Find verified buyers and sellers of c one injection in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- c one injection buyers in kazakhstan
- COD Kazakhstan
Kazakhstan Buyers of c one injection
LLP MEDAK PHARMA

KAZAKHSTAN
HS-Code
Products
3004900002
1 . METOJECT (METHOTREXATE) SOLUTION FOR INJECTION 50 MG/ML PO 20 MG/0.4 ML IN SYRINGE. ONE SYRINGE IN A BLACK PACK IN A CARDBOARD BOX
3004900002
1 . METOJECT (METHOTREXATE) SOLUTION FOR INJECTION 50 MG/ML PO 20 MG/0.4 ML IN SYRINGE. ONE SYRINGE IN A BLACK PACK IN A CARDBOARD BOX
3004900002
1 . METOJECT (METOJECT) SOLUTION FOR INJECTION 50 MG / ML PO 7.5 MG / 0.1 5 ML IN SYRINGE. ONE SYRINGE EACH IN A BELL PACK IN A CARDBOARD BOX S/Y: 29.06.2024
WEDEL PHARMA LLC

KAZAKHSTAN
HS-Code
Products
3004900002
1. METOJECT SOLUTION FOR INJECT.50 MG/ML - 17.5 MG 1 SYRINGE METOJECT® 50 MG/ML SOLUTION FOR INJECTION 17.5 MG/0.35 ML IN SYRINGE. ONE SYRINGE IN A CONTOUR BALL PACKAGING IN A CARDBOARD BOX. S/Y: 02/20/2025
3004900002
1. METOJECT SOLUTION FOR INJECT.50 MG/ML - 17.5 MG 1 SYRINGE METOJECT® 50 MG/ML SOLUTION FOR INJECTION 17.5 MG/0.35 ML IN SYRINGE. ONE SYRINGE IN A CONTOUR BALL PACKAGING IN A CARDBOARD BOX. S/Y: 03/05/2025
3004900002
1. METOJECT SOLUTION FOR INJECT. 50 MG/ML - 20 MG 1 SYRINGE METODJECT® 50 MG/ML SOLUTION FOR INJECTION 20 MG/0.4 ML PER SYRINGE. ONE SYRINGE IN A CONTOUR BALL PACKAGING IN A CARDBOARD BOX. S/Y: 05/15/2025
TOO ACINO KAZ

KAZAKHSTAN
HS-Code
Products
3004390001
1 . SIZEN® SOLUTION FOR INJECTION 1 2 MG. 3 ML CAPACITY CARTRIDGE OF CLEAR GLASS HYDROLYTIC CLASS I (EF*) SECURED WITH A BROMOBUTYL PISTON ON ONE SIDE AND A BROMOBUTYL DISC AND ALUMINUM COVER WHICH ATTACHES THE DISC TO THE CARTRIDGE ON THE OTHER SIDE. 1 CARTRIDGE IS PLACED IN A PALLET. 1 PALLET WITH 1 CARTRIDGE TOGETHER WITH INSTRUCTIONS FOR MEDICAL USE PRODUCTION DATE: 05/2022 EXPIRY DATE: 04/2024
3004390001
1 . SIZEN® SOLUTION FOR INJECTION 20 MG. 3 ML CAPACITY CARTRIDGE OF CLEAR GLASS HYDROLYTIC CLASS I (EF*) SECURED WITH A BROMOBUTIL PISTON ON ONE SIDE AND A BROMOBUTYL DISC AND ALUMINUM COVER WHICH ATTACHES THE DISC TO THE CARTRIDGE
3004390001
1 . SIZEN® SOLUTION FOR INJECTION 1 2 MG. 3 ML CAPACITY CARTRIDGE FROM CLEAR GLASS HYDROLYTIC CLASS I (EF*) SECURED WITH A BROMOBUTIL PISTON ON ONE SIDE AND A BROMOBUTYL DISC AND ALUMINUM COVER WHICH ATTACHES THE DISC TO THE CARTRIDGE
LLP KAZAKH PHARMACEUTICAL COMPANY MEDSERVICE PLUS

KAZAKHSTAN
HS-Code
Products
3004900002
1 . TENOLIOF LYOPHILISATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION) 20 MG. PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD.
3004900002
1 . TENOLIOF LYOPHILISATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION) 20 MG. PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD.
3004900002
1 . TENOLIOF LYOPHILISATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION) 20 MG. PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD.
TOO RAUZA ADE

