Find verified buyers and sellers of c reactive protein in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- c reactive protein buyers in ukraine
- COD Ukraine
Ukraine Buyers of c reactive protein

UKRAINE
HS-Code
Products
3002109100
1.REAGENTS DIAGNOSTIC OR LABORATORNITEST IMMULITE / IMMULITE 1000 NAVYZNACHENNYA HIGH-SENSITIVITY C-REAKTYVNOHOBILKA (CRP) TEST FOR HASTRYNUTEST FOR RAPID VYZNACHENNYATYREOTROPNOHO HORMONUTEST FOR DETERMINING EOZYNFILNOHOKATIONNOHO BILKATEST FOR VYZNACHENNYAADRENOKORTYKOTROPNOHO HORMONUTEST TO IDENTIFY ANTIBODIES TO THYROGLOBULIN TEST FOR THE DETECTION OF ANTIBODIES DOTYREOHLOBULINU IMMULITE 1000 ANTI-TG AB ( TEST FOR DETERMINING AUTOANTIBODIES DOTYREOHLOBULINU) IMMULITE / IMMULITE 1000 ANTI-TPO AB (TEST DEFINITIONS ANTYTILDO THYROID PEROXIDASE) TEST FOR THE DETECTION OF ANTIBODIES TO THYROID PEROXIDASE TEST FORDETERMINE ANTIBODIES TO THYROID PEROXIDASE TEST FOR FOLATE TESTNA DEFINITIONS INTACT PARATHYROID HORMONE TEST FOR DETERMINATION OF VITAMIN B1 2TEST ONDEFINITION HIGH-SENSITIVITY C-REACTIVE PROTEIN TEST IMMULITE / IMMULITE1 000 HOMOCYSTEINE ON DETERMINING HOMOCYSTEINE ??TEST DEFINITIONS FOR DETERMINING HOMOTSYSTEYINUTEST INSULIN-LIKE GROWTH FACTOR SOLVENT FOR CW TEST NAVYZNACHENNYA
3002109100
1.DIAGNOSTIC OR LABORATORY REAGENTSIMMUNOASSAY FOR QUANTIFICATION OF PROGESTERONETEST FOR QUANTIFICATION OF PROLACTINTHE TEST FOR QUANTIFICATION OF TOTAL TRIIODOTHYRONINE / ELECSYS T3IMMUNOTEST FOR QUANTIFICATION OF HUMAN TSH TEST FOR QUANTIFICATION OF PANCREATIC ALPHA-AMYLASE / ALPHA-AMYLASE EPS PANCREATIC (AMY-P) IMMUNOASSAY FOR DETERMINATION OF IGM AND IGG ANTIBODIES TO NUCLEAR ANTIGEN OF HEPATITIS B VIRUS 100 TESTS IMMUNOTEST FOR QUANTIFICATION OF HUMAN ANTIBODIES TO VERTIGOGLOBU O / ANTISTREPTOLYSIN O TEST FOR QUANTIFICATION OF CANCER ANTIGEN 15-3 / CA 15-3 II CANCER ANTIGEN 15-3 TEST FOR QUANTIFICATION OF C-REACTIVE PROTEIN TEST D
3002150000
1.DIAGNOSTIC OR LABORATORY REAGENTSTEST FOR QUANTIFICATION OF ALBUMIN / ALBUMIN IMMUNOTURBIDIMETRIC GEN.2 (ALB-T) TEST FOR QUANTIFICATION OF ALPHA-AMYLASE OF THE PANCREAS / ALPHA-AMYLASEEPS PANCREATIC (AMY-P) IMMUNOTEST FOR QUANTIFICATION OF VERTEBRATE ANTIBODIES TO ANTIBODIES TO FOR THE QUANTITATIVE DETERMINATION OF ANTIBODIES TO THYROPEROXIDASE VER. 3TEST FOR QUANTITATIVE IMMUNOLOGICAL DETERMINATION OF HUMAN ANTISTREPTOLYSINO / ANT ISTREPTOLYSIN O CALIBRATION KIT FOR TEST CA 19-9 TEST FOR QUANTIFICATION OF C-REACTIVE PROTEIN IMMUNOASSAY FOR IVD QUANTIFICATION OF FERRITIN IN PLASMA AND SERUM FOR ELECISM SYSTEMS TRIIODOTHIONINE IMMUNOTEST FOR QUANTIFICATION OF UNBOUND THYROXINE GEN. III TEST FOR QUANTIFICATION OF HEMOGLOBIN IMMUNOTEST FOR QUANTIFICATION OF INSULIN IMMUNOTEST FOR QUANTIFICATION OF LUTEINIZING HORMONE TEST FOR QUANTIFICATION OF PROLACTIN TEST FOR QUANTIFICATION OF RHEUMATOID ARTHRITIS TEST FOR QUANTIFICATION OF BLOOD PLASMA CONCENTRATION TEST FOR THE DETERMINATION OF TSH TEST FOR THE QUANTITATIVE DETERMINATION OF C-REACTIVE PROTEIN (CRP) 4 GEN. COBAS C 311/5 01/502 INTEGRA ROCHE DIAGNOSTICS GMBHGERMANY.

UKRAINE
HS-Code
Products
3002190000
1. DIAGNOSTIC SETS REAGENT IN THE COMPOSITION OF ANTIBODIES CONTAINING BOTH ANIMAL AND HUMAN ORIGIN CALIBRATORS AND CONTROL SUBSTANCE. MANUFACTURER INTENDED FOR USE IN VITRO DIAGNOSTIC IN THE STUDY OF BLOOD SAMPLES MAN. WORKING PRINCIPLE SETS BASED ON THE METHOD ELISA ON MICROPLATES AND REACTIONS ANTIGEN-ANTIBODY: 1125-300A ANTI-TPO - ANTI-THYROID PEROXIDASE ELISA - ELISA REAGENTS SET FOR DETERMINATION OF ANTI-TPO - ANTIBODIES TO TPO. SHT. 5 2425-300A INSULIN - ELISA REAGENTS SET FOR DETERMINATION OF INSULIN ; PCS-2; 3125-300A HS-CRP - HIGH SENSITIVITY CRP ELISA - ELISA KIT FOR DETERMINATION OF CRP - CONCENTRATIONS OF C-REACTIVE PROTEIN. PCS-1; 3825-300A CTNI-TROPIN ELISA REAGENTS FOR DETERMINATION OF CTNI - TROPONINE-I. PCS.-3; 7525-300A FOLATE ELISA-SET OF ELISA REAGENTS FOR DETERMINATION OF FOLAT. PCS-1 - ELITA AND 12IT DETERMINATION OF VITAMIN B-12 CONCENTRATION PCS-1; 9425-30 0A - 25 (OH) VITAMIN D TOTAL DIRECT (VIT D-DIRECT) TEST SYSTEM - ELISA KIT FOR DETERMINATION OF CONCENTRATIONS OF 25-ON TOTAL VITAMIN D. PCS-35; MULTILIGAND CONTROL - TRI-LEVEL PCS-3;
3002190000
1. SETS DIAGNOSTIC REAGENTS KITS RAPID TESTS WHICH IN ITS COMPOSITION CONTAINS ANTIBODIES OF ANIMAL AND HUMAN ORIGIN CALIBRATORS AND CONTROL SUBSTANCE. MANUFACTURER INTENDED FOR USE IN VITRO DIAGNOSTIC IN THE STUDY SAMPLES OF BLOOD OR URINE HUMAN: A00509 ASO (TURB.) - A SET OF REAGENTS ANTISTREPTOLYSIN (TURBIDYMETRYCHNYY). SHT.-6; A00505 A-2 MACROGLOBULIN (TURB.) - SET REAGENTS ALPHA-2 MICROGLOBULIN (TURBIDYMETRYCHNYY) SHT. 1; A00532 HAPTOGLOBIN (TURB. ) - A SET OF REAGENTS HAPTOGLOBIN (TURBIDYMETRYCHNYY) SHT. 1; A00800 PROTEIN CONTROL - CONTROL DETERMINATION OF PROTEIN SERUM. SHT. 1; A00806 APO A1 / A2 / B CONTROL (LYO) - TESTS SERUM DEFINITIONS APOLIPOPROTEIN A1 / A2 / B-1 SHT.; Y04604 HBA1C DIRECT CONTROL-SET - SET OF CONTROL SERA FOR CERTAIN HLIKOZOVANOHO HEMOGLOBIN (DIRECT METHOD) SHT. 1; A00519 CRP - DIAGNOSTIC REAGENTS FOR THE QUANTITATIVE DETERMINATION OF C-REACTIVE PROTEIN (SRB) B HUMAN SERUM BY TURBIDIMETRIC ANALYSIS. PCS-1; A00590 PROTEIN CONTROL - PROTEIN CONTROL FOR PROTEIN DETERMINATION PCS-3; A00506 APO A1 (TURB.) - SET OF REAGENTS FOR DETERMINATION OF APOLIPOPROTEIN A1 (TURBIDIMETRIC) PCS-1; A00506 APO A1 (TURB.) - SET OF REAGENTS FOR DETERMINATION OF APOLIPOPROTEIN A1 (TURBIDIMETRIC) PCS-1; A00800 PROTEIN CONTROL - PROTEIN CONTROL FOR PROTEIN DETERMINATION. PCS-1; D96568 ALPHA AMYLASE PANCR. ETG7PNP - SET OF REAGENTS FOR DETERMINATION OF ALPHA AMYLASE PANCREATIC ETG7PNP PCS-5; Z02550CE MTD-DIPSTICK - SET OF TEST STRIPS FOR DETERMINATION OF METHADONE (50 STRIPS IN THE SET). PCS-1; Y08310SV AMMONIA STANDARD 100 ΜM - STANDARD FOR DETERMINATION OF AMMONIA. PC.-2; Y08330 AMMONIA CTRL.-SET (2 LEVELS) - CONTROL SERUM FOR DETERMINATION OF AMMONIA (2 LEVELS). PC.-2; A00531 CERULOPLASMIN (TURB.) - SET OF REAGENTS FOR DETERMINATION OF CERULOPLASMIN (TURBIDIMETRIC) PCS-1; 507163SV COPPER STANDARD - STANDARD FOR DETERMINATION OF COPPER PCS-1; 507140 COPPER 3 5-DIBR-PAESA - SET OF REAGENTS FOR DETERMINATION OF COPPER 3 5-DIBR-PAESA PCS-1; 798001 URINE COMBY CONTROL PN - LIQUID URINARY CONTROL COMBY PN IS INTENDED FOR EVALUATION OF VISUAL INSPECTION AND RESULTS OF URINE ANALYSIS OBTAINED. PCS-5; 909680 SODIUM STANDARD SET (2 LEVELS) - SET OF STANDARDS FOR DETERMINATION OF SODIUM (2 LEVELS) PCS-1; 910190 POTASSIUM CONTROL SET (2 LEV.) - SET OF CONTROL SERUMS FOR DETERMINATION OF POTASSIUM (2 LEVELS) PCS-1; A00808 MICROALBUMIN CONTROL - CONTROL SERUM FOR DETERMINATION OF MICROALBUMIN PCS-1; G04010C URINE STRIP 10C - SET OF URINE STRIPS 10C (100 STRIPS IN A SET) PCS-100; Y04602 HBA1C DIRECT - SET OF REAGENTS FOR QUANTITATIVE DETERMINATION OF GLYCOSYLATED HEMOGLOBIN (DIRECT METHOD). PC.-4; Y04604 HBA1C DIRECT CONTROL-SET - SET OF CONTROL SERUMS FOR DETERMINATION OF GLYCOSED HEMOGLOBIN (DIRECT METHOD) PCS-2; Z06620CE MONONUCLEOSIS CASSETTE - A SET OF TEST CASSETTES FOR YOU
