH A STAND -1PC SCREENING RENAL BIOPSY (SCREEN RENAL BIOPSY): ART.T-4 - 9PCS EACH CONTAINS: PLASTIC TEST TUBE WITH BORIC ACID TABLET AND CLEAR LID 10ML -1PC PLASTIC STERILE PIPETTE WITH LABELS VOLUME 3.5ML -1PC ADDITIONAL BARCODE LABELS (SPARE) -2PCS LARGE LABEL FOR MIXED SAMPLES -1PC CONICAL TEST TUBE FOR TRANSPORTING SAMPLES WITH PRESERVATIVE TABLET 10ML -1PC MOISTURE ABSORBING PACKAGE -1PC CCLS APPLICATION FORM WITH BARCODE -1PC STERILE PLASTIC CONTAINER FOR TRANSPORTING URINE SAMPLES WITH STAND -1PC WEEK 52 KIDNEY BIOPSY (WEEK 52 RENAL BIOPSY): ART.T-5 -14PCS EACH CONTAINS: PLASTIC TEST TUBE WITH BORIC ACID TABLET AND TRANSPARENT LID 10ML -1PC PLASTIC STERILE PIPETTE WITH LABELS VOLUME 3.5ML -1PC ADDITIONAL BARCODE LABELS (SPARE) -2PCS VEL IKA LABEL FOR MIXED SAMPLES -1PC CONICAL TEST TUBE FOR TRANSPORTING SAMPLES WITH PRESERVATIVE TABLET 10ML -1PC PACKAGE FOR MOISTURE ABSORPTION -1PC APPLICATION FORM CCLS WITH BARCODE -1PC STERILE PLASTIC CONTAINER FOR TRANSPORTING URINE SAMPLES WITH STAND -1PC FAMA POPULATION (POPULATION PK): ART.T-6 -68PCS EACH CONTAINS: PLASTIC TEST TUBE WITH A TABLET OF BORIC ACID AND A TRANSPARENT LID 10ML -1PC PLASTIC STERILE PIPETTE WITH MARKS VOLUME 3.5ML -1PC ADDITIONAL LABELS BAR- CODES (SPARE) -2PCS LARGE LABEL FOR MIXED SAMPLES -1PCS CONICAL TEST TUBE FOR TRANSPORTING SAMPLES WITH PRESERVATIVE TABLET 10ML -1PC PACKAGE FOR MOISTURE ABSORPTION -1PCS CCLS APPLICATION FORM WITH BARCODE -1PCS STERILE PLASTIC CONTAINER FOR TRANSPORTING SAMPLES URINE WITH STAND -1PC AM PRIMARY ANALYSIS OF URINE SAMPLES (URINE FIRST AM VOID) ART.T-8 -43PCS EACH CONTAINS: PLASTIC TEST TUBE WITH A TABLET OF BORIC ACID AND A TRANSPARENT LID 10M
3923900000
1. CONCOMITANT PLASTIC CONSUMABLES FOR TRANSPORTING SAMPLES IN A CLINICAL TRIAL: KITS FOR LABORATORY TESTING (TYPE 4I) INCLUDING: DAY 78 ART.7 -2PCS EACH CONTAINS: APPLICATION FORM CCLS WITH BARCODE -1PCS PLASTIC TEST TUBE WITH DOUBLE BOTTOM WITH AMBER LID 5ML -1PC CONTAINER WITH TWO SLIDES -1PC CONICAL TEST TUBE WITH ORANGE LID WITH CANNING TABLET 10ML -1PC PLASTIC TUBE WITH SODIUM CITRATE WITH BLUE LID 1.8ML -1PC FOR PLASTIC WITH SCREW CAP 3.5ML -1PC PLASTIC TEST TUBE WITH DOUBLE BOTTOM WITH TRANSPARENT LID 5ML -2PCS PLASTIC TEST TUBE WITH PURPLE LID WITH K2 EDTA 6ML -2PCS TEST TUBE COVERED WITH SILICONE SHELL WITH RED LID - 10ML 2PCS PLASTIC STERILE CONICAL TEST TUBE 15ML -1PC PLASTIC TEST TUBE WITH PURPLE LID WITH K2 EDTA 10ML -1PC PLASTIC CONICAL CRYOTUBE WITH SCREW CAP 5 ML -1PC PLASTIC TEST TUBE WITH PURPLE LID WITH K2
3923900000
1. CONCOMITANT PLASTIC CONSUMABLES FOR TRANSPORTING SAMPLES IN A CLINICAL TRIAL: KITS FOR LABORATORY TESTS (TYPE 4I) CONSIST OF: END OF TREATMENT / AFTER TREATMENT WEEK 4 (EOT POST-TREATMENT WEEK 4): ART.T-1 -3PCS EACH CONTAINS: APPLICATION FORM CCLS WITH BAR CODE -1PC PLASTIC TEST TUBE WITH DOUBLE BOTTOM WITH TRANSPARENT LID 5ML -4PCS PLASTIC TEST TUBE WITH DOUBLE BOTTOM WITH AMBER LID 5ML -2PCS STERILE PLASTIC CONICAL TEST TUBE WITH CANNING TABLET -3 15M PLASTIC TEST TUBE FOR BLOOD SAMPLING WITH YELLOW-GOLD LID 3 5ML -2PCS TEST TUBE FOR BLOOD SAMPLING WITH GOLD CAP WITH GEL SEPARATOR 2 5ML -2PCS PLASTIC STERILE PIPETTE WITH MARKS VOLUME 3 1ML -4PCS DISPOSABLE PLASTIC TEST TUBE HOLDER NON-STERILE -1PC NEEDLE (MULTI-INTAKE) 21G X 1 25 IN. ECLIPSE -1PC PLASTIC CONICAL TEST TUBE FOR TRANSPORTATION WITH YELLOW LID 10ML -1PC PACKAGE FOR MOISTURE ABSORPTION -1PC TRANSPARENT PLASTIC
LLC TRADING YARD UTS

UKRAINE
HS-Code
Products
2501009900
1. TABLET SALT UNIVERSAL (SALT TABLETS). USED TO SOFTEN WATER IN HEATING SYSTEMS (RESTORATION OF ION EXCHANGE EXCHANGERS). NOT FOR HUMAN CONSUMPTION. PACKED IN BAGS OF 25 KG. NET. 1050 KG EACH. NET IN STRETCH-HOOD ON ONE PALLET. ONLY 60 PALLETS. TOTAL NET WEIGHT - 63000 KG. TRADEMARK - MOZYRSOL. MANUFACTURER - JSC MOZYRSOL. COUNTRY OF MANUFACTURE - BY.