KAZAKHSTAN
HS-Code
Products
3004900002
1 . TENOLIOF (TENOXICAM) 20 MG LYOPHILIZATE FOR SOLUTION FOR INJECTION COMPLETE WITH SOLUTION (WATER FOR INJECTION) DRUG IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD. S/Y 02/2025
3004900002
1 . TENOLIOF (TENOXICAM) 20 MG LYOPHILIZATE FOR PREPARATION OF SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION). PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD. S/Y 05/2025
3004900002
1 . TENOLIOF (TENOXICAM) 20 MG LYOPHILIZATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION). PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD. S/Y 1 1 /2025
TOO INKAR

KAZAKHSTAN
HS-Code
Products
3004900002
1 . TENOLIOF LYOPHILIZATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION). 20 MG. PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD. . SERIES 2203663. EXPIRED UNTIL 02. 25.
3004900002
1 . TENOLIOF LYOPHILIZATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION). 20 MG. PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD. . SERIES 2203663. EXPIRED UNTIL 02. 25.
3004900002
1 . TENOLIOF LYOPHILIZATE FOR PREPARING SOLUTION FOR INJECTION COMPLETE WITH SOLVENT (WATER FOR INJECTION). 20 MG. PREPARATION IN A GLASS VIAL. 2 ML SOLVENT IN GLASS AMPOULE. ONE VIAL WITH THE DRUG AND ONE AMPOULE WITH SOLVENT IN A BLACK PACK. ON 3 BELL PACKS IN A PACK FROM CARDBOARD. . SERIES 2203663. EXPIRED UNTIL 02. 25.
TOO SILKROAD TRADING

KAZAKHSTAN
HS-Code
Products
7017900000
1 . KITAZATO MT-INJ-2S55-35 ICSI PIPETTE I.D. DIAM. 5-5.5 ΜM EXT. DIAM. 6-7 MM ANGLE OF 35 ° (IN ONE PACKAGE OF 1 0PC)- GLASS DISHES PIPETTES MADE OF BOROSILICATE GLASS / ARE USED IN LABORATORIES NOT GRADUATED NOT CALIBRATED INJECTION ICIO AND HOLDING BOROSILICATE GLASS PIPETTES ARE USED FOR USED FOR USED FOR IMMOBILIZATION COLLECTION AND INTRODUCTION OF SPERMATOZOIDS INTO EGGS. EXPIRY DATE 25.08.2024 PART NO. 1 08038. MODEL MT-INJ-2S55-35. BRAND KITAZATO. MANUFACTURER KITAZATO CORPORATION JAPAN
7017900000
1 . KITAZATO 95301 MT-HD-1 00-35 PIPETTE HOLDING I.D. DIAM. 1 5-20 MKM EXT. DIAM. 1 00 MM ANGLE OF 35 ° (IN ONE PACKAGE OF 1 0PC)- GLASS DISHES PIPETTES MADE OF BOROSILICATE GLASS / ARE USED IN LABORATORIES NOT GRADUATED NOT CALIBRATED INJECTION ICIC AND HOLDING BOROSILICATE GLASS PIPETTES ARE USED FOR IMMOBILIZATION COLLECTION AND INTRODUCTION OF SPERMATOZOIDS INTO EGGS. EXPIRY DATE 25.01. 2025 PART NO. 201 002. KITAZATO BRAND. MODEL MT-HD-1 00-35 . MANUFACTURER KITAZATO CORPORATION JAPAN
7017900000
1 . KITAZATO 95361 MT-HD-1 20W-35 PIPETTE HOLDING I.D. DIAM. 25-30 MKM EXT. DIAM. 1 20 MM ANGLE OF 35 ° (IN ONE PACKAGE OF 1 0PC)- GLASS DISHES PIPETTES MADE OF BOROSILICATE GLASS / ARE USED IN LABORATORIES NOT GRADUATED NOT CALIBRATED INJECTION ICIO AND HOLDING BOROSILICATE GLASS PIPETTES ARE USED FOR IMMOBILIZATION COLLECTION AND INTRODUCTION OF SPERMATOZOIDS INTO EGGS. EXPIRY DATE 25.08.2024 PART NO. 1 08039. MODEL MT-HD-1 20W-35. BRAND KITAZATO. MANUFACTURER KITAZATO CORPORATION JAPAN
ARIFAR LTD LIABILITY COMPANY