3002190000
1. DIAGNOSTIC SETS REAGENT IN THE COMPOSITION OF ANTIBODIES CONTAINING BOTH ANIMAL AND HUMAN ORIGIN CALIBRATORS AND CONTROL SUBSTANCE. MANUFACTURER INTENDED FOR USE IN VITRO DIAGNOSTIC IN THE STUDY OF BLOOD SAMPLES MAN. WORKING PRINCIPLE SETS BASED ON THE METHOD ELISA ON MICROPLATES AND REACTIONS ANTIGEN-ANTIBODY: 2425-300A INSULIN - KIT OF ELISA REAGENTS FOR DETERMINATION OF INSULIN ; PC.-2; 325-300B TSH - THYROTROPIN ELISA - KIT OF 1 REAGENT-3 REAGENT. U-E3 - UNCONJUGATED ESTRIOL ELISA-ELISA REAGENTS SET FOR DETERMINING U-E3 - UNCONJUGATED ESTRIOL. SHT. 2; 6025-300A RAPID TSH ELISA - SET PRYZNACHNYY FOR THE QUANTITATIVE DETERMINATION OF THE CONCENTRATION THYROID STIMULATING HORMONE IN HUMAN SERUM BY ENZYME ANALYSIS. PCS.-7; 7625-300A - VITAMIN B-12 ELISA - KIT OF ELISA REAGENTS TO DETERMINE THE CONCENTRATION OF VITAMIN B-12. PCS-2; 8025-300B THYROID TANST: VA3 - THYROXINE TRIIODTHYRONINE THYROID STIMULATING HORMONE ELISA - ELISA KIT FOR DETERMINATION OF THYROID PANEL (VAST): T3 T4 TSH - THYROXIN TRIODIOTHYRONYONHINOGINO PCS-1; 8325-300B FERTILITY PANEL (VAST): HCG SPRL LH FSH - HUMAN CHORIONIC GONADOTROPIN HUMAN PROLACTIN HUMAN LUTEINIZING HORMONE FOLLICLE STIMULATING HORMONE ELISA FSH - HORIONIC HONODOTROPIN OF HUMAN PROLACTIN LUTEINIZING HORMONE FOLLICLE-STIMULATING HORMONE 192 DEFINITIONS. PCS-1; 9025-300A PTH INTACT - PARATHYROID HORMONE INTACT (2ND GENERATION) ELISA. PCS-12; 9425-300A - 25 (OH) VITAMIN D TOTAL DIRECT (VIT D-DIRECT) TEST SYSTEM - ELISA KIT FOR DETERMINATION OF 25-ON TOTAL VITAMIN CONCENTRATIONS PCS-50; 3125-300A HS-CRP - HIGH SENSITIVITY CRP ELISA - ELISA KIT FOR DETERMINATION OF CRP - CONCENTRATION OF C-REACTIVE PROTEIN. PC.-6; 325-300 TSH - THYROTROPIN - SET OF REAGENTS FOR DETERMINATION OF TSH - THYROTROPIC HORMONE PCS-5; 9425-300A - 25 (OH) VITAMIN D TOTAL DIRECT (VIT D-DIRECT) TEST SYSTEM - ELISA KIT FOR DETERMINATION OF CONCENTRATIONS OF 25-ON TOTAL VITAMIN D. PCS-40; 9725-300A AMH - ANTI-MULLERIAN HORMONE - A SET OF QUANTITATIVE DETERMINATION OF THE CONCENTRATION OF ANTIMULLER HORMONE IN SERUM OR PLASMODIS OF BLOOD. PCS-1;

UKRAINE
HS-Code
Products
3402209000
1. REAGENT FOR CLEANING THE LIQUID TRACT IN BIOCHEMICAL ANALYZERS FOR IN VITRO DIAGNOSTICS SERIES COBAS: PRECLEAN M IS USED TO REMOVE POTENTIALLY HAZARDOUS SUBSTANCES BEFORE THE APPEARANCE OF SIGNALS OF THE LAST STEPWISE ANALYTICAL PROCEDURE. 5 X 600 ML WASHING SOLUTION. PHOSPHATE BUFFER 10 MMOL / L; SODIUM CHLORIDE 20 M DETERGENT - 0.1% PRESERVATIVE PH 7.0 WASH SOLUTION PRECLEAN M LOT: 54110901 - 7 SET. WASH SOLUTION PRECLEAN M LOT: 5411 0901 - 1 SET. WASH SOLUTION PRECLEAN M LOT: 54110801 - 20 SET. FLUSHING SOLUTION PRECLEAN M LOT: 54110901 - 9 SET.SYSTEM SOLUTION FOR CLEANING THE NEEDLES OF THE SAMPLE INTAKES; PACKAGING -PLASTIC BOTTLE WITH A CAPACITY OF 70 ML .; DETERGENT - 1%; DOES NOT CONTAIN ABRASIVE POWDERS: CLEANING SOLUTION PROBEWASH M LOT: 1 997250. SOLUTIONPROBEWASH M LOT: 53972 501 - 2 PCS DILUENT UNIVERSAL IS USED FOR DILUTION OF THE SAMPLE WHEN PERFORMING TESTS USING ELECSYS REAGENTS CONTENTS: PROTEIN BASE PRESERVATIVE - 0.1% PACKAGING - 2 VIALS 36 ML EACH SOLVENT UNIVERSAL / UNIVERSAL DILUE NT LOT: 52370101 - 10 SETS. SMS WASHING SOLUTION FOR REAGENT SAMPLES AND / OR REACTION CHAMBERS IN ROCHE / HITACHICOBAS SYSTEMS WITH A 50 ML CASSETTE CONTAINING HCL 200 ?MOL / L. SMS WASHING SOLUTION FOR TEST REAGENT AND / OR REACTION CHAMBERS IN ROCHE / HITACH AND COBAS SYSTEMS C LOT: 54933701- 23 PCS. FLUSHING SOLUTION FOR CLEANING THE SYSTEM TUBES OF THE BIOCHEMICAL ANALYZER COBAS C6000; PACKING - A PLASTIC BOTTLE OF 58 7 ML .; DETERGENT 1% DOES NOT CONTAIN ABRASIVE POWDERS: WASHING SOLUTION OF NAOHD FOR SAMPLES OF REAGENT AND / OR REACTION CHAMBERS IN ROCHE / HITACH AND COBAS SYSTEMS C LOT: 54933501 - 72 PCS. SOLVENT FOR DILUTION OF THE SAMPLE WHEN PERFORMING TESTS USING REAGENTS ON SYSTEMS COBAS C (50 ML) TO THE BIOCHEMICAL ANALYZER COBAS C6000; PACKING - A PLASTIC BOTTLE OF 39 2 ML; DETERGENT 1% DOES NOT CONTAIN ABRASIVE POWDERS: SOLVENT FOR DILUTION OF THE SAMPLE WHEN PERFORMING TESTS USING REAGENTS ON SYSTEMS COBAS C (50 ML) LOT: 54130401 - 10 PCS. ISE SOLVENT IS INTENDED FOR USE IN CALIBRATION-SELECTIVE ELECTRODES ON ROCHE / HITACHI ANALYZERS AND COBAS C ANALYZERS. 5 PLASTIC VIALS EACH WITH 300 MLISE SOLVENT. P REACTIVE COMPONENTS: NA + K + CL NON-REACTIVE COMPONENTS: STABILIZER. SOLUTION FOR WASHING PROBES FOR SAMPLES ON ROCHE / HITACHI COBAS SYSTEMS C. CONTAINS NAOH 1 MMOL / LFLACON WITH A CAPACITY OF 59 ML. FLUSHING SOLUTION FOR SAMPLING PROBE LOT: 54329501 - 2 SETS. 9% NACL SOLVENT IS USED TO DILUTE THE SAMPLES IN COMBINATION WITH REAGENTS FOR QUANTIFICATION IN THE COBAS SYSTEM C 111. NACL: 9% PLASTIC VIAL (4 X 12 ML) SOLVENT FOR SAMPLE DILUTION WHEN PERFORMING TESTS USING REAGENTS ON COBAS C 111 SYSTEMS (4 X 12 ML) LOT: 53799201 - 3 SETS. WASHING SOLUTION FOR ADDITIONAL WASHING PROBE CYCLES FOR REAGENT / SAMPLE ON THE COBAS ANALYZER C 111. PLASTIC BOTTLE WITH A CAPACITY OF (4 X
3002150000
1. MEDICAL DEVICES INTENDED BY THE MANUFACTURER FOR IN VITRO USE FOR LABORATORY RESEARCH TO OBTAIN INFORMATION ON PHYSIOLOGICAL OR PATHOLOGICAL CONDITIONS: COMPLEX LABORATORY IMMUNOLOGICAL REAGENTS OF COMBINED ORIGIN OBTAINED USING BIOTECHNOLOGICAL PROCESSES: COMPLEX REAGENTS (ANALYTICAL) INTENDED FOR BLOOD TESTS: IMMUNOTEST HE4LOT: 53318001 - 1 AMOUNT TEST FOR THE QUANTITATIVE DETERMINATION OF C-PEPTIDE / C-PEPTIDELOT: 56380502 - 1 AMOUNT TEST FOR THE QUANTITATIVE DETERMINATION OF PROLACTIN / PROLACTINII LOT: 58163601 - 77 AMOUNT IMMUNOTEST FOR THE QUANTITATIVE DETERMINATION OF ANTIGENNUCLEOTIDE CARCINOMA / SCC LOT: 55183602 - 1 PIECE TEST FOR QUANTITATIVE DETERMINATION OF C-REACTIVE PROTEIN (CRP) 4 GEN. COBA SC 111 LOT: 59349901 - 20 SETS. IMMUNOTEST FOR QUANTITATIVE DETERMINATION OF FOLLICLE STIMULATING HORMONE / ELECSYS FSH LOT : 57175702 - 2 AMOUNT TEST FOR QUANTITATIVE DETERMINATION OF TRANSFERRIN / TRSF TINA-QUANTTRANSFERRIN VER.2 LOT: 59449201 - 1 AMOUNT
3402209000