2501009900
1. TABLET SALT UNIVERSAL (SALT TABLETS). USED TO SOFTEN WATER IN HEATING SYSTEMS (RESTORATION OF ION EXCHANGE EXCHANGERS). NOT FOR HUMAN CONSUMPTION. PACKED IN BAGS OF 25 KG. NET. 1050 KG EACH. NET IN STRETCH-HOOD ON ONE PALLET. ONLY 60 PALLETS. TOTAL NET WEIGHT - 63000 KG. TRADEMARK - MOZYRSOL. MANUFACTURER - JSC MOZYRSOL. COUNTRY OF MANUFACTURE - BY.
2501009900
1. TABLET SALT UNIVERSAL (SALT TABLETS). USED TO SOFTEN WATER IN HEATING SYSTEMS (RESTORATION OF ION EXCHANGE EXCHANGERS). NOT FOR HUMAN CONSUMPTION. PACKED IN BAGS OF 25 KG. NET. 1050 KG EACH. NET IN STRETCH-HOOD ON ONE PALLET. ONLY 60 PALLETS. TOTAL NET WEIGHT - 63000 KG. TRADEMARK - MOZYRSOL. MANUFACTURER - JSC MOZYRSOL. COUNTRY OF MANUFACTURE - BY.
LLC CREATIVE GROUPS OF THE COMPANY

UKRAINE
HS-Code
Products
2106909200
1. FOOD PRODUCTS TO THE DIET OF PEOPLE PACKAGED FOR RETAIL TRADE. COMPOSITE PRODUCTS THAT MEET THE CONDITIONS SPECIFIED IN PART ELEVEN OF ARTICLE 41 OF THE LAW OF UKRAINE ON STATE CONTROL OVER COMPLIANCE WITH LEGISLATION ON FOOD FEED ANIMAL BY-PRODUCTS HEALTH AND WELFARE ANIMALS AS WELL AS PACKAGED FOR THE FINAL CONSUMER DIETARY SUPPLEMENTS: -ZINCIT - DIETARY SUPPLEMENT EFFERVESCENT TABLETS WEIGHING 4.5 G ?20 IN PLASTIC TUBES (10 TABLETS EACH) PACKED IN A CARDBOARD BOX SERIES: 21C259 -3000UPMANUFACTURER: VORVAG PHARMA PHARMA GM . KG GERMANYCOUNTRY OF PRODUCTION: DETRAND: IN THE FORM OF DRAWING
2106909200
1. FOOD PRODUCTS; DIETARY SUPPLEMENTS PACKED FOR RETAIL: -VITAGAMA D3 2000 IU DIETARY SUPPLEMENT TABLETS 10 TABLETS IN A BLISTER; 5 BLISTERS IN A CARDBOARD BOX SERIES: 19L055 -1600UP-GLUCOSE VITAL - A DIETARY SUPPLEMENT TO THE DIET 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX SERIES: 19P086 -648UP-ZINCIT - DIETARY SUPPLEMENT EFFERVESCENT TABLETS 10 TABLETS EACH) PACKED IN A CARDBOARD BOX SERIES: 19D246 -734UPMANUFACTURER: VORVAG PHARMA GMBH & CO. KG GERMANYCOUNTRY OF PRODUCTION: DETRAND: IN THE FORM OF DRAWING
2106909200
1. FOOD PRODUCTS; DIETARY SUPPLEMENTS FOR PEOPLE NOT OF ANIMAL ORIGIN PACKED FOR RETAIL TRADE. COMPOSITE PRODUCTS THAT MEET THE CONDITIONS SPECIFIED IN PART ELEVEN OF ARTICLE 41 OF THE LAW OF UKRAINE ON STATE CONTROL OVER COMPLIANCE WITH LEGISLATION ON FOOD PRODUCTS ANIMAL BY-PRODUCTS HEALTH AND ANIMAL WELFARE PACKAGED FOR THE END CONSUMER DIETARY SUPPLEMENTS: -VITAGAMA D3 2000 IU DIETARY SUPPLEMENT TABLETS 10 TABLETS IN A BLISTER; 5 BLISTERS IN A CARDBOARD BOX SERIES: 20G110 -2750UP-ZINCIT - DIETARY SUPPLEMENTS DIETARY SUPPLEMENTS 5G ?20 IN PLASTIC TUBES (10 TABLETS IN EACH) PACKED IN A CARDBOARD BOX SERIES: 20H216 -5284UP MANUFACTURER: VORVAG PHARMA GMBH AND CO. KG GERMANY COUNTRY OF MANUFACTURE: DETRAND: IN THE FORM OF A PICTURE
TOV VENTA LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. EVKABAL® DROPS FOR CHILDREN DROPS 0.5 MG / ML IN A 10 ML VIAL 1 VIAL IN A CARDBOARD PACKAGE / 1 ML OF SOLUTION CONTAINS XYLOMETAZOLINE HYDROCHLORIDE 0.5 MG / ESCUZAN® LONG PROLONGED-RELEASE CAPSULES OF 50 MG 10 CAPSULES IN A BLISTER 6 BLISTERS IN A CARDBOARD BOX / 1 CAPSULE CONTAINS DRY EXTRACT OF HORSE CHESTNUT SEEDS (HORSE CHESTNUT SEEDS EXTRACT) (4.5-5.5: 1) 240-290 MG CORRESPONDING TO THE CONTENT OF TRITERPENE GLYCOSIDES IN TERMS OF ANHYDROUS ESCIN - 50 MG / TEMMLER IRELAND ISLAND LIMITED (PRODUCTION IN BULK BATCH CONTROL) IRELANDDRAGENOPHARM APOTHECARY PÜSCH GMBH (PRIMARY AND SECONDARY PACKAGING) GERMANYSWISS CAPS GMBH (PRIMARY AND SECONDARY