KAZAKHSTAN
HS-Code
Products
3004900002
1 . MEDICINE: TENOXICAM-SELTFAR. LYOPHILIZATE FOR PREPARING SOLUTION FOR INTRAVENOUS AND INTRAMUSCULAR INTRODUCTION COMPLETE WITH SOLVENT. ON 20 MG OF THE DRUG IN A VIAL. 2 ML SOLVENT (WATER FOR INJECTION) IN AMPOULE. 1 VIAL WITH PREPARATION AND 1 AMPOULE WITH SOLVENT IN A CARDBOARD BOX. ONE VIAL CONTAINS: ACTIVE SUBSTANCE - TENOXICAM** 20 MG AUXILIARY SUBSTANCES: SODIUM HYDROXIDE 3.68 MG MANNITOL TROMETHAMOL HYDROGENIC ACID ** - INCLUDING 1 0% EXCESS (V/V) ONE AMPOULE WITH SOLVENT: WATER FOR INJECTION 2 ML RECONQUERATED SOLUTION TENOXICAM CONTAINS 10 MG/ML TENOXICAM
3004900002
1 . MEDICINE: TENOXICAM-SELTFAR. LYOPHILIZATE FOR PREPARING SOLUTION FOR INTRAVENOUS AND INTRAMUSCULAR INTRODUCTION COMPLETE WITH SOLVENT. ON 20 MG OF THE DRUG IN A VIAL. 2 ML SOLVENT (WATER FOR INJECTION) IN AMPOULE. 1 VIAL WITH PREPARATION AND 1 AMPOULE WITH SOLVENT IN A CARDBOARD BOX. ONE VIAL CONTAINS: ACTIVE SUBSTANCE - TENOXICAM** 20 MG AUXILIARY SUBSTANCES: SODIUM HYDROXIDE 3.68 MG MANNITOL TROMETHAMOL HYDROGENIC ACID ** - INCLUDING 1 0% EXCESS (V/V) ONE AMPOULE WITH SOLVENT: WATER FOR INJECTION 2 ML RECONQUERATED SOLUTION TENOXICAM CONTAINS 10 MG/ML TENOXICAM
3004900002
1 . MEDICINE: TENOXICAM-SELTFAR. LYOPHILIZATE FOR PREPARING SOLUTION FOR INTRAVENOUS AND INTRAMUSCULAR INTRODUCTION COMPLETE WITH SOLVENT. ON 20 MG OF THE DRUG IN A VIAL. 2 ML SOLVENT (WATER FOR INJECTION) IN AMPOULE. 1 VIAL WITH PREPARATION AND 1 AMPOULE WITH SOLVENT IN A CARDBOARD BOX. ONE VIAL CONTAINS: ACTIVE SUBSTANCE - TENOXICAM** 20 MG AUXILIARY SUBSTANCES: SODIUM HYDROXIDE 3.68 MG MANNITOL TROMETHAMOL HYDROGENIC ACID ** - INCLUDING 1 0% EXCESS (V/V) ONE AMPOULE WITH SOLVENT: WATER FOR INJECTION 2 ML RECONQUERATED SOLUTION TENOXICAM CONTAINS 10 MG/ML TENOXICAM
MS STOCK LLP (MS STOCK)

KAZAKHSTAN
HS-Code
Products
3002150000
1. HEMLIBRA SOLUTION FOR INJECTION 30 MG/1 ML DRUG IN A BOTTLE OF 1 BOTTLE IN A CARDBOARD BOX SERIES: B0166B16 MANUFACTURE DATE: 06.28.2023 EXPIRATION DATE: 06.30.2025 PACKED IN ONE DISPOSABLE THERMAL CONTAINER
3002150000
1. ACTEMRA SOLUTION FOR HYPOCUTANEOUS INJECTION 162MG/0.9 ML 0.9 ML SOLUTION IN SYRINGE TUBE 4 SYRINGE TUBES IN A CARDBOARD PACK SERIES: B5031B13 PRODUCTION DATE: 04/21/2023 DATE Y ONE: 04/30/2025 PACKED IN A DISPOSABLE THERMAL CONTAINER
3002150000
1. HEMLIBRA SOLUTION FOR INJECTION 30 MG/1 ML DRUG IN A BOTTLE 1 BOTTLE IN A CARDBOARD BOX SERIES: B0171B05 MANUFACTURE DATE: 10/16/2023 EXPIRY DATE: 10/31/2025 PACKED IN ONE DISPOSABLE THERMAL CONTAINER
LTD PARTNERSHIP KNYAZ PHARMA LTD