1. REAGENT FOR CLEANING THE LIQUID TRACT IN BIOCHEMICAL ANALYZERS FOR IN VITRO DIAGNOSTICS OF THE COBAS SERIES: DILUENT UNIVERSAL IS USED TO DILUTE THE SAMPLE WHEN PERFORMING TESTS USING ELECSYS REAGENTS. CONTENT: PROTEIN BASE; PRESERVATIVE LESS THAN OR EQUAL TO 0.1% PACKAGING - 2 VIALS 36ML EACH: UNIVERSAL DILUENT LOT: 52370101 - 15 SETS. SOLVENT FOR DILUTION OF SAMPLES WHEN PERFORMING TESTS USING REAGENT SYSTEMS COBAS C (50 ML) TO THE BIOCHEMICAL ANALYZER COBAS C6000; PACKAGING - PLASTIC BOTTLE 39.2 ML; DETERGENT 1%; DOES NOT CONTAIN ABRASIVE POWDERS: SOLVENT FOR DILUTION OF THE SAMPLE WHEN PERFORMING TESTS USING REAGENTS ON THE SYSTEMS COBAS C (50 ML) LOT: 54130401 - 2 PCS. ISE DEPROTEINIZER IS A CLEANING AGENT INTENDED FOR USE WITH MODULES AND ISBI COBASINTEGRA AND COBAS C 111 ICE FOR CLEANING ION-SELECTIVE ELECTRODES CONTAINS SODIUM HYPOCHLORITE (NAOCL): ABOUT 1.2% PACKAGING - PLASTIC BOTTLE (2 X 11 ML) : ISE CLEANING SOLUTION FOR COBAS SYSTEM C 111 LOT: 51202701 - 6 PER SIDE TEST TO DETERMINE THE ACCURACY AND PRECISION OF THE SAMPLE AND REAGENT KIT USING A PIPETTE WITH DYE SOLUTION AND ISOTONIC SODIUM CHLORIDE SOLUTION X SYSTEM ROCHE / HITACHI COBAS C. ; PACKAGING -P ELASTIC CASSETTE CONTAINING SODIUM CHLORIDE: 1.54 MOL / L: TEST TO DETERMINE THE ACCURACY AND PRECISION OF THE SETSAMPLE AND REAGENT / INSTRUMENT CHECK LOT: 51153701 - 1 SET. DILUENTUNIVERSAL 2 IS USED TO DILUTE THE TEST WHEN PERFORMING TESTS WITH THE USE OF REAGENTS ELECSYS. CONTENTS: PROTEIN BASE; PRESERVATIVE LESS THAN 0.1% PACKAGING - 2 VIALS 36 ML EACH. P SOLVENT UNIVERSAL 2 / UNIVERSALDILU ENT II LOT: 48859501 - 3 SETS. SYSTEM SOLUTION FOR CLEANING THE NEEDLES OF THE SAMPLER; PACKAGING-PLASTIC BOTTLE WITH A CAPACITY OF 5X100ML .; DETERGENT - 1%; NO ABRASIVE POWDERS: CLEANING SOLUTION ISE CLEANING SOLUTION / ELECSYSSYSCLEAN L OT: 53145301 - 3 SETS. BLANKCELL IS USED TO CALIBRATE ITS TOOLS BY ROCHE TECHNICAL SERVICE. THE REAGENT IS INTENDED FOR USE BY TECHNICAL PERSONNEL FOR THE PURPOSE OF: INITIAL CALIBRATION OF BLANKCELL ADJUSTMENT OF A PHOTOELECTRONIC MULTIPLIER OF HIGH VOLTAGE (P MT HV). TESTING OF ARTIFICIAL MEDIA. REACTIVE WORKING SOLUTIONS: 2 X 50 ML FOR 225 MEASUREMENTS (ACCORDING TO 75 INITIAL CALIBRATIONS OF BLANKCELL) BCR1 PHOSPHATE BUFFER 300 MMOL / L TRIPROPYLAMINE 180 MOL / L SURFACTANT LESS THAN OR EQUAL TO 0.1% PRESERVATIVE PH 6.8. BCR2 PHOSPHATE BUFFER 300 MMOL / L TRIPROPYLAMINE 180 MMOL / L TRIS (2 2'-BIPYRIDYL) RUTHENIUM (II) -COMPLEX10 NMOL / L SURFACTANT LESS THAN OR EQUAL TO 0.1% PRESERVATIVE PH 6 8: SERVICE CALIBRATION SOLUTION ELECSYS COBAS E LOT: 50 389701 - 2 SET. UNIVERSAL SOLVENT DILUENT UNIVERSA L IS USED AS A REFERENCE SOLVENT IN COMBINATION WITH REAGENTS FOR ANALYSIS ELECSYS. PROTEIN BASE; PRESERVATIVE LESS THAN OR EQUAL TO 0.1% PACKAGING VIALS 2 X 16 ML. SOLVENT UNIVERSAL DILUENT LOT: 45659701 - 3 SETS. PACKED

UKRAINE
HS-Code
Products
3002190000
1.MEDICAL DEVICE FOR IN VITRO DIAGNOSTICS (CLINICAL LABORATORY DIAGNOSTICS) FOR EXAMINATION OF BLOOD AND URINE SAMPLES IN LABORATORIES NOT ON A SUBSTRATE IN THE FORM OF PENS (FELT-TIP PENS) FOR ALLERGENS AND PLASTIC CONTAINERS WITH LIQUID. IMMUNOLOGICAL PRODUCTS FOR SIEMENS ANALYZERS FOR CLINICAL AND LABORATORY CHEMICAL AND IMMUNOBIOLOGICAL STUDIES OBTAINED USING BIOTECHNOLOGICAL PROCESSES THAT DO NOT CONTAIN ANIMAL BLOOD DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS DOES NOT CONTAIN GMOS: REAGENT ADVIA CHEMISTRY FOR THE DETERMINATION OF C-REACTIVE PROTEIN 2 (ADVIA CHEMISTRY C-REACTIVE PROTEIN 2 REAGENTS) ART.06522059- 60PCS; CYSTATIN C_2 REAGENT ADVIA CHEMISRTY (CYSC_2) (ADVIA CHEMISTRY CYSTATIN C_2 REAGENT (CYSC_2)) ART.10734503-1PCS; COUNTRY OF MANUFACTURE GBSIEMENS BRANDMANUFACTURER RANDOX LABORATORIES LTD. (UK) FOR SIEMENS HEALTHCARE DIAGNOSTICS INC. (USA)
3002109900
1. MEDICAL DEVICE FOR IN VITRO DIAGNOSTICS (CLINICAL LABORATORY DIAGNOSTICS) FOR EXAMINATION OF BLOOD AND URINE SAMPLES IN LABORATORIES NOT ON A SUBSTRATE IN THE FORM OF PENS (FELT-TIP PENS) FOR ALLERGENS AND PLASTIC CONTAINERS WITH LIQUID. IMMUNOLOGICAL PRODUCTS FOR SIEMENS ANALYZERS FOR CLINICAL AND LABORATORY CHEMICAL AND IMMUNOBIOLOGICAL STUDIES OBTAINED USING BIOTECHNOLOGICAL PROCESSES THAT DO NOT CONTAIN ANIMAL BLOOD DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS DO NOT CONTAIN GMOS: ADVIA REAGENT FOR THE DETERMINATION OF APOLIPOPROTEIN A-1 (APO-A1) (ADVIA CHEMISTRY APOLIPOPROTEIN A1 REAGENT) ART.3050910-4PCS; ADVIA REAGENT FOR DETERMINATION OF APOLIPOPROTEIN B (APO-B) (ADVIA CHEMISTRY APOLIPOPROTEIN B REAGENT) ART.3055068-2PCS; ADVIA REAGENT FOR DETERMINATION OF CHOLESTEROL 2 (ADVIA CHEMISTRY CHOLESTEROL 2) .10376501-3PCS; ADVIA REAGENT FOR DETERMINATION FOR DETERMINATION OF C-REACTIVE PROTEIN 2 (ADVIA CHEMISTRY C-REACTIVE PROTEIN 2 REAGENTS) ART.6522059-7PCS; ADVIA REAGENT FOR DETERMINATION OF HAPTOGLOBIN (3 X 18
3002190000
1.MEDICAL DEVICE FOR IN VITRO DIAGNOSTICS (CLINICAL LABORATORY DIAGNOSTICS) FOR EXAMINATION OF BLOOD AND URINE SAMPLES IN LABORATORIES NOT ON A SUBSTRATE IN THE FORM OF PENS (FELT-TIP PENS) FOR ALLERGENS AND PLASTIC CONTAINERS WITH LIQUID. IMMUNOLOGICAL PRODUCTS FOR SIEMENS ANALYZERS FOR CLINICAL AND LABORATORY CHEMICAL AND IMMUNOBIOLOGICAL STUDIES OBTAINED USING BIOTECHNOLOGICAL PROCESSES THAT DO NOT CONTAIN ANIMAL BLOOD DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS DOES NOT CONTAIN GMOS: REAGENTS ADVIA CHEMISTRY FOR THE DETERMINATION OF AMYLASE (7X175 TESTS) (ADVIA CHEMISTRY AMYLASE REAGENT (7X175 TESTS)) - ART.07498 1PACK .; ADVIA CHEMISTRY REAGENT FOR DETERMINATION OF CHOLESTEROL 2 (ADVIA CHEMISTRY CHOLESTEROL 2) ART.10376501-3PCS; ADVIA CHEMISTRY REAGENTS FOR DETERMINATION OF C-REACTIVE PROTEIN 2 (ADVIA CHEMISTRY C-REACTIVE PROTEIN 2 REAGENTS) ART.06522059-24PCS; ADVIA REAGENT CHEMISTRY FOR DETERMINATION OF IMMUNOGLOBULIN A_2 (IGA_2) (ADVIA CHEMISTRY IMMUNOGLOBULIN A_2 REAGENT) ART.02194102-1PCS; VIA CHEMISRTY FOR DETERMINATION OF LP LACTATE DEHYDROGENASE (7X274 TESTS) (ADVIA CHEMISTRY LACTATE DEHYDROGENASE LP REAGENTS (7 X 274 TESTS)) ART.07502115-2PACK .; ADVIA CHEMISTRY REAGENT FOR DETERMINATION OF LIPASE IN BLOOD (ADVIA CHEMISTRY LIPASE REAGENT) ART.01984894-1PCS; ADVIA CHEMISTRY REAGENTS FOR DETERMINATION OF MICROALBUMIN 2 (ΜALB_2) (ADVIA CHEMISTRY MICROALBUMIN_2 REAGENT) ART.03051194-2PCS; ADVIA CHEMISRTY REAGENT FOR DETERMINATION OF TRANSFERRIN (ADVIA CHEMISTRY TRANSFERRIN REAGENTS) ART.03059160-1PCS; ADVIA CHEMISTRY REAGENT FOR DETERMINATION OF IRON 2 (7 X 145 TESTS) (ADVIA CHEMISTRY IRON 2 (7 X 145 TESTS)) ART.02194838-1PACK .; ADVIA CHEMISRTY REAGENTS FOR DETERMINATION OF HIGHLY SENSITIVE C-REACTIVE PROTEIN (HSCRP) (ADVIA CHEMISTRY CARDIOPHASE HIGH SENSITIVITY C-REACTIVE PROTEIN REAGENTS) ART.06837459-1PCS; ADVIA CHEMISTRY REAGENT FOR DETERMINATION OF FRUCTOSAMINE (ADVIA CHEMISTRY FRUCTOSAMINE REAGENT) ART.04862501-1PCS; ADVIA CHEMISRTY REAGENT FOR CYSTATIN C (ADVIA CHEMISTRY CYSTATIN C REAGENT) ART.10734503-1PCS; ADVIA CHEMISTRY REAGENTS FOR DEFINITION OF COMPLEMENT C4 (ADVIA CHEMISTRY COMPLEMENT C4 REAGENT) ART.03060770-1 PIECES; CALIBRATOR ADVIA CHEMISRTY CHOLESTEROL (HDL / LDL CHOLESTEROL CALIBRATOR) (ADVIA CHEMISTRY HDL / LDL CHOLESTEROL CALIBRATOR) ART.00309530-1PCS; CALIBRATOR ADVIA CHEMISRTY SPECIFIC PROTEINS (ADVIA CHEMISTRY LIQUID SPECIFIC PROTEIN CALIBRATORS) ART.07711199-2PCS; ADVIA CHEMISTRY REAGENT FOR DETERMINATION OF FRUCTOSAMINE (ADVIA CHEMISTRY FRUCTOSAMINE REAGENT) ART.04862501-1PCS; CALIBRATOR ADVIA CHEMISRTY LIPOPROTEIN A (ADVIA CHEMISTRY LIPOPROTEIN (A) CALIBRATOR) ART.04865268-1PCS; COUNTRY OF MANUFACTURE GBSIEMENS BRANDMANUFACTURER RANDOX LABORATORIES LTD. (UK) FOR SIEMENS HEALTHCARE DIAGNOSTICS INC. (USA)