PACKAGING) GERMANYESPARM GMBH (BATCH RELEASE) GERMANYSUSTAMAR® FILM-COATED TABLETS10 480 EACH; BLISTER IN A BLISTER; 5 BLISTERS IN A CARDBOARD BOX / 1 FILM-COATED TABLET CONTAINS DRY EXTRACT OF FRAGRANT MARTINI ROOT (EXTRACTUM HARPAGOPHYTUM PROCUMBENS SICCUM) 4.5–5: 1 (EXTRACTANT? ETHANOL 60%) 480 MG / PHARMA WERNIGERODE GMBH ) GERMANYSPHARMA PHARMA SERVICES GMBH (SECONDARY PACKAGING) GERMANYADVANCE PHARMA GMBH (PRODUCTION OF BULK PRODUCT PRIMARY PACKAGING SECONDARY PACKAGING QUALITY CONTROL BATCH PRODUCTION)) GERMANY
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. MEDOGISTIN TABLETS OF 16 MG; 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS BETAHISTINE DIHYDROCHLORIDE 16 MG / 190826-001 / UA / 13526/01/01 UNLIMITED FROM 16.08.2017MEDOKEMI LTD (CENTRAL PLANT) CYPRUSMEDOCHEMIE CYMEDOGISTIN TABLETS OF 24 MG 10 TABLETS IN EACH; 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS BETAHISTINE DIHYDROCHLORIDE 24 MG / 191122-001 / UA / 13526/01/02 UNLIMITED FROM 16.08.2017MEDOKEMI LTD (CENTRAL PLANT) CYPRUSMEDOCHEMIE CYMEDOKARDIL TABLETS OF 6.25 MG; 10 TABLETS; 3 BLISTERS IN A CARTON / 1 TABLET CONTAINS CARVEDILOL 6.25 MG / E4A027 / UA / 9479/01/01 UNLIMITED FROM 13.06.2019 AND FILM-COATED 10 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS MEMANTINE HYDROCHLORIDE10 MG / E4E075 / UA / 16845/01/01 13.07.2018-13.07.2023MEDOKEMI LTD (CENTRAL PLANT) PRODUCTION ON A FULL CYCLE) CYPRUSMEDOKEMI LTD (PLANT AZ) (PRIMARY AND SECONDARY PACKAGING OF THE FINISHED DRUG) CYPRUSMEDOKEMI LTD (CENTRAL PLANT) (PRODUCTION ON A FULL CYCLE) KIPRMEDOCHEMIE CYAKTIPROL TABLETS IN 3 MG TABLETS OF 10 MG 10 BOX / 1 TABLET CONTAINS AMISULPRIDE 100 MG / E4H034 / UA / 17654/01/01 13.09.2019-13.09.2024MEDOKEMI LTD (CENTRAL PLANT) (FULL CYCLE PRODUCTION) CYPRUSMEDOKEMI LTD (PLANT AZ) (PRIMARY AND SECONDARY PACKAGING) KIPRMEDOKEMI LTD (CENTRAL PLANT) (FULL CYCLE PRODUCTION) KIPRMEDOCHEMIE CYMAGUROL TABLETS OF 4 MG ? 20 (10X2) IN BLISTERS / 1 TABLET CONTAINS DOXAZOSINUM MESYLATE 4.86 MG EQUIVALENT TO DOXAZOSIN 4 MG / 02/433/120 .2016-16.03.2021MEDOKEMI LTD (THIS NEUTRAL PLANT) CYPRUSMEDOCHEMIE CYMEDOGREL FILM-COATED TABLETS OF 75 MG 10 TABLETS IN A BLISTER OF POLYVINYL CHLORIDE AND ALUMINUM FOIL 3 BLISTERS IN A BOX; / 1 TABLET CONTAINS CLOPIDOGREL BISULFATE 97.86 MG WHICH IS EQUIVALENT TO CLOPIDOGREL 75 MG / E4J008 / EN / 12149/01/01 UNLIMITED FROM 13.05.2017MEDOKEMI LTD (CENTRAL PLANT) (PRODUCTION OF BULK PRODUCT QUALITY CONTROL PERMISSION TO RELEASE A SERIES) CYPRUS PLANT AZ) (PRIMARY AND SECONDARY PACKAGING) CYPRUSMEDOKEMI LTD (CENTRAL PLANT) (PRODUCTION OF BULK PRODUCT QUALITY CONTROL PERMISSION TO MEDOCHEMIE CYRAMIMED TABLETS OF 10 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS / E4E028 / EN / 10153/01/03 UNLIMITED FROM 31.10.2019 MEDOKEMI LTD (CENTRAL PLANT) (PRODUCTION OF BULK PRODUCT QUALITY CONTROL BATCH PRODUCTION PERMIT) CYPRUSMEDOKEMI LTD (PLANT AZ) (PRIMARY AND SECONDARY PACKAGING) CYPRUS MEDOKEMI CENTRAL FACTORY)
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. LIPRIMAR® TABLETS FILM-COATED 20 MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS ATORVASTATIN CALCIUM EQUIVALENT TO 20 MGATORVASTATIN / PFIZER PHARMACEUTICALS LLS (PRODUCTION IN BULK QUALITY CONTROL) USA. INSPRA® FILM-COATED TABLETS 50 MG AND 10 TABLETS EACH; 3 BLISTERS IN A CARTON / 1 TABLET CONTAINING 50 MG OF EPLERENONE / PFIZER PHARMACEUTICALS LLS (PRODUCTION IN BULK AND QUALITY CONTROL) USA.