KAZAKHSTAN
HS-Code
Products
3002410000
1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPHTHERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-341 500 PACKS. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. NOT FOR VETERINARY. DO NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. 1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-85000 PACK. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. MANUFACTURER - SANOFI PASTER S.A. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 1 0 QUANTITY:85000 PCS ; 2. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLLESS) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. SOFTWARE 0. 5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-85000 PACKS. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. MANUFACTURER - SANOFI PASTER S.A. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 1 1 QUANTITY:85000 PCS ; 3. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLLESS) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-85000 PACKS. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. MANUFACTURER - SANOFI PASTER S.A. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 1 2 QUANTITY:85000 PCS ; 4. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARD
3002410000
1 . HEXAXIM VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZA TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZA TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND AGAINST BACTERIAL VACCINES. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. HEXAXIM ® 1 SYRINGE + 2 NEEDLES BATCH: U3E021 V. TOTAL - 30 PACKS. REGISTER CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. PRODUCTION DATE - 23. 02. 2020 / EXPIRY DATE UP TO - 31 . 01 . 2024. VKA DELIVERY ACCORDING TO THE APPENDIX TO THE DELIVERY CONTRACT: 30 DOSES FREE OF CHARGE WITH EACH BATCH SUPPLIED. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
3002410000
1 . HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES SUSPENSION FOR INJECTION. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZA TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. BATCH: U3E021 V. TOTAL - 1 4 PACKS. REGISTER CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. DATE OF PRODUCTION - 23. 02. 2020/ EXPIRY DATE UP TO - 31 . 01 . 2024. NOT FOR VETERINARY. DOES NOT CONTAIN ASPIR T. DELIVERY ACCORDING TO THE APPENDIX TO THE DELIVERY CONTRACT: FREE DOSES TO REPLACE DAMAGED DOSES IN A PREVIOUS SHIPMENT LINKED TO ACCOUNT 901 281 01 DATED 27.01. 2022. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
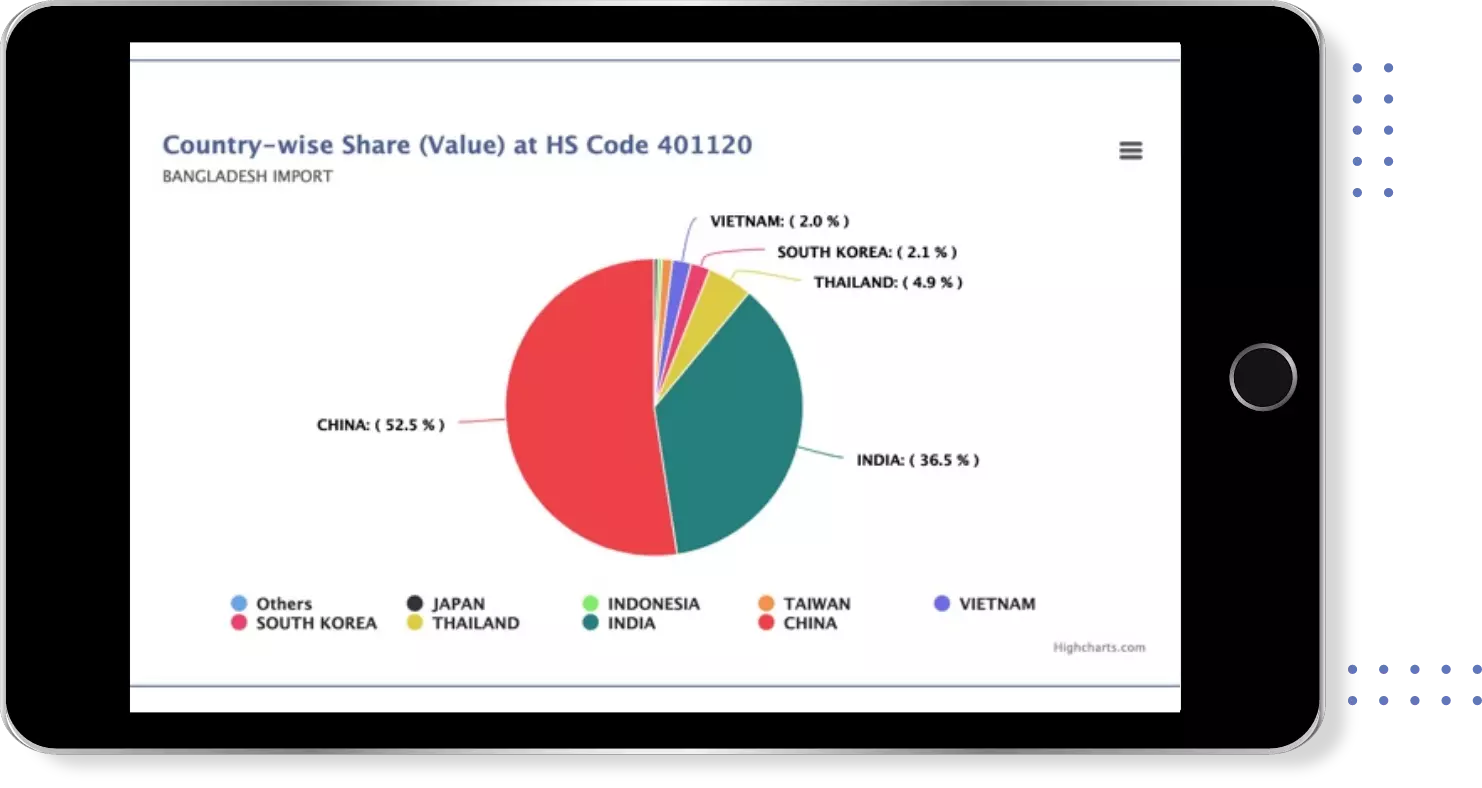
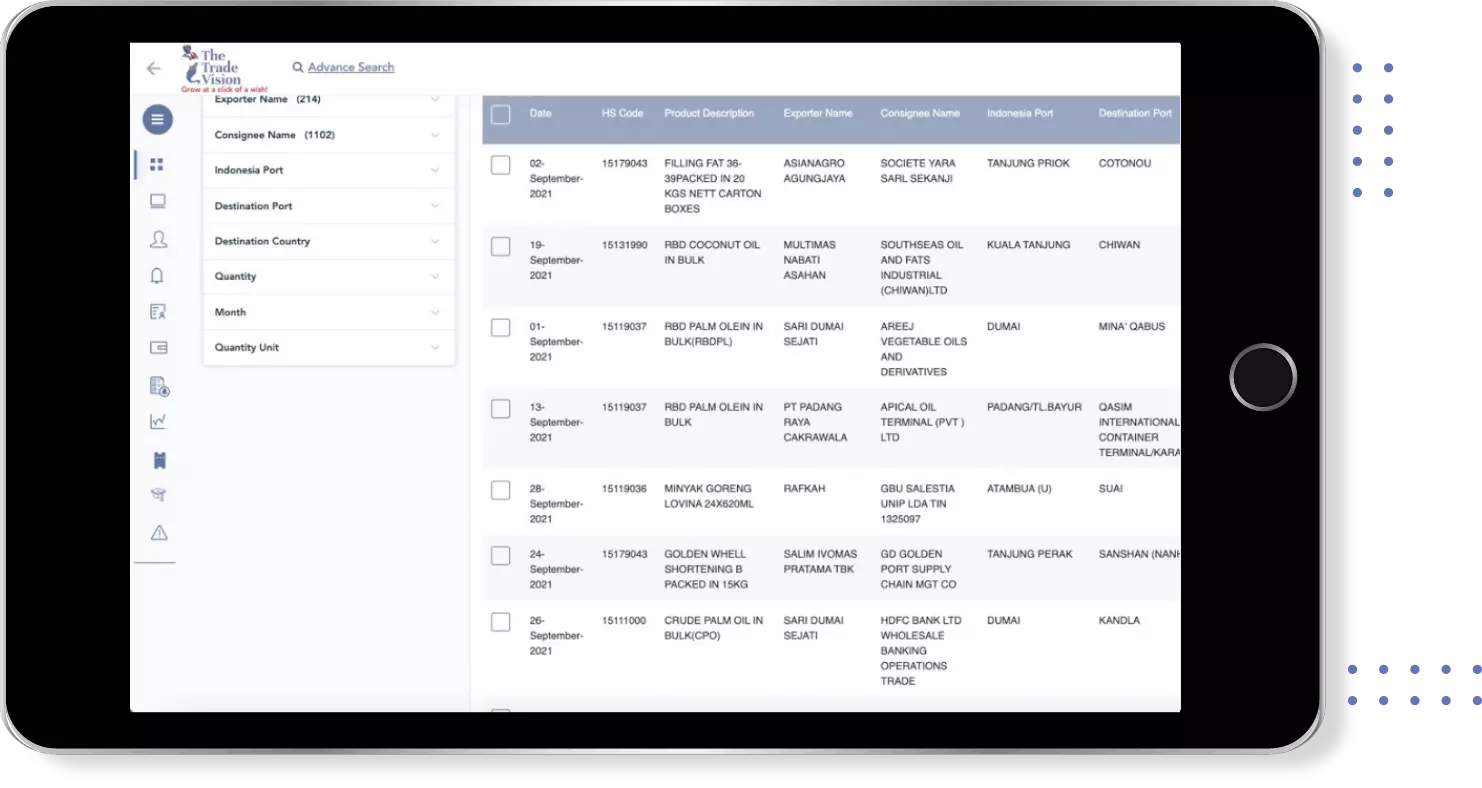
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.