UKRAINE
HS-Code
Products
3002120000
1.MEDICAL DEVICE FOR DIAGNOSTICSIN VITRO (CLINICAL LABORATORY DIAGNOSTICS FOR OBTAINING INFORMATION ON PHYSIOLOGICAL OR PATHOLOGICAL CONDITION OF A PERSON) -SERUMIMMUNE: -ARTICLE L2KPR2 IML2000 PROLAKTIN 200T TEST FOR PROLACTIN 1 PACK; -ARTICLE L2KTS6 IML. 2000 3RD GRD. 600T THIRD GENERATION THYROID STIMULATING HORMONE TEST 1 PACK ARTICLE L2KPS2 CAT ? 10380986 IML 2000 PSA KIT 200T IMMULITE 2000 PSA TEST FOR FREE PSA 1 PACK ARTICLE L2KHPG2 IML.2000 H. PYLORI SEMI QUANT 200T TEST FOR DETERMINATION OF IGG ANTIBODIES TO H. PYLORI 1 PACK; -ARTICLE L2KCG2 CAT. ? 10381206 IML2000 HCG 200T TEST FOR DETERMINATION OF HUMAN CHRONIC GONADOTROPIN 1 PACK; FOR DETERMINATION OF N-TERMINAL BRAIN NATRIURETIC PROPEPTIDE IMMULITE 2000 NT-PROBNP 1 PACK; - ARTICLE L2KPS2 CAT. ? 10380986 IML. 2000 PSA KIT 200T TEST IMMULITE 2000 PSA KIT FOR DETERMINATION OF FREE PSA 1 PACK; - ARTICLE L2KDD2 IML 2000 D-DIMER K IT 200T TEST FOR DETERMINATION OF D-DIMER IMMULITE 2000 D-DIMER 1 PACK; -ARTICLE L2KAP2 CAT. ? 10381187 IML.2000 AFP KIT 200T TEST FOR DETERMINATION OF ALPHA-FETOPROTEIN 1 PACK; -ARTICLE L2KCE2 IML. 2000 CEA KIT 200T TEST TO DETERMINE THE ISOFORM OF MB CREATINE KINASE IMMULITE 2000 CK-MB 1 PACK; -ARTICLE L2KPF2 CAT. ? 10380984 IML. 2000 FREE PSA KIT 200T IMMULITE 2000 FREE PSA TEST FOR FREE PROSTATE-SPECIFIC ANTIGEN 1 PACK ARTICLE L2KIN2 IML.2000 INSULIN KIT 200T INSULIN TEST 1 PACK ARTICLE L2KFB2 CAT. ? 10381175 IML2000 FREIES B-HCG 200T CONTROL MODULE OF THE FREE BETA SUBUNIT OF HUMAN CHORIONIC GONADOTROPIN IMMULITE (FREE BETA HCG) 1 PACK; -ARTICLE L2KCO6 IML. 2000 CORTISOL KIT 600T TEST FOR DETERMINATION OF CORTISOL 1 PACK; -ARTICLE L2KCRP2 IML.2000 HIGH SENS CRP KIT 200T TEST FOR DETERMINATION OF HIGHLY SENSITIVE C-REACTIVE PROTEIN 1 PACK; -ARTICLE L2KSH2 IML2000 SHBG 200T TEST FOR DETERMINATION OF GLOBULIN BINDING BINDING HORMONES 1 PACK; -ARTICLE L2KDD2 IML. 2000 D-DIMER KIT 200T TEST FOR DETERMINATION OF D-DIMER IMMULITE 2000 D-DIMER 6 PACK; -ARTICLE L2KRUB2 CAT ? 10381338 IML.2000 RUBELLA QUANT IGG KIT 200T TEST IMMULITE 2000 TOXOPLASMA QUANTITATIVE IGG FOR QUANTITATIVE DETERMINATION OF IGG ANTIBODIES TO THE VIRUS RUBELLA 1 PACK; -ARTICLE L2KFB2 CAT. ? 10381175 IML2000 FREIES B-HCG 200T CONTROL MODULE OF THE FREE BETA SUBUNIT OF HUMAN CHORIONIC GONADOTROPIN IMMULITE (FREE BETA HCG) 4 PACK; -ARTICLE L2KPC2 CAT. ? 10381213 IML2000 PAPP-A 200T EATING WEIGHT ASSOCIATED 2 PACK; -ARTICLE L2KIE2 IML2000 GESAMT IGE 200T TEST FOR DETERMINATION OF THE GENERAL LEVEL OF ANTIBODIES OF THE IGE CLASS 2 PACK; -ARTICLE L2KCRP2 IML.2000 HIGH SENS CRP KIT 200T TEST FOR DETERMINATION OF HIGHLY SENSITIVE C-REACTIVE PROTEIN 4 PACK; -ARTICLE L2CGZ CAT.? 10387051 MMULITE 2000 HCG SAMPLE DILUE 50ML SOLVENT FOR THE TEST FOR DETERMINATION OF HUMAN CHORIONIC GONADOTROPIN 1 PACK; -ARTICLE L2KPR2 IML2000 PROLAKTIN 200T TEST FOR DETERMINATION
3822000000
1.MEDICAL DEVICE FOR IN VITRO DIAGNOSTICS (CLINICAL LABORATORY DIAGNOSTICS FOR OBTAINING INFORMATION ON PHYSIOLOGICAL OR PATHOLOGICAL CONDITION OF A PERSON) -DIAGNOSTIC OR LABORATORY REAGENTS ON A SUBSTRATE AND PREPARED (READY) DIAGNOSTIC OR LABORATORY REAGENT SUBSTRATE (ON DISKS): - ARTICLE 10491445 500 TESTS FOR METHOD OF IN VITRO DIAGNOSTICS IN THE QUANTIFICATION OF ESTRADIOL IN HUMAN SERUM AND PLASMA (HEPARINIZED AND TREATED WITH EDTA) USING THE ADVIA CENTAUR CP SYSTEM. 100 TESTS FOR IN VITRO DIAGNOSTICS IN THE QUANTIFICATION OF ESTRADIOL IN HUMAN SERUM AND PLASMA (HEPARINIZED AND AD-TREATED AD SYSTEM) CENTAUR CP. 1 PACK; -ARTICLE DF37 FLEX CARTRIDGE WITH REAGENTS FOR QUANTITATIVE DETERMINATION OF C-REACTIVE PROTEIN (CRP) FOR BIOCHEMICAL ANALYZER DIMENSION 7 PACK; -ARTICLE DC85 IRON CALIBRATOR (IRON CAL)) FOR BIOCHEMICAL ANALYZER DIMENSION 1 PACK; -ARTICLE DF77 FLEX CARTRIDGE WITH REAGENTS FOR THE QUANTIFICATION OF URIC ACID
3002120000
1.MEDICAL DEVICE FOR IN VITRO DIAGNOSTICS (CLINICAL LABORATORY DIAGNOSTICS FOR OBTAINING INFORMATION ON PHYSIOLOGICAL OR PATHOLOGICAL HUMAN CONDITION) -SERUM IMMUNE: -ARTICLE L2KLH2 TEST FOR LUTEINIZING HORMONE 1 PACK; -ARTICLE L2KPC2 PLASTIC TEST ASSOCIATED WITH 1 PREGNANT WOMAN PACK; -ARTICLE L2KBR2 TEST FOR DETERMINATION OF CANCER ANTIGEN 15-3 IMMULITE 2000 BR-MA / CA 15-3 1 PACK; -ARTICLE L2KGI2 TEST FOR DETERMINATION OF CANCER ANTIGEN CA19.9 1 PACK; -ARTICLE L2KOP2 TEST FOR DETERMINATION OF CANCER CARBOHYDRATE ANTIGEN CA125 1 PACK; -ARTICLE L2KPS2 TEST IMMULITE 2000 PSA KIT FOR DETERMINATION OF FREE PSA 1 PACK; -ARTICLE L2KPF2 TEST IMMULITE 2000 PSA FOR DETERMINATION OF PROSTATE-SPECIFIC ANTIGEN 1 PACK; -ARTICLE L2KTY2 TEST IMMULITE 2000 FOR DETERMINATION OF THYROGLOBULIN PACK; -ARTICLE L2KDS2 TEST FOR DETERMINATION OF DEHYDROEPIANDROSTERONE SULFATE IMMULITE 2000 DHEA-SO4 1 PACK; -ARTICLE L2KAP2 TEST FOR DETERMINATION OF ALPHA-FETOPROTEIN 1 PACK; -ARTICLE L2KRT6 TEST FOR RAPID DETERMINATION OF OF OTROPIC HORMONE 1 PACK; -ARTICLE L2KLH2 TEST FOR DETERMINATION OF LUTEINIZING HORMONE 1 PACK; -ARTICLE L2KE22 TEST FOR DETERMINATION OF ESTRADIOL IMMULITE 2000 ESTRADIOL 1 PACK; IMMULITE 2000 HBSAG TEST FOR DETERMINATION OF HEPATITIS B VIRUS SURFACE ANTIGEN 1 PACK; -ARTICLE L2KIE2 TEST FOR DETERMINATION OF TOTAL LEVEL OF IGE ANTIBODIES 1 PACK; -ARTICLE L2KPF2 IMMULITE 2000 PSA TEST FOR DETERMINATION OF PROSTATE-SPECIFIC ANTIGEN 1 PACK; -ARTICLE L2 PROGESTERONE TEST 1 PACK; -ARTICLE L2KTS6 THIRD-GENERATION TEST FOR THYROID-STIMULATING HORMONE 1 PACK; -ARTICLE L2KAP2 ALPHA-FETOPROTEIN TEST 1 PACK; -ARTICLE L2KPF2 IMMULITE 2000 PSA TEST FOR PROSTATE-SPECIFIC ANTIGEN; -ARTICLE L2KIN2 TEST FOR DETERMINATION OF INSULIN 1 PACK; -ARTICLE L2KCE2 TEST FOR DETERMINATION OF CANCER-EMBRYONIC ANTIGEN IMMULITE 2000 CEA 1 PACK; -ARTICLE L2KFB2 CONTROL MODE UL OF FREE BETA-SUBUNIT OF HUMAN CHORIONIC GONADOTROPIN IMMULITE (FREE BETA HCG) 1 PACK; -ARTICLE L2KCRP2 TEST FOR DETERMINATION OF HIGHLY SENSITIVE C-REACTIVE PROTEIN 1 PACK; -ARTICLE L2KFT46 TEST IMMULITE 2000 FOR DETERMINATION OF FREE T4 1 PACK; -ERT TEST FOR DETERMINATION OF TOTAL LEVEL OF IGE CLASS ANTIBODIES 1 PACK; -ARTICLE L2KE22 TEST FOR DETERMINATION OF ESTRADIOL IMMULITE 2000 ESTRADIOL 1 PACK; -ARTICLE L2KCL2 TEST FOR DETERMINATION OF CALCITONIN 1 PACK; -ARTICLE L2KCE2 TEST FOR DETERMINATION OF CANCER-EMBRYONIC2 ANTIGEN 1 PACK; -ARTICLE L2KPF2 IMMULITE 2000 PSA TEST FOR PROSTATE-SPECIFIC ANTIGEN 1 PACK; -ARTICLE L2KPEP2 TEST FOR DETERMINATION OF C-PEPTIDE 1 PACK; -ARTICLE L2KCRP2 TEST FOR DETERMINATION OF HIGHLY SENSITIVE C-REACTIVE PROTEIN 2 PACK; -ARTICLE L2KIE2 TEST TO DETERMINE THE TOTAL LEVEL OF IGE ANTIBODIES 3 PACK; -ARTICLE L2KUE32 TEST IMMULITE 2000 TO DETERMINE UNCONJUGATED ESTRIOL 1 PACK; -ARTICLE L2KPC2 TEST FOR PREGNANCY-ASSOCIATED PLACENTAL BI