TOV VENTA Â LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE FREE OF: ANTIBIOTICS ALKALOIDS HORMONES VITAMINS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: ESTULIK® TABLETS OF 1 MG 10 TABLETS IN A BLISTER 2 BLISTERS IN A CARDBOARD BOX / EACH TABLET CONTAINS 1 MG 1.15 MG GUANFACINE HYDROCHLORIDE) / ALOTENDIN TABLETS 5 MG / 5 MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARTON / 1 TABLET CONTAINS 5 MG OF BISOPROLOL FUMARATE AND 5 MG OF AMGLADIPINE CORRESPONDING TO 6.95 MG OF AMLODIPINE BESYLATE / ALOTENDIN TABLETS OF 5 MG / 10 MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARTON / 1 TABLET CONTAINS 5 MG OF BISOPROLOL FUMARATE AND 10 MG OF AMGLADIPINE WHICH CORRESPONDS TO 13.9 MG OF AMLODIPINE BESYLATE / SUPRASTIN® TABLETS OF 25 MG 10 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS 25 MG OF CHLOROPYRAMINE HYDROCHLORIDE; / LUCETAM® FILM-COATED TABLETS 800 MG 15 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS 800 MG OF PIRACETAM
3004900000
1. MEDICINES FOR HUMANS FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. BETALOK ZOK FILM-COATED TABLETS WITH SLOW-RELEASE MG 14 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS 23.75 MG OF METOPROLOL SUCCINATE CORRESPONDING TO 25 MG OF METOPROLOL TARTRATE / BETALOK ZOK FILM-COATED TABLETS WITH A SLOW RELEASE OF 100 MG 30 TABLETS IN A VIAL; 1 VIAL IN A CARDBOARD BOX / 1 TABLET CONTAINS 95 MG OF METOPROLOL SUCCINATE CORRESPONDING TO 100 MG OF METOPROLOL TARTRATE / BETALOK ZOK FILM-COATED TABLETS WITH A SLOW RELEASE OF 50 MG 30 TABLETS IN A VIAL; 1 VIAL IN A CARDBOARD BOX / 1 TABLET CONTAINS 47.5 MG OF METOPROLOL SUCCINATE CORRESPONDING TO 50 MG OF METOPROLOL TARTRATE / BRILINTA FILM-COATED TABLETS 60 MG; 14 TABLETS EACH
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. METOTAB TABLETS OF 7.5 MG ?30 IN VIALS / 1 TABLET CONTAINS 7.5 MG OF METHOTREXATE (AS METHOTREXATE DISODIUM) / EXCELLA GMBH (MANUFACTURER RESPONSIBLE FOR THE