UKRAINE
HS-Code
Products
3002190000
1. REAGENTS DIAGNOSTIC ON THE SUBSTRATE ARE TEST SYSTEMS DESIGNED FOR QUANTITATIVE RAPID DIAGNOSIS OF METABOLIC DISORDERS AND DISEASE DIAGNOSIS USED IN MEDICINE: FINECARE CRP / FINECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSIS OF C-REACTIVE PROTEIN-100PCS. ART.W201FINECARE NT-PROBNP TEST / TEST-SYSTEMFINECARE FOR QUANTITATIVE RAPID DIAGNOSIS OF BRAIN NATRIURETIC PEPTIDE-125PCS. ART.W202 FINECARE CTNI TEST / TEST SYSTEM FINECARE FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF TROPONIN I - 1000 PIECES. ART.W203 FINECARE MYOTEST / FINECA RE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF MYOGLOBIN-375PCS. ART.W204 FINECARE CK-MB TEST / TEST SYSTEM FINECA RE FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF CREATINE GAS MV -375PCS. ART.W205 FINECARE MAU TEST / FINECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSIS OF MICROALBUMINURIA-500PCS. ART.W206 FINECARE HBA1C TEST / TEST SYSTEM FINECA RE FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF GLYCATED HEMOGLOBIN-2000 PIECES.ART.W207 FINECARE AFPTEST / TEST SYSTEM FINECARE FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF ALPHA-FETOPROTEIN (AFP) PSAAA TESTA-25050. / FINECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSTICS OF PROSTATIC SPECIFIC ANTIGEN-375PCSART.W209 FINECARE CYS-C TEST / FINECAR E TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF CYSTATIN C (CYS-C) -125PCSART.W2T FESTCARE TS / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF THYROID TROPIC HORMONE (TSH) - 750 PIECES OF ART. W220 FINECARE SAA / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF SERUM AMYLOID - 50 PIECES. ART.W221. FINECAREBNP / FINECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSIS OF SERUM NATRIURETIC PEPTIDE-125PCS. ART.W222. FINECARE CTNI / NT-PROBNP / FIN ECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSIS OF SERUM TROPONIN I / CEREBRAL NATRIURETIC PEPTIDE-125PCS. ART.W222. FINECARE B-HCG TEST / TEST SYSTEM FINECA RE FOR QUANTITATIVE EXPRESS DIAGNOSIS OF HUMAN CHORIONIC GONADOTROPIN-375PCS. ART.W225 FINECARE T3 TEST / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS TRIO OF DTYRONINE (T3) -125 PIECES.ART.W231 FINECARE VITDTEST / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF VITAMINS D-60 PIECES.ART.W241 FINECARE -FINECARE SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF ESTRADIOL -125PCSART.W247 FINECARETESTOSTERONE / FINEC TEST SYSTEM ARE FOR QUANTITATIVE EXPRESS DIAGNOSTICSTESTOSTERONE -50PCSART.W248 REAGENT PACKAGE FOR BLOOD GAS ANALYZER AND 6 ELECTROLYTES. ART.W451-ZOP4-I FINECARE PT / INR TEST / TEST SYSTEMFINECARE FOR DIAGNOSTICS OF PROTHROMBIN TIME - 96 PIECES. ART. W480 FINECARE APTTTEST / TEST SYSTEM FINECARE FOR DIAGNOSTICS OF THE ACTIVATED PARTIALLY THROMBOPLASTIN TIME - 96 PIECES. ART. W481 TEST - FINECARE F FINECARE SYSTEM FOR DIAGNOSTICS OF FIBRINOGEN - 120 PIECES OF ART. W482 FINECARE ACT TEST / FINECARE TEST SYSTEM FOR DIAGNOSTICS OF THE ACTIVATED TIME OF COAGULATION - 120 PIECES OF ART. W483 FINECARE TT TEST / FINECARE TEST SYSTEM FOR DIAGNOSTICS OF THROMBIN TIME - 120 PIECES OF ART. W484 TEST AMYLASE (BY DRY CHEMISTRY) WONDFO CHEMCAR
3002190000
1. REAGENTS DIAGNOSTIC ON THE SUBSTRATE ARE TEST SYSTEMS DESIGNED FOR QUANTITATIVE RAPID DIAGNOSIS OF METABOLIC DISORDERS AND DISEASE DIAGNOSIS USED IN MEDICINE: FINECARE CTNI TEST / TEST SYSTEM FINECARE FOR QUANTITATIVE RAPID DIAGNOSIS OF TROPONIN I - 1500PCS. ART.W203FINECARE HBA1C TEST / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF GLYCATED HEMOGLOBIN-375 PIECES.ART.W207FINECARE PCT TEST / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF PROCALCITONIN -1100 PIECES. ART.W210 FINECARE CTNI / CK-MB / MYO TEST / TEST SYSTEMFINECARE FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF TROPONIN I / CREATINE KEGASMV / MYOGLOBIN -150PCS. ART.W216 FINECARE CRP / PCT TEST / FINECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSTICS OF C-REACTIVE PROTEIN / PROCALCITON NU-125PCS.ART.W218. FINECARE VITD TEST / FINECARE TEST SYSTEM FOR QUANTITATIVE EXPRESS DIAGNOSTICS OF VITAMIN D - 500 PIECES OF ART. W241 MANUFACTURER GUANGZHOU WONDFO BIOTECH CO. LTD. FINECARE TRADEMARK. COUNTRY OF PRODUCTION CN.
3002190000
1. REAGENTS DIAGNOSTIC ON THE SUBSTRATE ARE TEST SYSTEMS DESIGNED FOR QUANTITATIVE RAPID DIAGNOSIS OF METABOLIC DISORDERS AND DISEASE DIAGNOSIS USED IN MEDICINE: FINECARE CRP / FINECARE TEST SYSTEM FOR QUANTITATIVE RAPID DIAGNOSIS OF C-REACTIVE PROTEIN-1250PCS. ART.W201FINECARE CTNI TEST / TEST SYSTEM FINECAREFOR QUANTITATIVE RAPID DIAGNOSIS OF TROPONIN I - 1000PCS. ART.W203FINECARE D-DIMER TEST / TEST SYSTEM FINE CARE FOR QUANTITATIVE EXPRESS DIAGNOSTICSD-DIMER-3500PCS. ART.W211MANUFACTURER GUANGZHOU WONDFO BIOTECH CO. LTD.FINECARE BRAND.COUNTRY OF PRODUCTION CN.

UKRAINE
HS-Code
Products
3002109900
1. PRODUCTS OBTAINED ON THE BASIS OF ANIMAL BLOOD FOR DIAGNOSTIC USE: HIGH-DENSITY CHOLESTEROL-C IMMUNO FS (5X20ML / 1X25ML 99 10 021) CAT.?1 3521 -7UP .; C-REACTIVE PROTEIN FS (5X25ML / 1X25ML 99 10 021) CAT.?1 7002-1UP .; RHEUMATOID FACTOR FS (5X25ML / 1X25ML 99 10 021) CAT.?1 7022-1UP .; D-DIMER (2X12ML / 1X8ML 99 10 935) CAT.?1 7268-3UP .; ONE HBA1CFS (2X15ML / 1X10ML / 1X5ML 99 90 380) CAT.?13329-5P .; ONE HBA1C FS (2X15ML / 1X10ML / 1X5ML 99 90 380) CAT.?1 3329-5UP .; ONEHBA1C FS (100 TESTS 99 10 760) CAT.?13329-2UP. CREATINE KINASE-MB FS (4X120 TEST 99 10 921) CAT.?11641-5UP .; PANCREATIC AMYLASE CC FS (4X120 TEST 99 10 921) CAT.?10551-2UP .; URINE ALBUMIN / CSF FS MICROALBUMIN (4X100 TEST 99 10 921) CAT.?10242-2UP .; APOLIPOPROTEIN B FS (4X100 TEST 99 10 921) CAT.?1 7112-2UP .; APOLIPOPROTEIN A1 FS (4X100 TEST 99 10 921) CAT.?1 7102-2UP .; ANTISTREPTOLYSIN O FS (4X100 TEST 99 10 921) CAT. ?1 7012-4UP .; C-REACTIVE PROTEIN FS (4X200TEST 99 10 920) CAT. ?1 7002-6UP .; C-REACTIVE PROTEIN U-HS FS (4X100 T
3002120000
1. OTHER BLOOD FRACTIONS: ALBUMIN IN URINE / CSF FS (MICROALBUMIN) (5X25ML / 1X25ML 99 10 021) CAT. 1 7268-1P .; CALCIUM FS (4X200 TEST 99 10920) CAT.?1 1181-5UP .; ANTISTREPTOLYSIN O FS (4X100 TEST 99 10 921) CAT.?1 7012-1UP .; C-REACTIVE PROTEIN FS (4X200 TEST 99 10 920) CAT. 7001 7002-2 PACK; RHEUMATOID FACTOR FS (4X100 TEST 99 10 921) CAT. 7017022-1 PACK. 10 045) CAT.?1 7170-1UP .; CALIBRATOR FOR CRP (5X2ML 99 10 039) CAT. CAT.?5 9100-1UP .; MULTICALIBRATOR UNIVERSAL TRUCAL U (20X3ML 99 10 063) CAT.?5 9100-5UP .; CONTROL MATERIAL FOR URINE ALBUMIN U / CSF LEVEL 1 (3X1 ML 99 10 046) CAT. ?5 9710-1UP .; CONTROL MATERIAL FOR CRP LEVEL 1 (3X2ML 99 10 045) CAT. 965 9600-2UP .; CONTROL MATERIAL TRULAB L LEVEL 1 (3X3ML 99 10 065) CAT. 905 9020-1UP.; ?5 9000-4UP .; TRULAB PATHOLOGY CONTROL SERUM (20X5ML 99 10 062) CAT. 905 9050- 1UP.