PRODUCTION OF THE FINISHED DOSAGE FORM PRIMARY AND SECONDARY PACKAGING LABELING OF PRIMARY PACKAGING CONTROL / TESTING SERIES) GERMANYGEMCITABIN MEDAC POWDER FOR SOLUTION PREPARATION 38 MG / ML) 1500 MG EACH; 1 VIAL WITH POWDER IN A CARDBOARD BOX / 1 ML OF RECONSTITUTED SOLUTION FOR INFUSION CONTAINS 38 MG OF GEMCITABINE (AS GEMCITABINE HYDROCHLORIDE) / MEDAK GESELSHAFT FÜR KLINICHE SPETSIALPREPARATE MBH (MANUFACTURER RESPONSIBLE FOR REPACKAGING APPLICATION OF PROTECTIVE FILM ON THE VIAL) SERIES) GERMANYONKOTEK PHARMA PRODUCTION GMBH THE MANUFACTURER RESPONSIBLE FOR THE PRODUCTION OF THE FINISHED DOSAGE FORM
HFC BIOCON LLC

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR HUMANS PACKAGED FOR RETAIL TRADE: -VIRDAK 60 (ACTIVE SUBSTANCES: 1 TABLET CONTAINS: DACLATASVIRUDIHYDROCHLORIDE EQUIVALENT TO DACLATASVIRU60MG) FILM-COATED TABLETS 60MG EACH 28MG 28 TABLETS IN CONTAINER2 CONTAINER; 1719UP MANUFACTURER: GETERO LABZ LIMITED INDIACOUNTRY OF PRODUCTION: INBRAND: VIRDAK 60 EXPIRY DATE: 30.09.2023