3002120000
1.MEDICAL DEVICES FOR IN VITRO DIAGNOSTICS: REAGENTS THE ACTIVE COMPONENTS OF WHICH ARE IMMUNE SERA AND OTHER BLOOD FRACTIONS: 99 10 935 D-DIMER FS (CAT. ? 1 7268) - 3 PACK.99 10 921 APOLIPOPROTEIN B FS (CAT. ?? 1 7112) - 2 UNITARY ENTERPRISE 99 10 921 APOLIPOPROTEIN A1 FS (CAT. ? 1 7102) - 2 UNITARY ENTERPRISE 10 1020 C-REACTIVE PROTEIN FS (CAT. ? 1 7002) - 2 UNITARY ENTERPRISE.99 10 920 C-REACTIVE PROTEIN UNIVERSAL WITH HIGH SENSITIVITY (CAT. ? 1 7045) - 10 UNITARY ENTERPRISE. 99 10 921 RHEUMATOID FACTOR FS (CAT. ? 1 7022) - 2 UNITARY ENTERPRISE.

UKRAINE
HS-Code
Products
3822000000
1. CERTIFIED COMPLEX LABORATORY REAGENTS AND REFERENCE MATERIALS ARE INTENDED FOR USE IN AUTOMATIC ANALYZERS OF AVH PENTRA AND PENTRA: CRP CAL / CALIBRATOR CRP (C-REACTIVE PROTEIN) - 6 PIECES; HDL CAL / CALIBRATOR HDL (CHOLESTEROL HDL) - 8 PIECES; LDL CAL / LDL CHOLESTEROL CALIBRATOR - 6 PCS; FERRITIN CAL / FERRITIN CALIBRATOR -2 PCS; RF CAL / RF CALIBRATOR (RHEUMATOID FACTOR) -5 PCS; IMMUNO I CONTROL L / H / IMMUNO CONTROL IL / H-14 PCS; ASO 2 CP / ASO 2 CP ( ANTISTREPTOLYSIN) - 20 PIECES; FERRITIN 2 CP / FERRITIN 2 CP - 4 PIECES; CRP CP / CRP CP (C-REACTIVE PROTEIN) - 160 PIECES; HDL DIRECT CP / HDL DIRECT CP (HDL CHOLESTEROL) - 80 PIECES; LDL DIRECT CP / LDL DIRECT WED (LDL CHOLESTEROL) - 20 PCS; RF CP / RHEUMATOID FACTOR CP - 40 PCS. TRADEMARK: HORIBA. MANUFACTURER: HORIBA ABX SAS. COUNTRY OF MANUFACTURE: JP
3822000000
1. CERTIFIED COMPLEX LABORATORY REAGENTS AND REFERENCE MATERIALS ARE INTENDED FOR USE IN AUTOMATIC ANALYZERS AVH PENTRA AND PENTRA: FERRITIN 2 CP / FERRITIN 2 CP - 6 PIECES CRP CP / CRP CP (C-REACTIVE PROTEIN) - 40 PIECES TRADEMARK: HORIBA. MANUFACTURER: HORIBA ABX SAS. COUNTRY OF MANUFACTURE: JP
3822000000
1. CERTIFIED COMPLEX LABORATORY REAGENTS AND REFERENCE MATERIALS ARE INTENDED FOR USE IN AUTOMATIC ANALYZERS AVH PENTRA AND PENTRA: CRP CAL / CALIBRATOR CRP (C-REACTIVE PROTEIN) - 10PCS; HDL CAL / CALIBRATOR HDL (CHOLESTEROL LVH) - 12PCS; LDL CAL / CALIBRATOR LDL CHOLESTEROL - 6 PCS; FERRITIN CAL / FERRITIN CALIBRATOR -6 PCS; ASO 2 CAL / ASO 2 CALIBRATOR (ANTISTREPTOLYSIN) - 12 PCS; RF CAL / RF CALIBRATOR (RHEUMATOID FACTOR) -10 PCS .; IMMUNO II CONTROL L / H / IMMUNO CONTROL II L / H- 4PCS; LOW CRP CONTROL / CONTROL CRP LOW LEVEL (C-REACTIVE PROTEIN) -3 PCS; ASO 2 CP / ACO 2 CP (ANTISTREPTOLYSIN) - 20PCS; FERRITIN 2 CP / FERRITIN 2 CP- 5 PCS; CP / CRP CP (C-REACTIVE PROTEIN) - 40PCS HDL DIRECT CP / HDL DIRECT CP (HDL CHOLESTEROL) - 80PCS; LDL DIRECT CP / LDL DIRECT CP (LDL CHOLESTEROL) - 80PCS; RF CP / RHEUMATOID FACTOR CP - 40PCS. TRADEMARK: HORIBA. MANUFACTURER: HORIBA ABX SAS. COUNTRY OF MANUFACTURE: JP

UKRAINE
HS-Code
Products
3002109900
1. MEDICAL DEVICES FOR IN VITRO DIAGNOSTICS (NOT FOR VETERINARY MEDICINE) (DO NOT CONTAIN NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS): TEST FOR C-REACTIVE PROTEIN CASSETTE 10T / EMB. SERIES FCRP20030002 MOD. FI-CRP -200 PIECES FAST TEST FOR GLYCATED HEMOGLOBIN HBA1C CASSETTE 10 T / EMBANKMENT SERIES FHBA19110001 MOD. FI-HBA-402 -200 PIECES TEST STRIPS FOR GLUCOSE ALLCHEKTM 25 STRIPS IN A TUBE 50 STRIPS IN A PACKAGE - 100 SERIES GLAGS S50-40 UP. FAST TEST FOR COTININE (COT20) CASSETTE 25T / EMBANKMENT. SERIES COT20020009 MOD. DCT-802 -1000 PIECES. QUICK TEST FOR TROPONIN I CASSETTE 10T / EMBANKMENT. SERIES CTNI20020020 MOD. CTI-402 -1000 ??. QUICK TEST FOR TROPONIN T CASSETTE 10T / EMBANKMENT SERIES CTNT20020003 MOD. CTNT-402 -500 ??. RAPID TEST FOR MYOGLOBIN / CREATINE KINASE (KK-MV) / TROPONIN I COMBO CASSETTE 10T / EMBANKMENT. SERIES CMA20020004 MOD. CMA-435 -500 ??. RAPID TEST FOR ROTAVIRUS TEST CASSETTE 25T / EMB. SERIES ROT20020005 MOD. IRO-602 -500 ??. RAPID TEST FOR ADENOVIRUS RESPIRATORY SYNCYTIAL VIRUS AND INFLUENZA A + B COMBO CASSETTE 10T / EMB. SE
3002109900
1. MEDICAL DEVICES FOR IN VITRO DIAGNOSTICS (NOT FOR VETERINARY MEDICINE) (DO NOT CONTAIN NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS): RAPID TEST FOR MONONUCLEOSIS CASSETTE 20T / EMB. SERIES MONO20030007 MOD. IMO-402 -300 PIECES FAST TEST FOR NOROVIRUS / ROTAVIRUS / ADENOVIRUS COMBO CASSETTE 10 T / EMBANKMENT SERIES MVD20030002 MOD. IMVD-645 -200 PIECES FAST TEST FOR ROTAVIRUS TEST CASSETTE 25 T / SET SERIES ROT20030008 MOD. IRO-602 -500 ??. RAPID TEST FOR SYPHILIS TEST CASSETTE 40T / EMB. SYP20030042 SERIES MOD. ISY-402 -1000 ??. QUICK TEST FOR TROPONIN I CASSETTE 10T / EMBANKMENT. SERIES CTNI20030013 MOD.CTI-402 -1000PCS. RAPID TEST FOR SEMI-QUANTITATIVE DETERMINATION OF C-REACTIVE PROTEIN CASSETTE 10T / EMB. CRP20030004 SERIES MOD. CCR-T402 STOCK - CA USA -300 ??. RAPID TEST FOR PROCALCITONIN (PCT) CASSETTE 10T / EMBANKMENT. PCT20030003 SERIES MOD. CPC-402 -200 PIECES. QUICK FOB TEST TEST CASSETTE 25T / EMB. SERIES FOB20030029 MOD. TFO-602 -1000 ??. TEST STRIPS FOR URINE ANALYSIS 3 PARAMETERS (KET GLU PRO) ?100 (100 STRIPS IN A TUBE) SERIES URS20030024 MOD.U031-03
3002190000
1. MEDICAL DEVICES FOR DIAGNOSTICS IN VITRO (NOT FOR VETERINARY MEDICINE) (DO NOT CONTAIN NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS): TEST FOR C-REACTIVE PROTEIN) CASSETTE 10T / SET SERIES FCRP20110003 MOD.FI-CRP-402 -2000PCSTROPONIN AND TEST CASSETTE 10T / SET FCTI20090001 SERIES MOD.FI-CTI-402 -1000PCSQUICK TEST FOR GLYCATED HEMOGLOBINHBA1C CASSETTE 10T \ SET SERIES FHBA20120002 MOD.FI-HBA-402 -500PCSMANUFACTURER CO. LTD. PRODUCTION - CN

UKRAINE
HS-Code
Products
3002150000
1.MEDICAL PRODUCTS FOR DIAGNOSTICS: IMMUNOLOGICAL PRODUCTS PACKAGED IN DOSAGE FORM OR FOR RETAIL TRADE: IN VITRO REAGENT KITS DIAGNOSTIC FOR QUANTITATIVE DETERMINATION OF REAGENTS IN PLASMA SERA IN CLINICAL DIAGNOSTIC MEDICAL LABORATORIES. NOT FOR VETERINARY MEDICINE. INGREDIENTS: ANTIBODY FRAGMENTS ENZYMES SERUMS OF AN ANIMAL ORIGIN.-HDL 80 / HDL 80 (SERIES 2106148) - 10 UNITARY ENTERPRISE; - HDL C 160 / HDL HALL 160 (SERIES 2111084) - 10 UNITARY ENTERPRISE; ; -HDL / LDL CAL / HDL / LDL CALIBRATOR (SERIES 2109017; 2111054) - 10 UNITS; - CHOL 440 / CHOLESTERINE 440 (SERIES 2110004) - 20 UNITS; - HDL PREC 100 / HDL SEDIMENT 100 (SERIES 2108113) - 1 ; -CHOL 5X50 / HOL 5X50 (SERIES 2108037) - 10 UNITS; - CHOL 250 / CHOLESTERINE 250 (SERIES 2110006) - 30 UNITS; - LDH 100 / LDH 100 (SERIES 2108117) - 5 UNITS; - CRP / C-REACTIVE PROTEIN (SRB) (SERIES 2111244; 2110175) - 70 UNITARY ENTERPRISE. KITS OF REAGENTS IN VITRO DIAGNOSTIC FOR QUANTITATIVE DETERMINATION OF REAGENTS IN PLASMA SERUM IN CLINODIAGNOSTIC MEDICAL LABORATORIES. A
3002150000