0000210690
1. DIETARY SUPPLEMENT TO THE DIET OF MAN PACKAGED FOR RETAIL NON-ANIMAL ORIGIN: -ZINCIT - DIETARY SUPPLEMENT EFFERVESCENT TABLETS WEIGHING 4.5 G ?20 IN PLASTIC TUBES (10 TABLETS EACH) PACKED IN A CARDBOARD BOX SERIES: GMBH & CO. KG GERMANYCOUNTRY OF PRODUCTION: DETRAND: IN THE FORM OF DRAWING EXPIRATION DATE: 31.05.2024
0000300660
1.PHARMACEUTICALS CHEMICAL CONTRACEPTIVES BASED ON HORMONES PACKAGED FOR RETAIL TRADE: -VENDIOL FILM-COATED TABLETS 0.06 MG / 0.015 MG FOR 28 TABLETS IN A BLISTER (24 YELLOW ACTIVE TABLETS AND 4 GREEN PLACEBO TABLETS); 1 BLISTER (EACH BLISTER IN A LAMINATED SACHET WITH A FLAT CARDBOARD CASE FOR STORING THE BLISTER AND A WEEKLY CALENDAR-STICKER) IN A CARDBOARD PACKAGE SERIES: U15004B -2508UP MANUFACTURER: OJSC GEDEON RICHTER HUNGARY: 31 MAY: UKRAINE
LLC SERVYE UKRAINE

UKRAINE
HS-Code
Products
3004900000