1. MEDICAL DEVICES: IMMUNOLOGICAL PRODUCTS PACKAGED IN DOSAGE FORM OR FOR RETAIL TRADE: SETS OF REAGENTS IN VITRO DIAGNOSTIC FOR QUANTITATIVE DETERMINATION OF REAGENTS IN PLASMA SERA IN CLINICAL DIAGNOSTIC MEDICAL LABORATORIES. NOT FOR VETERINARY MEDICINE. INGREDIENTS: ANTIBODY FRAGMENTS ENZYMES SERUMS OF ANIMAL ORIGIN.-HDL 80 / HDL 80 (SERIES 2005130) - 3 UNITARY ENTERPRISE; - LDL 80 / LDL 80 (SERIES 2010099) - 5 UNITARY ENTERPRISE; - LDH 110 / LDH 110 (SERIES 2101078) - 5 UNITARY ENTERPRISE; - CRP / C-REACTIVE PROTEIN (CRP) (SERIES 2011158) - 30P .; SETS OF REAGENTS IN VITRO DIAGNOSTIC FOR THE QUANTITATIVE DETERMINATION OF REAGENTS IN PLASMA SERUM IN CLINICAL DIAGNOSTIC MEDICAL LABORATORIES.IN THE COMPOSITION OF ANTISERUM: -CRP READY-TO-USE / C-REACTIVE PROTEIN READY -USE (SERIES 2102184) - 2 UNITS; - ERBA STANDARD PLASMA (STANDARD SERIES 2012011) - 1 UNIT; - ASO CALIBRATOR / ASL-O CALIBRATOR (SERIES 2102030) - 3 UNITS; - RF CALIBRATOR / RF CALIBRATOR (SERIES 2010094) - 3 UNITS; - ASL (O) CONTROL / ASL-O CONTROL (SERIES 2010052) - 3 UNITS; - ASO / ANTISTREPTOLYSIN O (ASL-O) (SERIES 2006005) - 5 UNITS; - CRP CONTROL LOW / SRB CONTROL LOW (SERIES 2011159) - 4 UNITS; - HBA1C CALIBRATOR SET / HBA1C CALIBRATOR (SERIES 2102097) - 1 UNIT; - HBA1C CONTROL HIGH / HBA1C CONTROL HIGH (SERIES 2102147) - 3 UNITS; - HBA1C CONTROL LOW / HBA1C CONTROL LOW (SERIES 2102084) - 3 UNITS; - HBA1C / GLYCATED HEMOGLOBIN DIRECT (HBA1C) (SERIES 2009009) - 25 UNITS; - RF / RHEUMATOID FACTOR (RF) (SERIES 2102040) - 15 UNITS; - RF CONTROL / RF CONTROL (SERIES 2011161) - 3 UNITS; - CRP STANDARD / CRP STANDARD (SERIES 2102183) - 1 UNIT; - CRP CALIBRATOR / PSA CALIBRATOR (SERIES 2102031) - 4 UNITS; - CRP CONTROL HIGH / CRP CONTROL HIGH (SERIES 2006092) - 2 UNITS. MODIFIED IMMUNOLOGICAL PRODUCTS REAGENT KITS IN VITRO DIAGNOSTIC FOR QUANTITATIVE DETERMINATION OF REAGENTS IN BIOLOGICAL MEDIA IN HONEY. LABORATORIES. AS A PART OF SERUM OF A HUMAN ORIGIN: BILIRUBIN STANDARD / BILIRUBIN STANDARD (SERIES 2101123) - 10 UNITARY ENTERPRISE. MANUFACTURER: ERBA LACHEMA SROCOUNTRY OF PRODUCTION: CZ.
3002101000
1. MEDICAL PRODUCTS FOR DIAGNOSTICS: MODIFIED IMMUNOLOGICAL PRODUCTS: SETS OF REAGENTS IN VITRO. DIAGNOSTIC FOR QUANTITATIVE DETERMINATION OF REAGENTS IN PLASMA SERA IN CLINICAL DIAGNOSTIC MEDICAL LABORATORIES. AS A PART OF ANTISERUM: -CRP READY-TO-USE / C-REACTIVE PROTEIN READY- TO-USE (SERIES 2003189) - 1 UNIT; - RF CALIBRATOR / RF CALIBRATOR (SERIES 2003153) - 1 UNIT; - CRP CONTROL LOW / CRP CONTROL LOW (SERIES 2002152) - 1 UNIT; - HBA1C / GLYCATED HEMOGLOBIN DIRECT (HBA1C) (SERIES 2003205) - 5 UNITS; - HBA1C CALIBRATOR SET / HBA1C CALIBRATOR (SERIES 1912016) - 1 UNIT; - HBA1C CONTROL HIGH / HBA1C CONTROL HIGH (SERIES 1912017) - 1 UNIT; - HBA1C CONTROL L HBA1C CONTROL LOW (SERIES 1912018) - 1 UNITARY ENTERPRISE. MANUFACTURER: ERBA LACHEMA SRO COUNTRY OF MANUFACTURE: CZ.
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
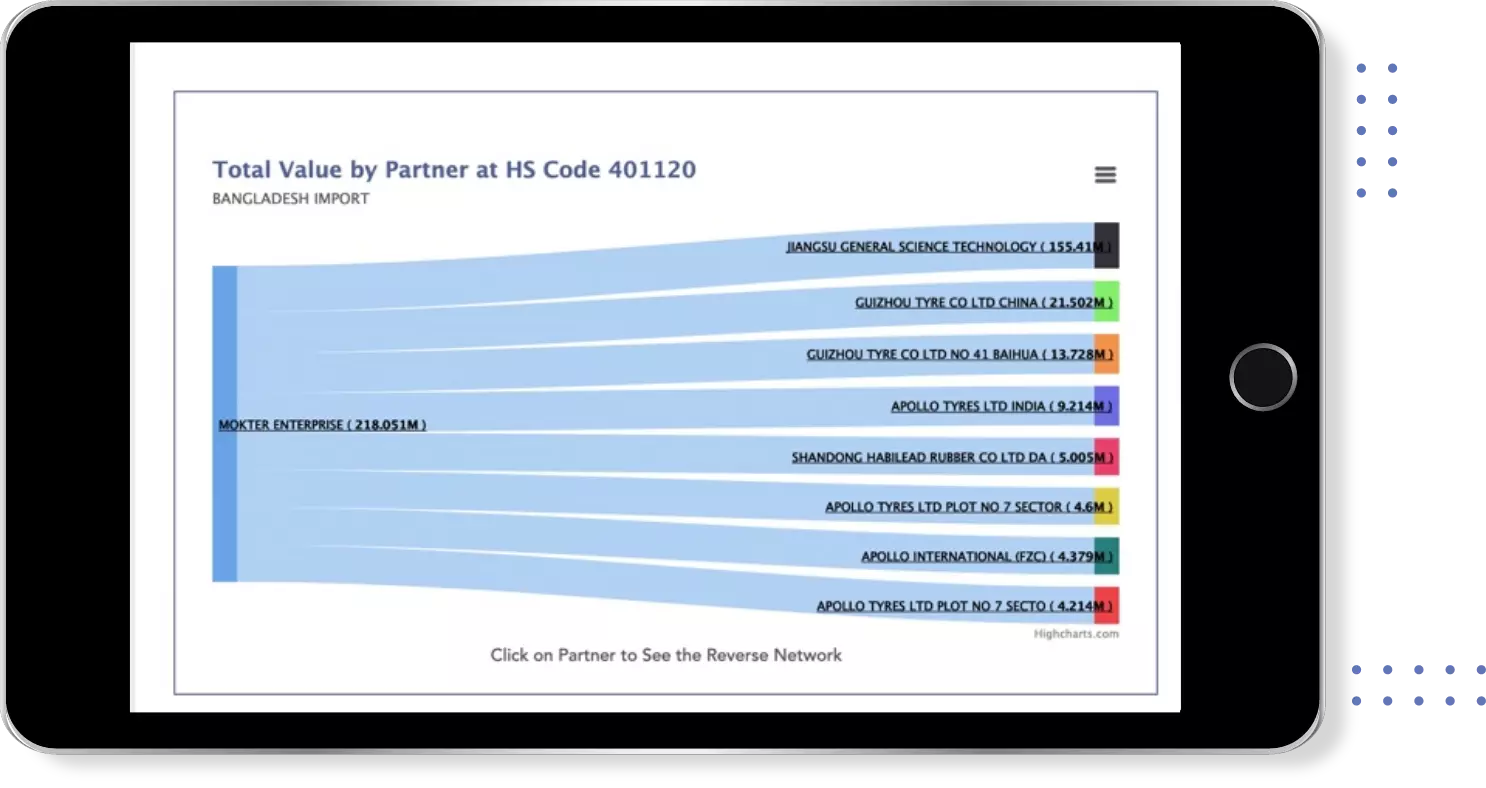
Get most profitable Buyers & Suppliers
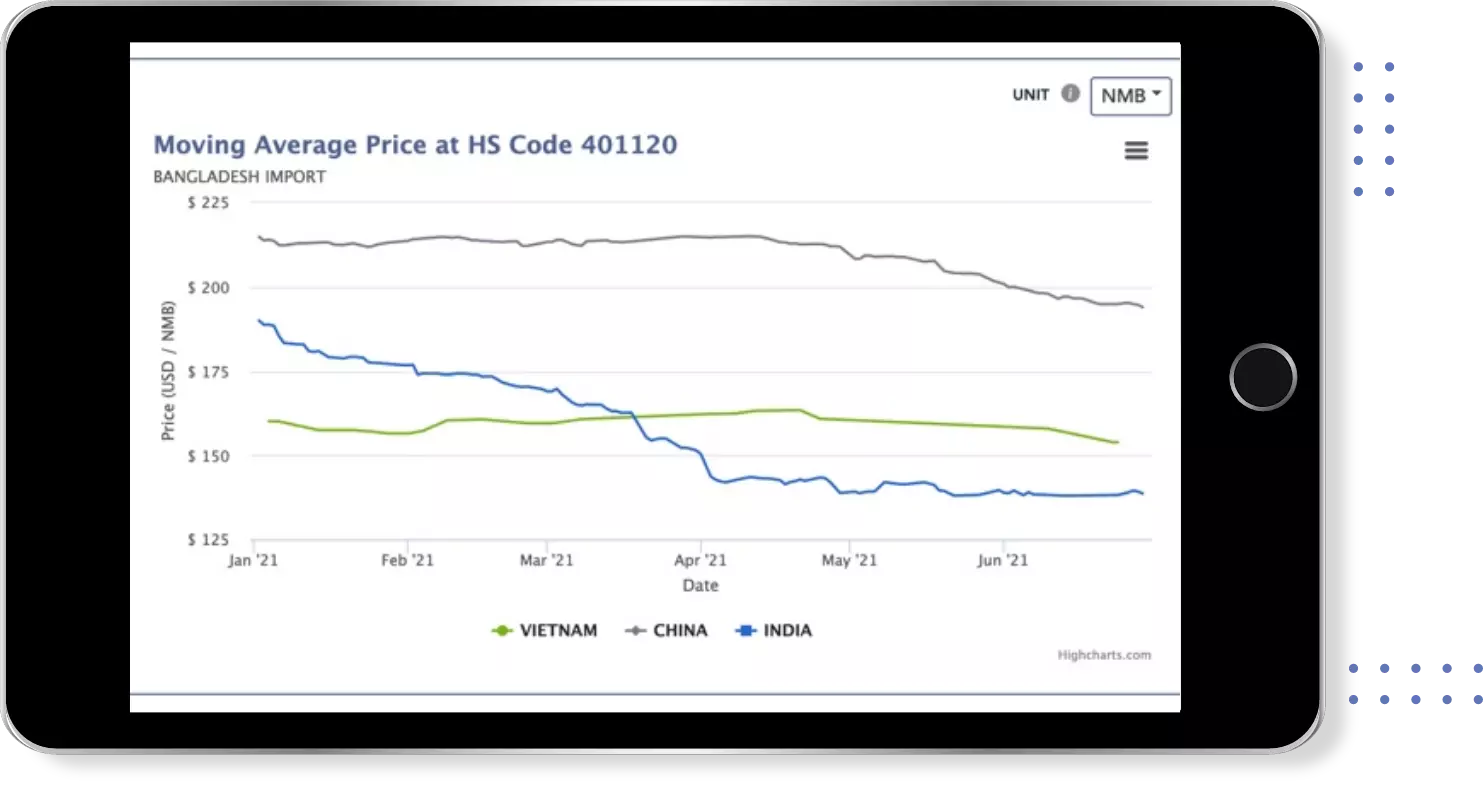
Instant insights on Price , Quantity & Value trends
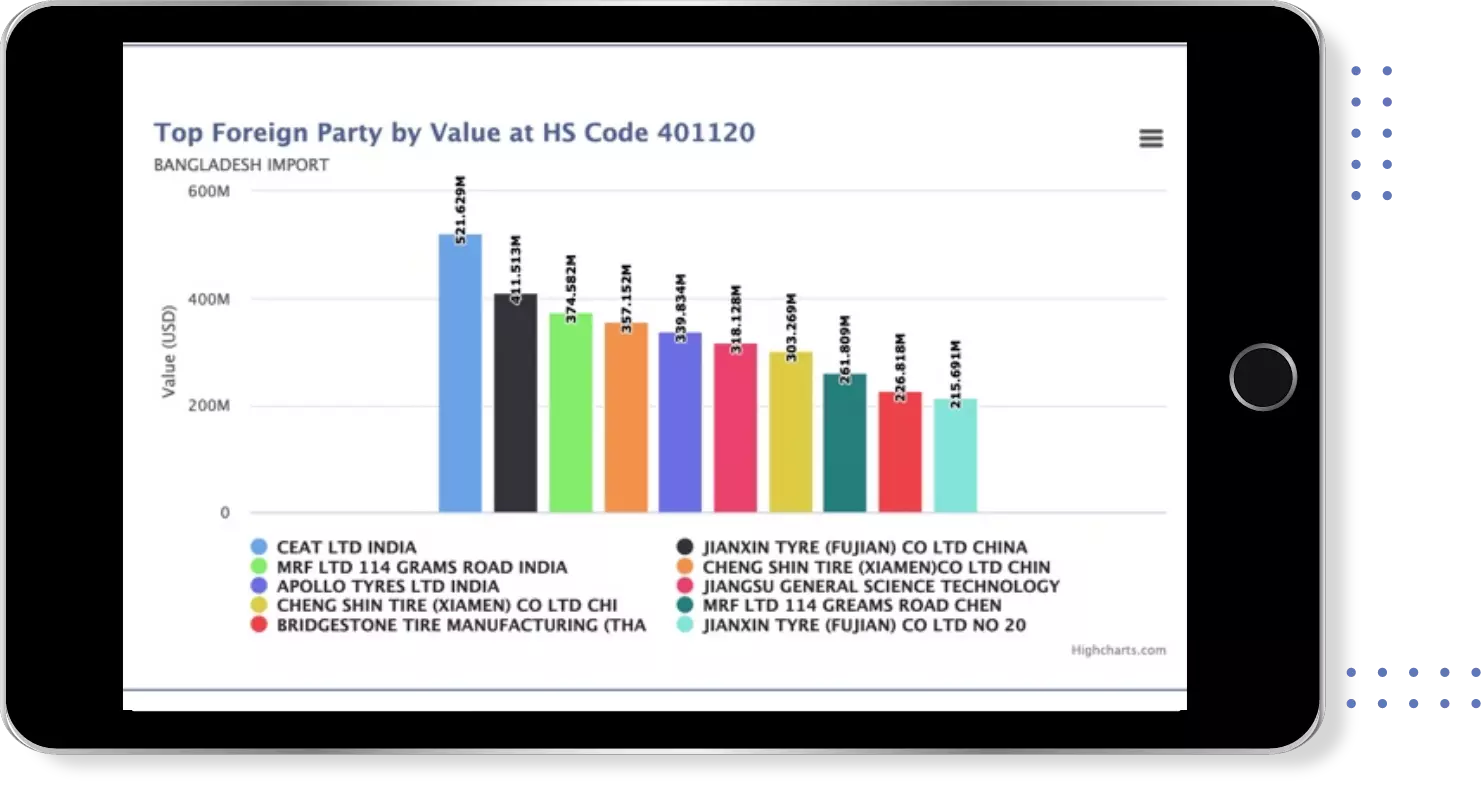
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
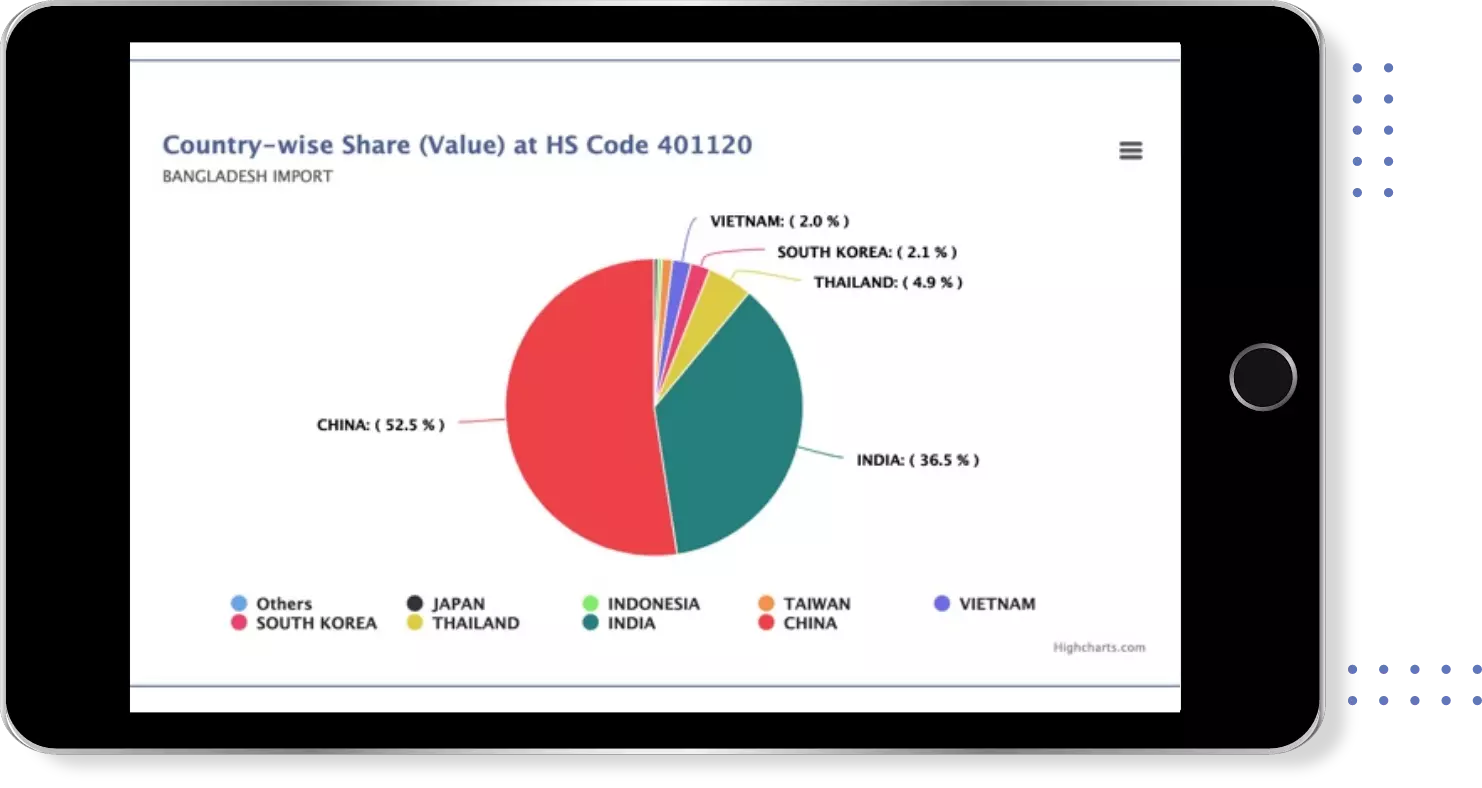
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.