1. MEDICINAL PRODUCTS FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL SALE: TRIPLIXAM® 5 MG/1.25 MG/10 MG FILM-COATED TABLETS 5 MG/1.25 MG/10 MG EACH; 30 PILLS IN A PILL CONTAINER;
3004900000
1. MEDICINAL PRODUCTS FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL SALE: DETRALEX® 1000 MG FILM-COATED TABLETS 1000 MG EACH; 10 TABLETS IN A BLISTER; 3 BLISTERS EACH IN A CARDBOARD BOX SIZE
3004900000
1. MEDICINAL PRODUCTS FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL SALE: TRIPLIXAM® 10MG/2.5MG/5MG FILM-COATED TABLETS 10MG/2.5MG/5MG 30 TABLETS IN A CONTAINER FOR TABLETS 1 CONE EACH
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
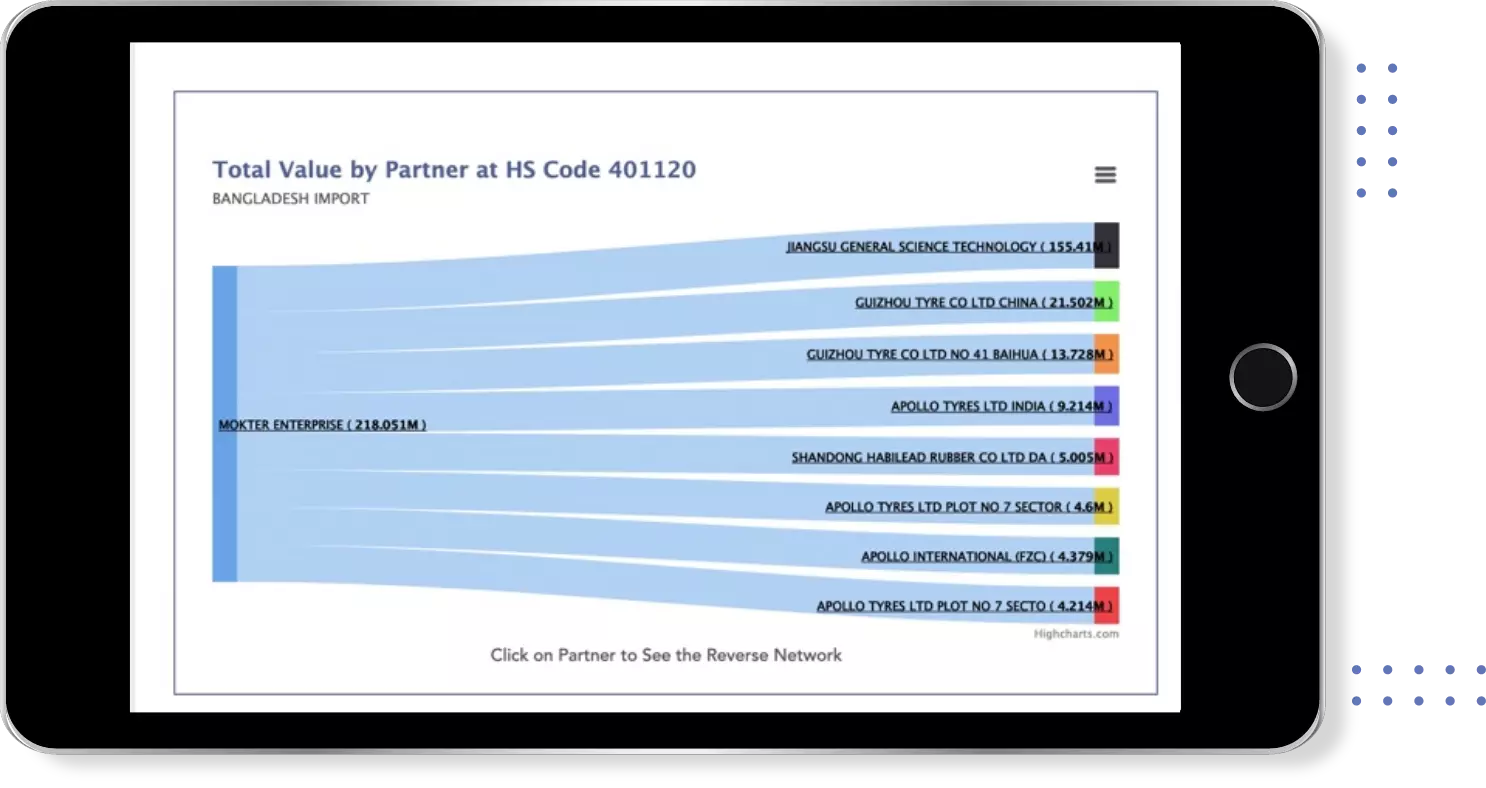
Get most profitable Buyers & Suppliers
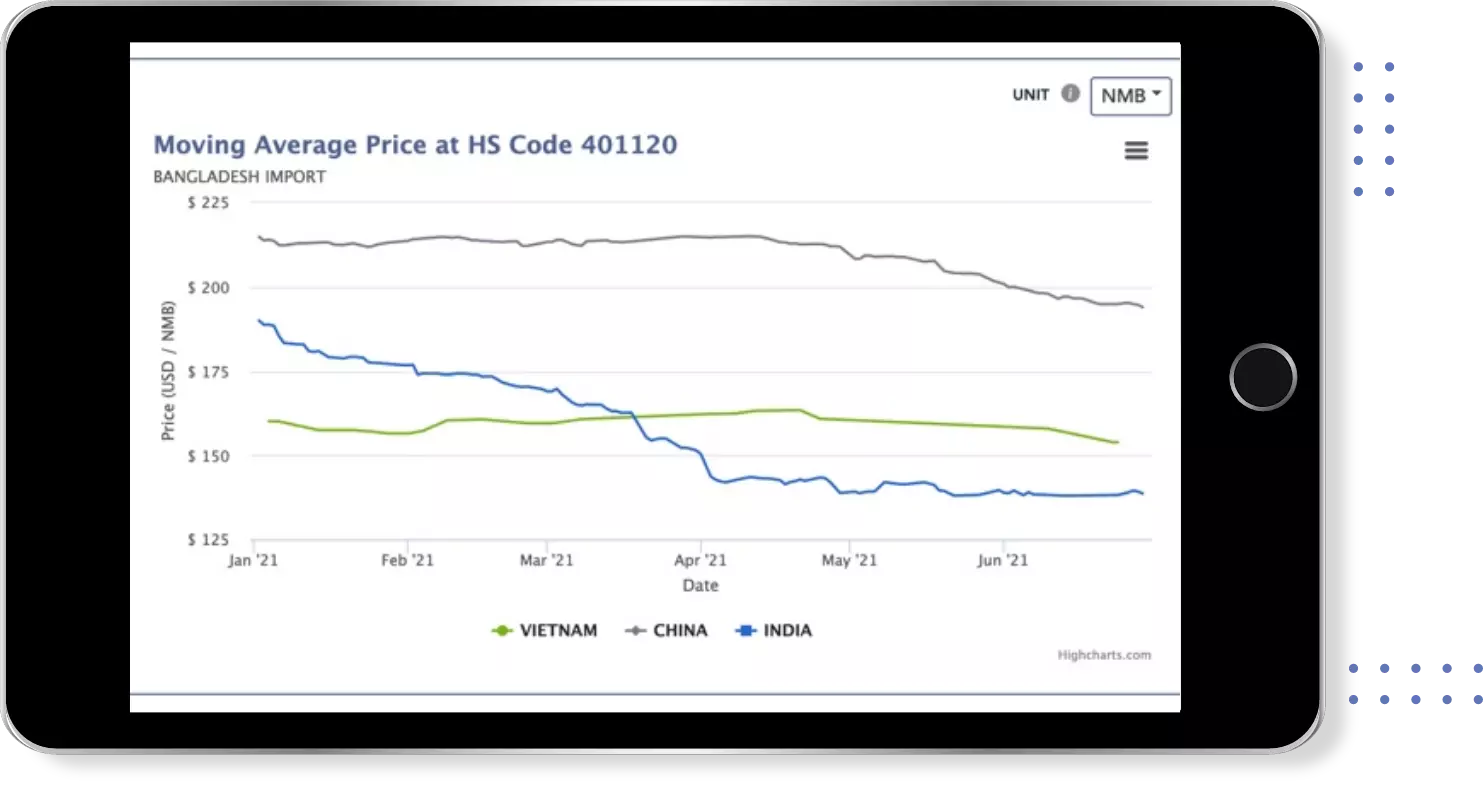
Instant insights on Price , Quantity & Value trends
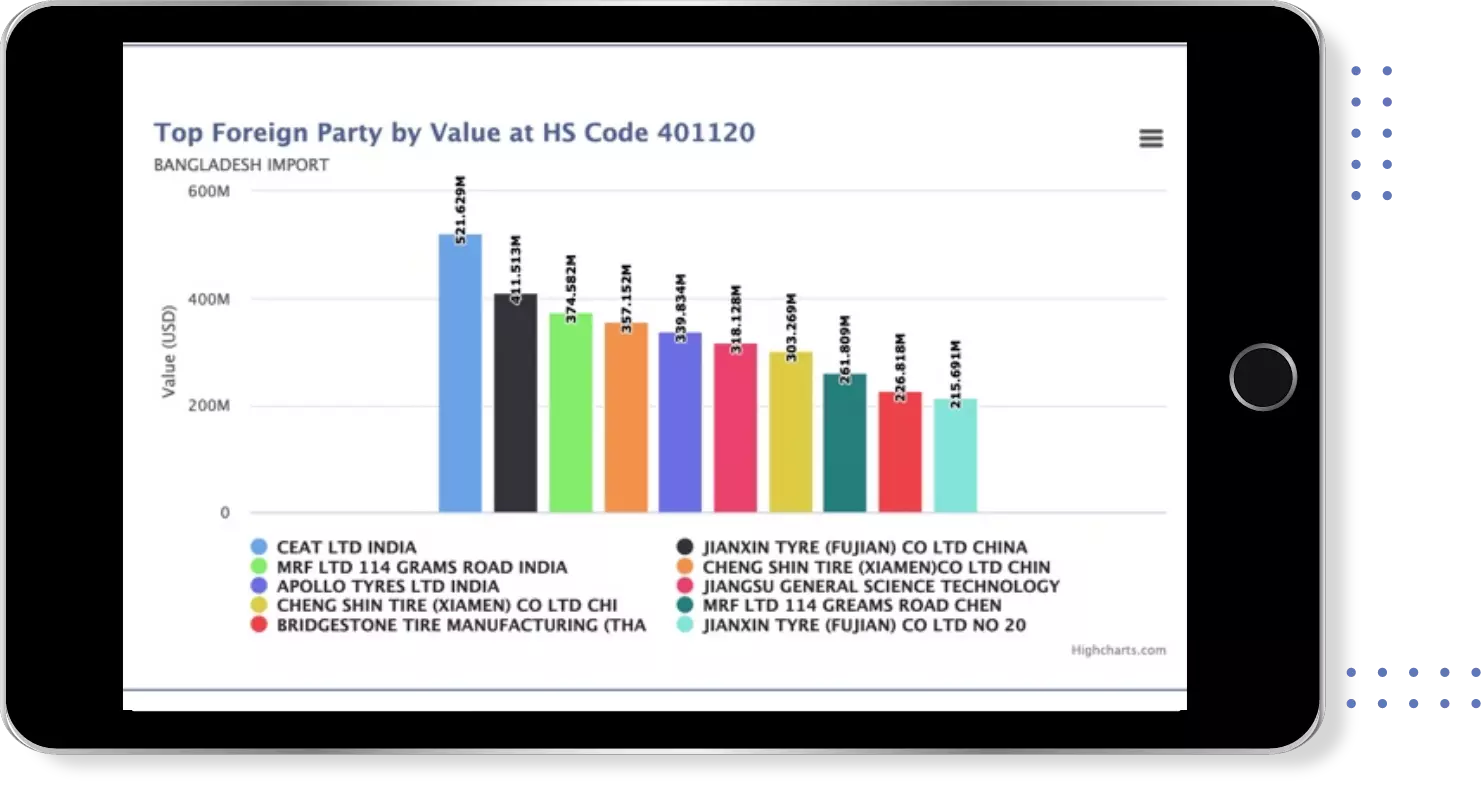
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
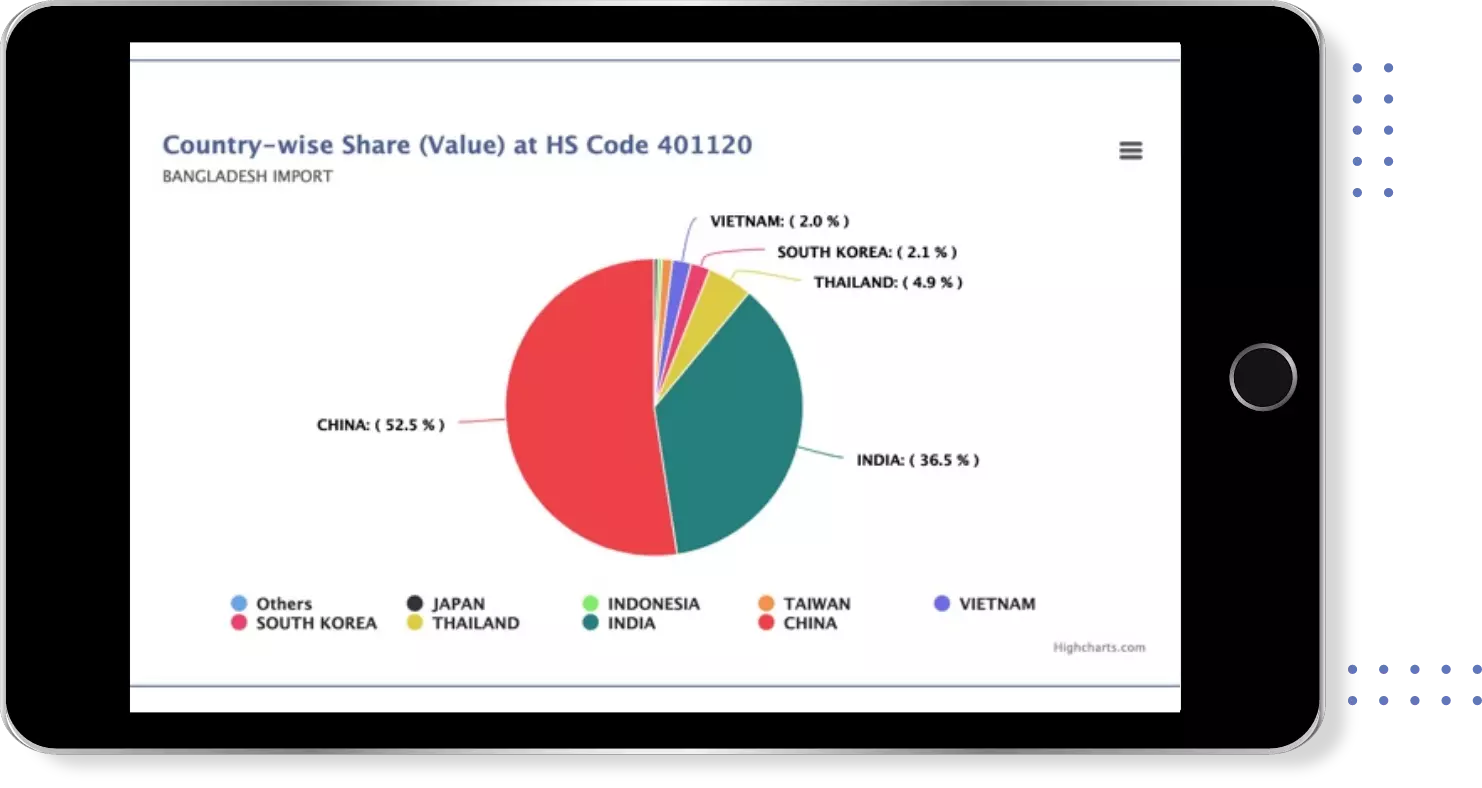
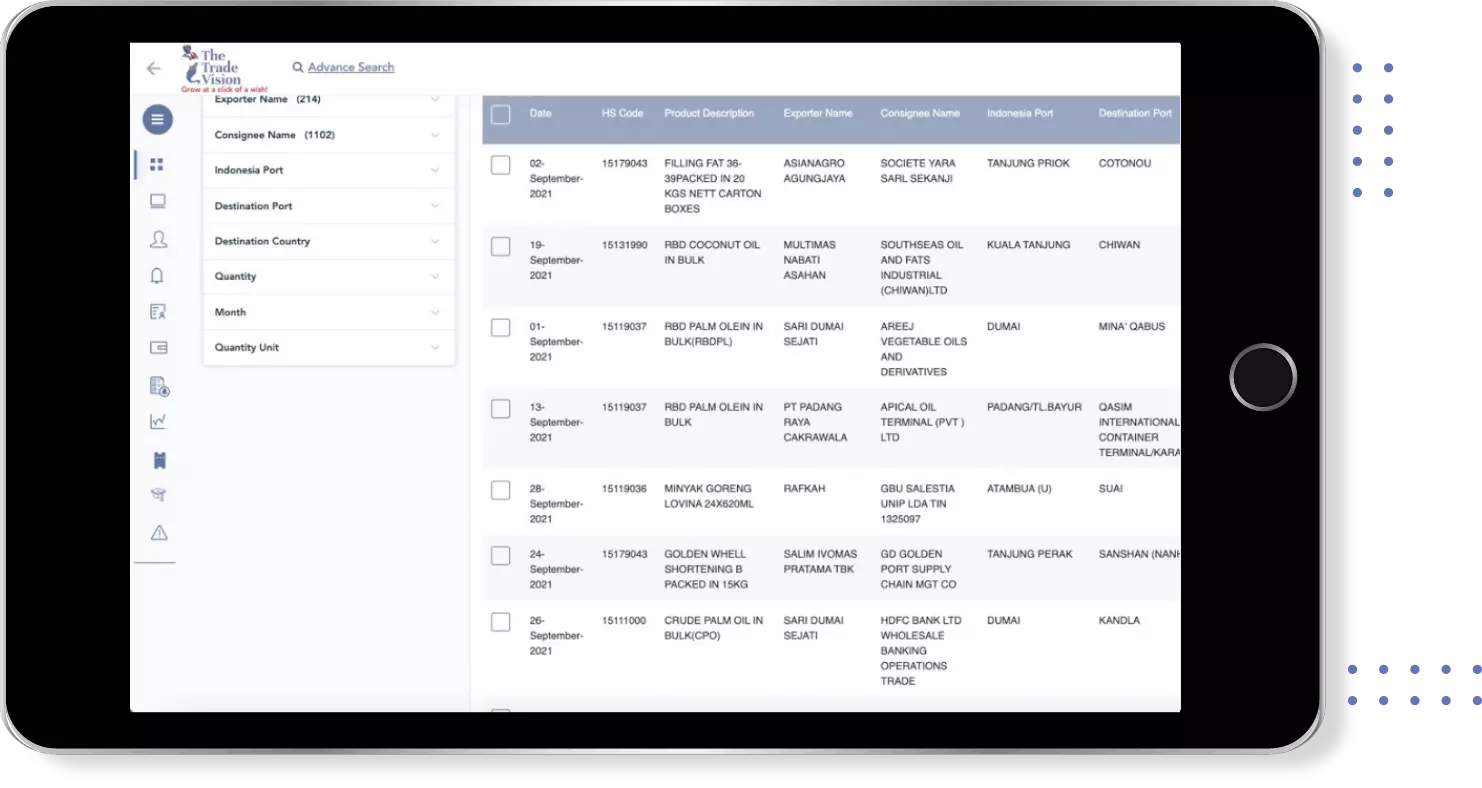
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.